Rhawn Gabriel Joseph, Ph.D.
BrainMind.com
Congenital malformations arise in the central nervous system, more so than any other bodily organ (Norman & Ludwin 2008; Packer, 2-14). It has been estimated that approximately 1% of newborns suffer from major defects and malformations of the central nervous system (Gilles 2008). Although cradled within the comforting confines of the mother's womb, the developing fetal brain is particularly vulnerable to environmental, chemical, nutritional, and stress-related insults, including those in which the mother is physically or emotionally traumatized, thus inducing fetal stress.
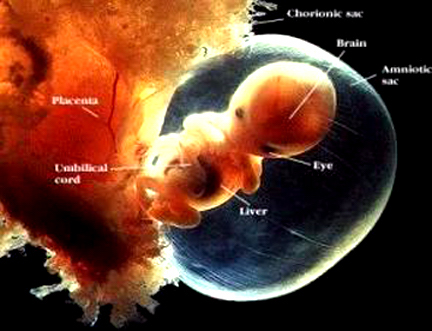
Severe and common congenital disturbances in brain formation are generally attributed to hypoxia, anoxia, lowered pH, cyanosis, and excessive levels of carbon dioxide (Gilles 2008), though they may also be induced by fetal stress. About 12% to 20% of all congenital cerebral abnormalities are due to environmental fators including toxins, maternal infections, drugs and malnutrition (reviewed in Norman & Ludwin 2008) as well as perinatal cerebrovascular accidents.
However, be they physical, chemical, emotional, nutritional, or genetic in origin, depending on the timing and duration of the causative events, lesions to or abnormalities of the developing fetal brain can range in severity and diversity from complex malformations to a simple loss of neurons in a discrete region or layer of tissue. For example, prenatal disturbances of early onset may selectively interfere with forebrain formation due to massive neuronal migration failure. These disturbances may range from anencephaly and microencephaly to more modest anomalies in gyri, sulci and fissure formation (reviewed in Norman & Ludwin 2008), including abnormal limbic system synaptic development.
Disturbances later in the fetal period may instead result in abnormalities confined to the upper layers of the neocortex and/or specific brain regions, such as the frontal, temporal, or inferior parietal lobes, or cerebellum or specific limbic system nuclei. However, like those of earlier onset, these disturbance may be due to abnormalities in neuronal migration, fetal stress, drugs, malnutrition, toxic exposure or direct injury in which case glia scars may also form.
In general, congenital disturbances can be considered of two types, primary and secondary. Primary malformations are due to an intrinsically abnormal developmental process which in turn is related to genetic or chromosomal abnormalities. These genetic abnormalities may result in massive neuronal migration failure, and in the extreme, anencephaly and a complete failure of the forebrain and upper brainstem to develop such that the infant may only possess a spinal cord and caudal medulla (Encha-Razavi 2015). However, it has been estimated that only 1% to 6% of congenital disturbances in brain formation are due to major chromosomal abnormalities (Norman & Ludwin 2008).
Severe and more common congenital disturbances in brain formation are generally attributed to secondary factors, including hypoxia, anoxia, lowered pH, cyanosis, and excessive levels of carbon dioxide (Gilles 2008). About 12% to 20% of all congenital cerebral abnormalities are due to environmental fators including toxins, maternal infections, drugs and malnutrition (reviewed in Norman & Ludwin 2008) as well as perinatal cerebrovascular accidents. These disturbances typically result in massive neural migration failures, the premature arrest of migrating neurons (heterotopias), or their migration to inappropriate substrates, and/or the establishment of inappropriate synaptic connections, atrophy, the loss of or failure to properly myelinate, as well as seizure activity.
It is sometimes difficult to determine if fetal brain abnormalities are due to primary or secondary factors, particularly if an environmental insult was experienced early in fetal brain development. Both primary and secondary congenital abnormalities may be characterized by a retardation in neural growth, a failure to acquire subsequent developmental components such as myelin (Gilles 2008), the establishment of abnormal synaptic interconnections, seizure activity, or the arrest of neurons which cease to migrate and congregate beneath their normal terminal substrate thus forming heterotopic nests.
For example, Galaburda et al. (1983) found islands of heterotopic neurons beneath as well as within the necortex and inferior parietal lobe (IPL) of individuals diagnosed as dyslexic. As the IPL is directly implicated in the ability to read and write and serves as a phonological storehouse (see chapters 11, 20), injury to this region secondary to migration failure, appears to have been causative in these particular cases. In fact, often these congenital abnormalities involve the adjoining auditory cortex, including the planum temporal temporal, thus giving rise to a variety of language disorders and associated abnormalities of thought including "schizophrenia."
In general, the more immature the organism, the more susceptible it is to neurological injury and the more widespread and serious will be the consequences even though not all regions of the CNS mature at the same time. For example, injuries to the CNS after 17 to 18 weeks of gestational age, are usually characterized by the presence of gliosis, macrophages, necrosis, and calcification, whereas those experienced earlier in fetal development may result in microencephaly (Norman & Ludwin 2008).
Hence, abnormalities early in development can result in wide spread cell death and neural migration failure and thus ancencephaly and microencephaly, whereas at the other extreme even functional abnormalities may not be readily detectable and the lesion may be confined to a specific area of the brain (Norman & Ludwin 2008).
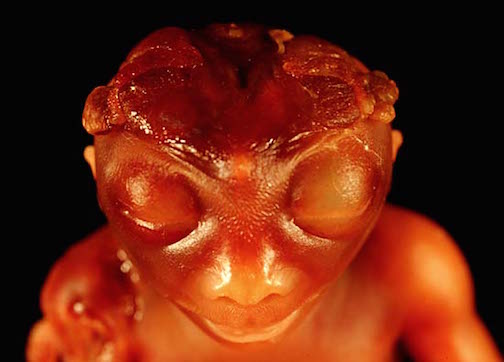
For example, environmental insults experienced late in the third trimester may result in abnormalities confined to the upper layers of the neocortex and hippocampus, and thus focal or lateralized cortical abnormalities, including polymicrogyria, macrogyria, and heterotopias. However, in cases where the upper layers of the neocortex are abnormally affected, given that these layers are made up of interneurons which enable widespread areas of the neocortex to communicate, deficits in multimodal informational analysis and cognitive functioning including disturbances such as schizophrenia (Benes 2015; Weinberger 1987) would be a likely sequalae.
Of course, the brain also remains vulnerable to physical and non-physical insults, including emotional stress, experienced well after birth, including traumas suffered during adulthood (chapter 30). The immature cerebrum is especially vulnerable as the developing brain requires specific types of environmental input and stimulation in order for proper synaptic interconnections to be established and maintained (e.g. Casagrande & Joseph 1978, 1980; Greenough & Chang 1988; Joseph, 1999b). If that "experience-expectant" input is denied early in postnatal development, those areas depending on that input in order to develop normally will be adversely affected.
Unfortunately, be it due to drug exposure, neglect and malnurishment, physical abuse and blows to the head, and so on, in general it is only the more severe disturbances which are likely to come to the attention of a physician (e.g. Courtois 2015; Leestma 2015).
Nevertheless, although the initial deficit may be overlooked even when physicians are in attendance (such as at birth), it is not uncommon for individuals who are traumatized prenatally or during the first few years of life to later suffer from a variety of cognitive, intellectual, behavioral, and emotional problems which are a direct consequence of these cerebral irregularities and injuries (Joseph, 1998b, 1999b).
Moreover, cognitive, intellectual, and emotional abnormalities which are a direct consequence of an early cerebral trauma may not become fully manifest until later in life, because these functions are not required until later in development (e.g. Goldman-Rakic, 1974); at which point these disturbances may be mistakenly assumed as having their onset at this later time period. This includes abnormalities referred to as "autism" and "schizophrenia" (e.g. Benes 2008, 2015; Carpenter et al. 2008; Crow et al. 1989) as well as a variety of "learning disabilities."
Unfortunately, pregnant mothers not uncommonly abuse alcohol or drugs and are sometimes repeatedly beaten and emotionally traumatized by husbands or boyfriends. It is also not uncommon for infants to be shaken, beaten and subject to head injuries and physical and sexual abuse. In either case, be it prenatal or postnatal, the developing brain will be negatively impacted. As these individuals grow older they may suffer from low intelligence, learning disabilities, poor impulse control as well as a host of disturbances involving motor, intellect, cognition, and emotional functioning.
ONTOGENY OF ABNORMAL FETAL BRAIN DEVELOPMENT
Following fertilization, three principle germ layers are rapidly formed, the ectoderm, mesoderm, and endoderm. The mesoderm gives rise to the chordomesoderm and acts to stimulate the ectoderm to form the neuroectoderm and later, the neuroepithelium. The neuroepithelium will give rise to those cells which are to form the neural tube and then the spinal cord and central nervous system.
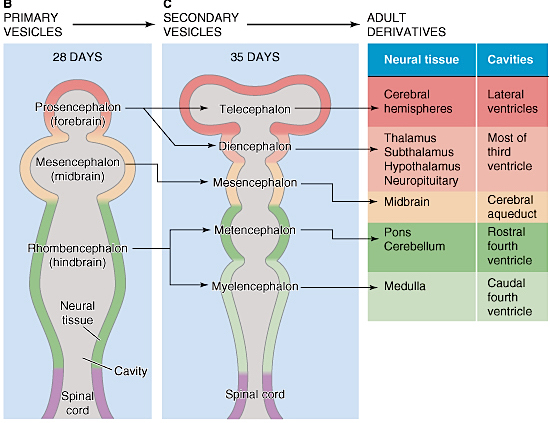
Specifically, at about the fourth week, when the neonate is about 4 mm in length, and affter the neural tube is formed, the internal neural ectodermal tissue, referred to now as the neuroepithelium continues to differentiate and neuroblasts emerge and begin to migrate toward their terminal substrate (Bayer et al. 2015; Sidman & Rakic, 1973, 1982). As neuronal development and migration continues the most anterior half of the neural tube begins to swell in three different places. These three swelling constitute what will become the midbrain (where the optic and auditory lobes will form) the hindbrain (from which the pons and medulla will be fashioned) and the telencephalon/forebrain from which the limbic system, striatum, cerebral hemispheres, and neocortex will form. It is the caudal 50% of the neural tube which eventually becomes the spinal cord.
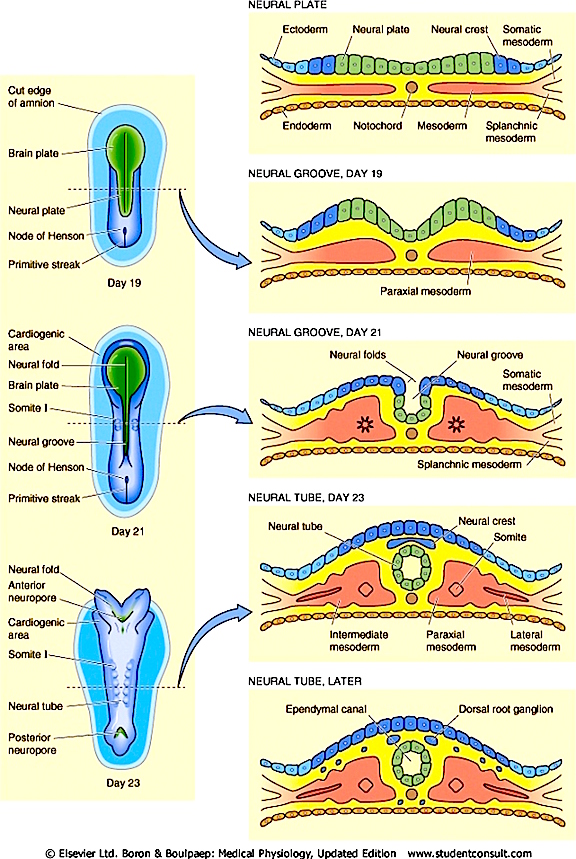
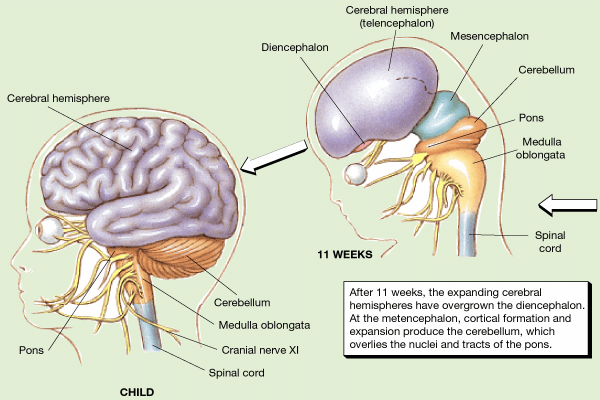
Due to the continued proliferation and migration of neuroblasts, the three vesicles continue to swell, thereby forming five vesicles, the myelincephalon, metencephalon, mesencephalon, diencephalon, and telencephalon. As detailed in chapter 23, the location and different growth stages of the neuroepithelium hearlds and predicts the lateral morphological differentiation and organization of the mature CNS. That is, specific sites within the neuroepithelium give rise to discrete neuronal populations, wave upon wave of which will migrate to and thus form specific brain tissues. According to Bayer et al. (2015, p. 100), "each neuronal population in the central nervous system has three defining characteristics. (1) Each is generated in a specific site in the neuroepithelium. (2) Each one has a unique timetable of neurogenesis that is (3) linked to developmental patterns in other populations with which it will have strong interconnections."
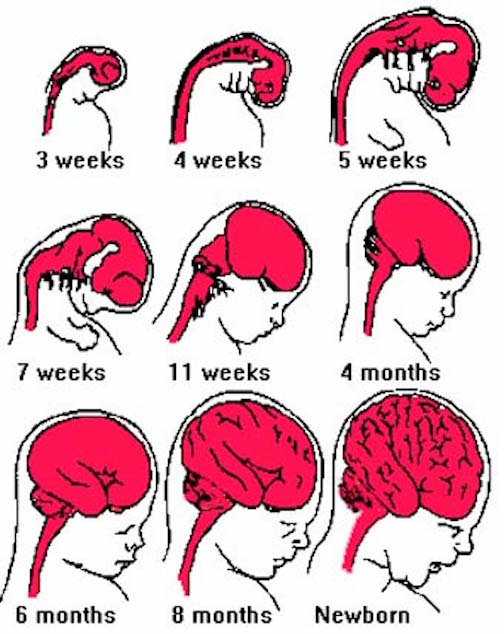
In addition, after the formation of the neural tube, the brain develops in a caudal to rostral arc, such that the brainstem is established in advanced of the forebrain and the limbic forebrain is established in advance of the neocortex. Moreover, different layers of neocortex are established at different time periods and consist of wholly different cells which appear to be generated in different regions of the ventricular neuropetheilium. For example, layers I and VII appear to be formed as an outpouching of that portion of the neural tube associated with the midbrain. By contrast, the neurons which will eventually comprise the layers of the neocortex are predominantly generated in the germinal region of the ventricular surface of the fifth most anterior vessical which corresponds to what will become the telencephalon. Once generated these immature neurons migrate outwards in successive waves toward the surface and insert themselves between layers I and VII, thus forming layers VI, then V, then IV, then III, then last of all layer II (Sidman & Rakic 1973, 1982).
Specifically, migration of the neuroblasts begins after they complete their last mitotic division. They form an association with the glial fibers, and then develop a leading process (what will become a dendrite) which is directed toward the pial surface of the developing brain. They then begin to engage in a crawling-like movement along these glial bridges and thus migrate in successive waves, only to cease their movement temporarily as they wait beneath layer VII. Upon receiving some signal to continue, and/or following the establishment of their terminal substrate and/or following the reception of axons from the thalamus or other cortical/subcortical regions, these neurons resume their migration, sandwiching themselves between layers I and VII.
However, because these and other immature neurons are generated at different time periods, and due to these time dependent relationships, insults to the brain can effect specific neural populations and thus specific regions of the developing brain while seemingly sparing other tissues. For example, if the insult is experienced early during neurogenesis then the precursors to neurons in the germinal matrix are destroyed in vast quantities.
"In cases where neuronal populations are produced over a short time span, a brief, but massive, insult may lead to the permanent decimation of the entire population" (Bayer et al. 2015). Moreover, because the generation of one population of neurons and their migration can then trigger the generation and migration of a second population of neurons, if the first wave are decimated, abnormalities in the second wave will also occur, although the initial trauma, infection, etc., has long passed (Bayer et al. 2015; Rakic 1988).
DISTURBANCES OF NEURONAL MIGRATION: MIGRATION FAILURE & HETEROTOPIAS
In some cases, disorders of neural migration may result in the formation of anomalous and aberrant interconnections between tissues that normally do not directly communicate. Similarly, some neurons may migrate to the wrong region of the brain and establish abnormal interconnections, and/or exert abnormal trophic influences on surrounding tissues. Likewise, if the migrating neurons lose their way or if their movement is prematurely arrested, they may form anomalous groupings in the white matter; i.e., heterotopic nests.
HETEROTOPIAS
Under some conditions the migration of a certain subset of neurons may be delayed such that, in consequence, their terminal and potential synaptic substrate comes to be occupied by other neurons and/or their axons and dendrites. If this occurs, the migration of the appropriate (albeit delayed) neurons may be cut short such that they form anomalous nuclei, beneath their terminal substrates, within the white matter. These displaced nerve cell masses are referred to as heterotopias.
Conversely, in some cases there may result in excessive production of neuroblasts. However, because they are produced in excess there is insufficient cortical space available. That is, all cortical space comes to be quickly occupied such that those neurons that were produced in excess cease their migration while they are still in route toward the neocortex (Roessman 2015). Like their delayed counterparts, presumably these excess cells become isolated within white matter.
There are two types of hetertopia: laminar and nodular (Roessman 2015). The laminar heterotopias are characterized by diffuse, bilateral, sometimes symmetrical gray matter deposits within the white matter. Nodular heterotopias tend to collect near the horns and walls of the lateral ventricles or they may be diffusely scattered throughout the white matter. The convolutions of the hemispheres, however, may appear normal (Norman & Ludwin 2008). In fact, in some cases seemingly "normal" patients are subsequently discovered to have this condition but to have an otherwise normal appearing neocortex (Roessman 2015).
HETEROTOPIAS, TROPHIC INFLUENCES & NEURAL GRAFTS
Although seemingly isolated, and although these displaced heterotopic neurons may form interconnections only with each others (such that they seemingly "talk" only to themselves), it is likely that they exert abnormal trophic influences on adjacent axons that make up the white matter as they pass to and fro between divergent brain areas. Moreover, in many cases, these heterotopic nests may send forth axons and innervate nearby tissues and exert both indirect and direct influences.
Indeed, similar trophic influences and (in some cases) neural interconnections are formed by surgically implanted neural grafts. That is, it has been demonstrated that transplated (grafted) fetal tissue not only survives but exerts trophic influences and, in some cases, forms interconnections with the host tissue (see Lescauldron & Stein 2011; Sinden et al. 2012). Infant brains, however, are more likely to accept such grafts than adult brains, and they are also far more likely to establish interconnections with the grafted tissue.
For example, if somesthetic or visual cortex is implanted into the neonatal midbrain, visual fibers will sprout and innervate the superficial layers of the superior colliculus (which subserves visual functions), whereas the somesthetic fibers will innervated the intermediate layers which subserve movement and somesthesis (Marion & Lund 1987; cited by Lescaudron & Stein 2011). They thus form connections with functionally similar tissues. Moroever, in many instances of fetal transplants in adults brains ravaged by various diseases (e.g. Parkinson's), partial behavioral recovery ensues.
For example, Lindvall et al. (2012) transplanted midbrain fetal dopamine neurons into the putamen in two patients with Parkinson's disease. According to these authors, both patients demonstrated a significant, albeit gradual improvement in symptoms beginning 6 to 12 weeks after surgery; i.e. a reduction in rigidity and bradykinesia predominantly on the side of the body contralateral to the graft. Freed et al. (2011), Madrazo et al. (2011) and others have reported similar findings. Moreover, Lindvall et al. (2012, p. 156) report that transplated DA neurons not only survived, but that "fetal mesencephalic grafts can restore DA synthesis and storage in the denervated human striatum and that this restoration can lead to a significant and sustained improvement in motor function." However, recovery was nevertheless partial.
Although transplated embryonic tissue may survive, the connections established between this and the host tissue does not resemble the original or normal cytoarchitectural organization (Lescaudron & Stein 2011; Stein & Glasier 2012) and instead takes on a disorganized structural arrangement. Moreover, in some instances, wholly inapropriate tissues may be innervated. For example, Marion & Lund (cited Lescaudron & Stein 2011) found that neonatal visual cortex implants into the pons innervated the substantia nigra and even sent axons into the spinal cord.
Therefore, like heterotopias, grafted tissue appear to exert some type of trophic influence on the damaged tissue (Lescaudron & Stein 2011; Sinden et al. 2012), for even when synaptic connections fail to form, these grafts often exert a positive influence on functional recovery and act to reduce or diminish the degree of deficit or disability. In fact, even transplants of purified astrocytes and wound extracts are able to enhance behavioral recovery as much as neural tissue transplants. Hence, possibly the grafted tissue acts to release various hormones or neurotransmitters which act to diffusely influence adjoining tissues. In fact, these influences appear to be exceedingly widespread and can even influence the functional activity in the hemisphere contralateral to the lesion (Lescaudron & Stein 2011).
HETEROTOPIAS, MIGRATION ERRORS, & PSYCHOSIS
Unlike grafted and transplated fetal tissue which can exert positive influences on adjacent and distant brain structures, the trophic influences exerted by heterotopic neural nests appear to be abnormal. Because of these abnormal trophic influences, and/or their abnormal interconnections and abnormal activity they generate (thus influencing long distance axons and adjacent neurons), these nests of ectopic neurons are associated with the development of seizures, epilepsy (Palmini et al. 2008; Spencer et al. 1984), and psychosis (Cannon et al., 2014). That is, the establishment of abnormal interconnections or the presence of these neurons within inappropriate substrates can result in the generation of abnormal activity which leads to seizures, and which can lead to a variety of cognitive and learning related disorders including psychosis; particularly if the temporal lobe is affected (see chapter 21).
For example, Kovelman and Scheibel (1983) report that individuals diagnosed as schizophrenic display heterotopic neurons in the CA1 areas of the hippocampus. Even the offspring of schizophrenics demonstrate similar abnormalities (Cannon et al. 2014) which may well be due to being reared in a psychotic environment, and/or due to an abnormal maternal internal environment during the fetal period -such as viral infection or severe maternal stress.
Nevertheless, as detailed in chapter 28, 30, abnormal hippocampal and temporal lobe activity are frequently associated with epilepsy and psychotic disturbances, including the development of formal thought disorders. In part, this is due to abnormalities affecting the immediately adjacent amygdala (which has the lowest seizure threshold of all brain regions) and the overlying auditory speech areas, such that emotion and personality become disrupted, as does speech and thought formation secondary to disruptions involving the functional integrity of Wernicke's area.
It is noteworthy, however, that different subtypes of schizophrenia are associated with temporal vs frontal dysfunction; e.g., schizoaffective disorder coupled with auditory or visual hallucinations and a formal thought disorder, vs blunted schizophrenia with "negative" symptoms (chapter 11). However, in some cases both the frontal and temporal lobes may be abnormal. For example, Akbarian et al. (1993) have discovered populations of nicotineamide-adenine dinucleotide phosphate-diaphorase neurons abnormally located in the white matter of the frontal and temporal lobes of schizophrenics. Coupled with their 2015 data, Akbarian and colleagues (2015) suggest that these anomalies may be due to neuronal migration errors (Benes, 2015; Weinberger, 1987) or abnormalities involving the cortical subplate during embryonic formation of the brain, perhaps secondary to maternal viral infection (Crow et al. 1989).
Because the cortical subplate serves as the terminal junction for migrating neocortical neurons, abnormalities of early fetal onset, involving the subplate may in turn result in white matter abnormalities and diplaced (subplate) neurons, as well as reduced GAD expression within all seven layers of the neocortex of schizophrenics (see also Lewis 2015). However, given that all seven layers of the neocortex come to be formed, albeit abnormally, also raises the possibility that if due to infection or other environmental insult, this must have occurred late in fetal brain development, after the third trimester (Benes 2015).
That is, if due to environmental factors, these must have been experienced after the necessary neuroblasts were fashioned, perhaps during that phase of migration in which these cells congregate beneath the neocortex and while they were in wait of those trophic and other signals that beckon them to take their final position within one of the layers. This would explain the lack of gliosis or other indications of gross cerebral abnormalities in these particular cases (reviewed in Benes 2015).
Given that it has been discovered that neurons have the capability of undergoing cellular division, particularly those in the hippocampus (Joseph, 1998c,d,e), and that neural division may contribute to columnar organization as well as the layering of the neocortex, it is also possible that these migrating neurons, due to some infectious agent, may be stimulated to divide prematurely; that is, while still migrating or while in wait, thus forming heterotopias that are left behind.
In fact, abnormalities involving neuronal migration have been found to create structural lesions which may be typified by cortical cytoarchitectural disorganization coupled with abnormal laminar and columnar organization (Barth 1987; Rakic 1988).
On the other hand, given that a number of studies indicate significant interneuron reductions in layer II and III whereas the brain is otherwise free of gross cerebral abnormalities such as gliosis indicates that the onset may have occurred well after birth, perhaps during the first few years of life (reviewed in Benes 2015); perhaps as a consequence of genetic programming errors, or severe traumatic stress including insufficient or abnormal mothering (Joseph, 1999b). As detailed in chapter 28, abnormal rearing environments can in fact reduce neural density including neocortical thickness, and result in a massive loss of nerve cells.
As summed up by Benes (2015, p. 105) "studies of schizophrenia have revealed preferential volume loss in the entorhinal cortex, hippocampus, amygdala, globus pallidus... prefrontal, anterior cingulate, primary motor cortices," as well as reductions in neuronal "density in the entorhinal formation, hippocampal formation, medial dorsal thalamus, and nucleus accumbens."
However, as also detailed in chapters 28, 30, stress-induced abnormalities, particularly those affecting the norepinephrine and related neurotransmitter systems, may also induce abnormal neuroplasticity and proliferative growth. In consequence, abnormal interconnections are established between brain regions which normally should not directly communicate.
It is noteworthy that an increase in synaptic interconnections between layer I and II has also been reported in the brains of schizophrenics (Benes 2015). Normally these initial interconnections (which serve to nourish and maintain the cell until its true synaptic counterpart is available) drop out as surrounding neurons and those which are still arriving, begin to differentiate, mature, and grow axons and dendrites. Presumably, layer I neurons maintains these interconnections with layer II and III interneurons either due to the failure of their normal counterparts to successfully complete their migration (thus creating white matter heterotopias), or due to stress-induced neuroplasticity abnormal proliferative growth.
LATE ONSET SYMPTOMS
Schizophrenia has classically been associated with onset in the late teens and early twenties. Not uncommonly, although many of these individuals appear to have their "nervous breakdown" at this early age, it not uncommonly turns out that some of these indivudals were in fact always viewed as odd socially inept or withdrawn by school mates, teachers, siblings, and often parents.
Also, in many cases it is once these individuals leave the structured environment provided by school (including college) and are expected to fend for themselves, that there problems become fully manifest.
This is particularly true of males (e.g. Szymanski et al. 2015). In this regard, it is likely that their underlying problem may well have been of early onset, but due to the routine, structure, and presumably at least semi-supportive structure of their early home environment (though in some cases the home environment is also psychotic), their symptoms were masked, or perhaps existed only as possibility until triggered by stress or some other environmental insult.
NEUROTRANSMITTERS & BRAIN DEVELOPMENT
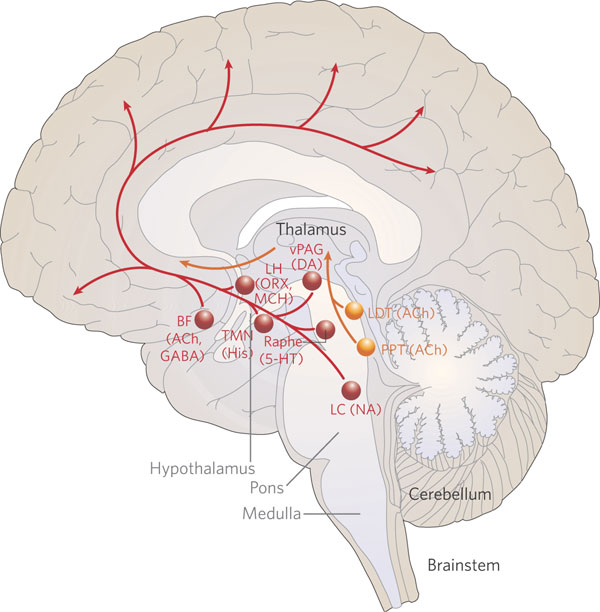
There are many and varied neurotransmitter systems each of which develop and mature in their own distinct and unique ontogenetic patterns in different regions of the brain. For example, depending on brain area, the norepinephrine, dopamine, serotonin, and the cholinergic systems tend to mature late and/or increase in concentration late in prenatal and early in postnatal development (Johnston 1988). However, there are exceptions depending on brain area.

For example, AChE activity in the substantia innominata (nucleus basalis) increases markedly from the 12 to 22nd weeks of fetal development. As the substantia innominata inervates wide areas of the neocortex and in conjunction with AChE, plays a significant role in maintaining neuronal functional viability, the early development and increases in AchE concentration thus appear to be exceedingly important in stimulating neural growth and neocortical formation. By contrast, damage to the substantia innominata and loss of AchE, is associated with wide spread neural degeneration and Alzheimer's disease (see chapter 16).
However, as per the neocortex, AChE, as well as NE and 5HT innervate different layers to different and varying degrees. These neural transmitters also appear to play different roles in maintaining or stimulating neural migration, neural growth, and the formation of the different layers of neocortex.
For example, NE has been shown electrophysiologically to inhibit or suppress irrelevant background activity, while simultaneously enhancing evoked responses in both inhibitory and excitatory afferent neural circuits associated with the processing of relevant environmental input (Foote et al. 1983). Hence, in the developing nervous system, NE may also act to suppress the establishment of irrelevant neural circuits, while simultaneously stabilizing and/or promoting the growth and formation of important synaptic neural networks (see Bear & Singer 1986; Pettigrew & Kasamatsu 1978).
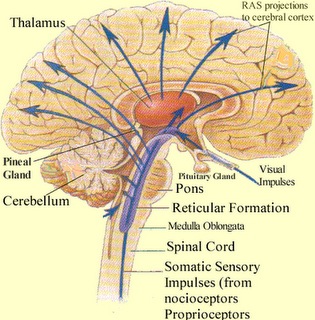
Specifically, NE axons arrive prior to migrating neocortical neurons, and quickly infiltrate neocortical layers I and VII, and then proceed to innervate the cortex in a caudal-ventral-medial to rostral-lateral to caudal-superior-lateral arc. By birth, both cerebral hemispheres have been densely infiltrated by NE axons (Levitt & Moore 1979) and NE levels have reached about 30-40% of adult levels (Johnston 1988). As detailed in chapter 17, the NE which innervates the neocortex is derived from the A6 catecholamine group in the locus coeruleus --a structure which develops well in advance of the neocortex, during the first two months of fetal development.
However, NE as well as 5HT innervation of the neocortex differs depending on brain areas. For example, in area 17, NE appears to be particularly dense in layers III, V and VI. By contrast, 5HT innervation is more extensive in IV (Foote & Morrison 1984). However, in area 18, layer IV contains more NE that 5HT. Because of these differing concentration it has been suggested that NE and 5HT exert some degree of counterbalancing or reciprocal influences on neurological development (Foote & Morrison 1984). For example, whereas both may promote neural growth, if 5HT levels becomes excessive whereas NE levels are reduced (such as in response to stress), abnormal neural circuits may be formed, particularly within the limbic system (see below).
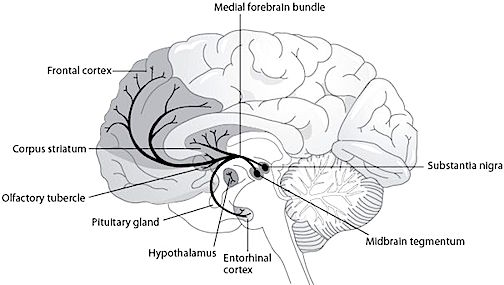
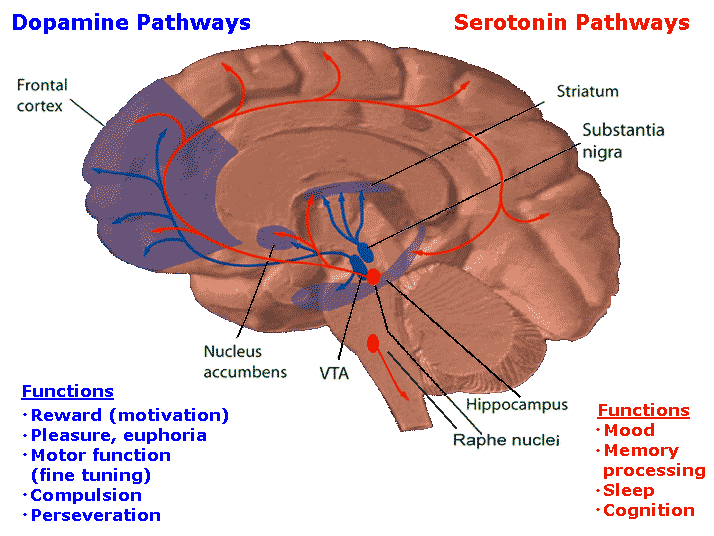
NE INFLUENCES ON NEOCORTICAL DEVELOPMENT
Since NE axons arrive early and quickly infiltrate the various neocortical layers, they can therefore act to influence if not regulate the developmental process including cell migration, neuronal and axonal maintenance, synaptic development, and neuronal differentiation (Parnavelas et al. 1988). Specifically, NE innervation first occurs in layers I and VII. These layers also serve as the site where the first NE synapses are formed; and it is the subplate layer VII (also referred to as layer VIb) where the first non-NE synapses are formed. Moreover, those migrating cells which differentiate first are first innervated by NE axons.
The initial innervation of the neocortex by NE is paralleled by the capacity of these axons to engage in NE reuptake (Levitt & Moore 1979). That is, these NE synapses are active and functional. However, NE neurotransmitter levels, storage capacity and rate of synthesis develops at a rather slower pace (Goldman-Rakic & Brown 1982); a process in primates that continues well after age 3. That is, it takes several years for NE synapses to reach functional maturity, and which, inadvertently predisposes the immature nervous system to becoming easily damaged by emotional stress (see below). Given that these NE synapses form when the capability of storing and synthesizing NE is not yet fully developed, indicates that these axons are not concerned with NE transmission per se. Rather they appear to be more concerned with exerting a trophic influence on migrating neurons, and perhaps regulating the developmental process including early synaptogenesis (Parnavelas et al. 1988). Indeed, this is the major role of cortical layers I and VII.
As noted above, migrating neurons may temporarily cease their journey and wait beneath layer VII. Likewise, thalamic axons also wait beneath layer VII, which suggests that layer VII is exerting some type of trophic influence which both attracts and halts neural migration, perhaps so as to coordinate their arrival with those neurons with which they are to establish synaptic connections. Indeed, thalamic axons appear to be waiting for the arrival of those neurons with which they are to establish synaptic interconnections, and they may establish preliminary synapses with appropriate neurons while they wait.. However, even then this waiting period may persist until until NE synaptogenesis has occurred, at which point these waiting neurons resume their journey and sandwich themselves between layers I and VII, thus successively forming layers VI, V, and so on.
Apparently, NE stimulates cyclic AMP formation in the developing cortex, which in turn regulates protein kinase phosphorylation of intracellular proteins, as well as energy metabolism via glycogenolysis stimulation (reviewed in Johnston 1988). Hence, NE is important in neural growth and neural metabolic activity and exerts an attractive, activating, and nurturing influences on arriving neurons, and thus promotes their growth and the formation of appropriate synapses.
NE plays an important role in neuronal plasticity and in guiding and inhibiting neural development even in non-NE systems. That is, NE acts so as to not only promote the establishment of synapses, but to coordinate their development with the experience-expectant stimulation required for them to thrive; stimulation which may be provided by NE as well as non-NE neurons. However, if that "experience-expectant" stimulation is not provided, abnormal neural circuits may be established, and/or entire populations of neurons may atrophy and die.
For example, if deprived of sensory input (e.g. patterned vision) early in post natal development, neurons and neuronal interconnections become abnormal in those pathways subserving the lost input. Moreover, even when visual input is later provided, the neurons which normally subserve visual perception, will fail to react, such that the animal or human may appear partially blind (Casagrande & Joseph, 1978, 1980; Joseph & Casagrande, 1978, 1980). However, if NE is depleted in advance, then subsequent deprivation has little or no influence on cortical structure (Kasamatsu & Pettigrew 1976; Pettigrew & Kasamatsu 1978) which presumably remains in an immature and plastic state. Thus, these neurons and synapses continue to wait until NE levels build to normal levels and experience-expectant stimulation is provided.
By contrast, if "experience-expectant" stimulation is provided in the absence of NE, there results a profound disturbance in pyramidal neuron morphology, coupled with reduction in cell densities (see Parnavelas et al. 1988). Hence, a complex relationship exist between NE and migrating neurons and the establishment of appropriate interconnections between cells.
FETAL STRESS AND ABNORMAL LIMBIC SYSTEM DEVELOPMENT
If an individual is sufficiently stressed, NE levels may be altered or temporarily depleted (Rosenblum et al., 2014; Southwick et al., 1993; Spoont,2012; Witvliet 1997), which in turn results in abnormal cellular development and/or atrophy (Joseph, 1998b). Likewise, if a pregnant woman is repeatedly subject to emotional and other forms of stress, her brain and that of her fetus may be abnormally effected.
However, these neurological abnormalities are not due to stress-induced NE depletion alone, but to the massive secretion of cortisol, enkephalins, 5HT, dopamine (DA) and other transmitters (Joseph, 1998b). As noted above, 5HT and NE appear to play counterbalancing roles in regard to neurophysiological and inhibitory vs excitatory functioning (see chapter 17). Under conditions of stress, however, 5HT and NE may be differentially effected, such that NE may be depleted. However, in the absence of NE, and under conditions of stress, there is a massive increase in cortical 5HT secretion.
Moreover, in the developing brain, there may also be an increase in 5HT axons which in turn result in increased 5HT secretion and related neuroplastic alterations. If this occurs early in development, this may result in increased 5HT innervation of target and non-target tissue (Blue & Milliver 1987), thus resulting in abnormal synaptic and cellular function and development including the establishment of inappropriate neural connections between tissues which normally do not directly communicate, especially within the limbic system. Indeed, these neurotransmitter fluctuations negatively impact pyramidal neurons, including those within the neocortex, as well as amygdala and hippocampal neurons, axons, dendrites, and their pre and post synaptic substrates (Cain, 2012; Goelet & Kandel, 1986; Kraemer, 2012; Krystal, 2011).
Indeed, stress increases NE turnover in the amygdala (Tanaka, et al. 1982) and increases the production of opiates (Krystal 2011). Opiate receptors are often located presynaptically on NE cells (Llorens et al. 1978), and opiate peptides appear to exert an inhibitory effects on the release of NE (Izquierdo & Graundenz 1980). Hence, there is a counterbalancing relationship between these substances, which, under conditions of prolonged stress may expose developing neurons to the deleterious effects not only of enkephalins in the absence of NE, but cortisol, all of which can significantly and permanently alter the neuronal interconnections, neuronal drop out rate, and thus the functioning of the wide areas of the cerebrum, especially within the amygdala (Cain 2012; Racine 1978) as well as the hippocampus (Joseph, 1998b, 1999d; Lupien & McEwen, 1997; Sapolsky, 1996) and hypothalamus.
For example, since NE also serves a neural protective function, if NE levels are reduced, neurons are exposed to the damaging effects of enkephalins and corticosteroids which at high levels attack and kill pyramidal neurons (Gahwiler 1983; Henriksen et al., 1978; Packan & Sapolsky, 2011), and which can induce neural atrophy (Lupien & McEwen, 1997; Sapolsky, 1996; Uno, Tarara, Else, & Sapolsky, 1989), as well as promote the formation of abnormal synapses.
Even if NE levels remain at normal levels, the stress-induced secretion of massive amount of corticosteroids can induce widespread damage, due in part to their suppressive influences exerted on membrane receptor proteins which are detached from their cellular receptor (Beaulieu, 1987). These conditions alter excitability and information transmission between neurons (Hua & Chen, 1989; Majewska, Harrison, Schwartz, Baker & Paul, 1986) and interfere with messenger RNA protein transcription. Hence, damaged cells cannot be repaired due to DNA/RNA interference, such that, in consequence, limbic system structures such as the amygdala and the hippocampus begin to atrophy or develop abnormal activity, including subclinical seizures, i.e., kindling.
Normally, the release of NE retards kindling in the amygdala. However, with repetitive stress and high levels of arousal, opiates will inhibit NE release, which can lead to permanent structural and functional alterations within infant and adult amygdala neurons, effecting their neocortical interconnections, postsynaptic densities and in the size of the presynaptic terminals as well as their capacity to process and transmit information (e.g. Cain 2012; Racine 1978). With repeated instances of activation or heightened activity, an abnormal form of neuronal plasticity and a lowered threshold of responding also results such that amygdala neurons develop sub-clinical seizure activity, i.e. kindling, which in turn may induce epileptiform after discharges, seizures and convulsions (Cain 2012; Racine 1978).
Moreover, heightened activity within the immature amygdala and associated limbic system neurons, such as the septal nuclei, can induce the growth of additional dendrites which in turn act to attract axons from alternate sites (e.g. Raisman 1969). In consequence, abnormal neural networks and pathways may be formed between nuclei that "normally" do not directly interact. Conditions such as these could predispose the individual to behaving or reacting in an emotionally abnormal fashion, and to processing and even storing information in an abnormal fashion, such that in consequence, the individual is predisposed to developing a host of debilitating emotional and psychotic disturbances (see chapters 2, 28, 30, 31).
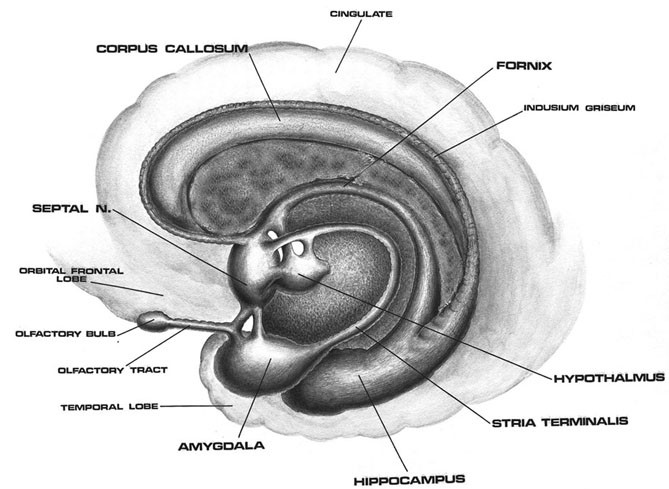
FETAL STRESS AND ABNORMAL SEXUAL DIFFERENTIATION
As detailed in chapter 25, the body and the brain first become sexually differentiated at about the third fetal month. Prior to this age, although genetically male or female, the fetus is physically/sexually-neutral. With the formation of the testes, and the secretion of testicular androgen, target tissues in the spinal cord, brainstem, and cerebrum become activated and transformed into "male" neurons, and form "male" patterns of neural development and neural circuitry, including the pattern of dendritic interconnections between target tissues in the hypothalamus, amygdala, hippocampus, cingulate gyrus, and the bed nucleus of the stria terminals and thus the septal nuclei as well as the amygdala (Bleier et al. 1982; Bubenik & Brown, 1973; Dorner, 1976; Gorski et al. 1978; Nishizuka & Arai, 1981; Rainbow et al. 1982; Raisman & Field, 1971, 1973). It is also well established that the presence (or absence) of testosterone directly effects and determines sexual and cognitive sex differences in mammals including humans (Barnett & Meck, 2011; Beatty, 2012; Collaer & Hines 2015; Dawson et al. 1975; Joseph et al., 1978; Masica et al., 1969; Resnick & Berenbaum 1982).
Presumably, these effects are initially triggered through the differential action of testosterone within target brain areas thus altering neuronal genomic expression (Breedlove 2012; McCarthy 2014 for review). By acting directly on target cell DNA, testosterone (or lack therefore) directly effects neural migration and proliferation vs programmed cell death, and thus cell growth, differentiation, density and myelination (Breedlove 2012; McCarthy 2014; Tobet & Fox 2012). In consequence, a "male" rather than a "female" brain is produced.
The effects of fetal androgens, however, are controlled by a variety of genetic mechanisms which modulate receptor topography and the density of receptor populations which differ during different stages of development and as a function of gender. For example, the genetic sex of the target tissue appears to effect receptor affinity for steroid binding, such as through the secretion of alpha-fetoprotein (Raynaud, Mercier-Bodard & Balieu 1971). These alpha-fetoproteins bind to all circulating estrogens and thus changes their external chemical configuration so that it cannot be recognized by neural receptors (Toran-Allerand 1986). Their chemical configuration is altered and they cannot be taken up by steroid sensitive neurons. Thus these protein act to suppress the influences of these hormones by making them unrecognizable so that they cannot act on the DNA of target neural tissue. Hence, if for any reason there is a failure to produce alpha fetal proteins, females may become masculinized by their own estrogen secretions or the secretions of androgens by the adrenals.
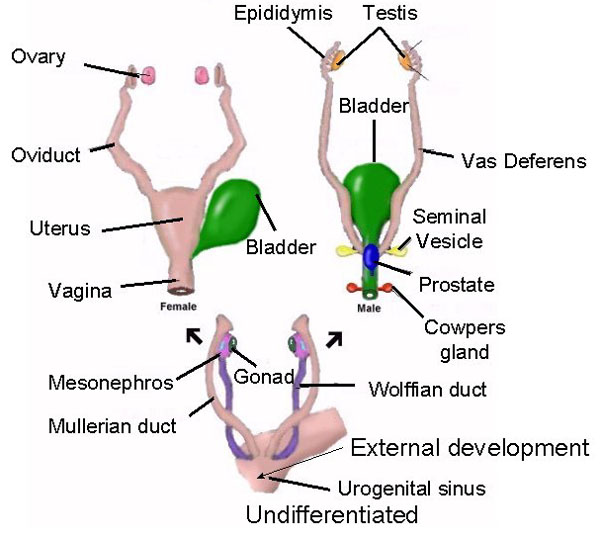
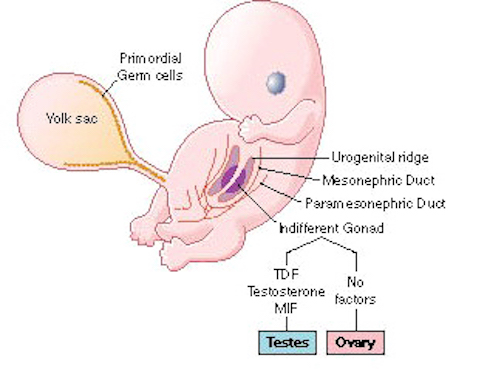
Testosterone however, does not act directly on the genomes of target tissues. Rather, fetal androgens must converted into dihydrotestosterone and into estradiol by an enzyme referred to as aromatase. Aromatase enables fetal androgens to bind to estrogen as well as to testosterone receptors (McCarthy 2014), whereas dihydrotestosterone acts selectively on testosterone receptors. Again, females are protected in this regard by the secretion of alpha-fetoprotein (Raynaud, Mercier-Bodard & Balieu 1971). However, if for any reason these fetal androgens fail to become converted, or if they are prevented from acting on target neural tissue, the female pattern of sexual differentiation will ensue. Although genetically male, the individual may be born with a "homosexual" brain.
Fetal stress may play a significant role in the failure of the male pattern of neural differentiation to unfold, and may well play a significant role in the etiology of homosexuality. Specifically, chronic stress can alter the binding of testosterone, and can prevent testosterone from binding with hypothalamic and amygdala neurons responsible for sexual behavior (Raab & Haedenkamp, 1981). If the fetus is subject to considerable stress, or if "mother" is in an abusive relationship or subject to other profound stress, the secretion of stress-steroids may effect the sexual differentiation and the development of the limbic system and induce the female pattern of neural development. That is, as these stress-steroids, e.g., cortisol and aldosterone, are not true androgens, rather than inducing a male pattern, they may block the reception and binding of fetal testosterone, thereby inducing the female developmental pattern. In fact, the secretion of cortisol and aldosterone--particularly if prolonged--can reduce the secretion of gonadropins (Moberg, 1985), which would also interfere with male sexual behavior and the development of the male pattern of neural development.
As noted, it has been shown that the ventromedial and anterior nuclei of the hypothalamus of male homosexuals demonstrate the female pattern of development (Levay, 2008; Swaab, 2011); sex-specific patterns which may have been induced by stress. If these stress induced steroidal changes also negatively impact the amygdala, affected individuals may not only become homosexual, or sexually and emotionally abnormal, but later engage in self-destructive sexual activities including prostitution and indiscriminate homosexuality (see chapters 13 and 28).
Corticosteroids when secreted at high levels, can in fact induce wide spread neural injury and abnormal neuroplastic changes throughout the brain. Pyramidal neurons in particular are especially vulnerable to the deleterious effects of stress and high levels of corticosteroids, especially those located in limbic system structures due the abundance of Type II adrenal steroid receptors which abound within this tissue (Lupien & McEwen, 1997; Pugh, Fleshner, & Rudy, 1997). Corticosteroids at high levels can exert a suppressive influences on membrane receptor proteins, thereby altering excitability and information transmission between neurons (Hua & Chen, 1989; Majewska, Harrison, Schwartz, Barker, & Paul, 1986), and can detach the cellular receptor from its attached protein (Beaulieu, 1987); a condition which interferes with messenger RNA protein transcription and thus the genetics of neural differentiation and neural plasticity, including, perhaps, those related to the sexual differentiation of the brain.
In fact, the hypothalamus may be particularly at risk as a number of its nuclei are not only sexually differentiated (Allen, et al., 1989; Bleier et al. 1982; Rainbow et al. 1982; Raisman & Field, 1973; Swaab, & Hoffman, 2011) but play different roles in sexual behavior. Moreover, the hypothalamic, pituitary, adrenal system (HPA) is critically involved in the adaption to stressful changes in the external or internal environment, such that the hypothalamus begins to secrete corticotropin releasing factor (CRF) which activates the andenohypophysis which begins secreting ACTH which stimulates the adrenal cortex which secretes corticosteroids (Hakan, Eyle, & Henriksen, 2014; Roozendall, Koolhaas & Bohus, 2012). These events in turn appear to be under the modulating influences of NE. As stress increases, NE levels may be altered or decrease (Bliss, Ailion, & Zwanziger, 1968; Rosenblum et al., 2014; Southwick et al., 1993; Spoont,2012; Witvliet 1997), which triggers the activation of the HPA axis and thus the secretion of corticostereoids which in the male may block the actions of fetal androgens, whereas these same stress-stereoids may induce a mild degree of masculinization in the female brain (e.g. Joseph et al., 1978). That is, these hormones prevent complete masculination in males but do not completely eliminate masculinization as they possibly have mild masculinizing effects. Hence, females may be mildly masculinized whereas males may be feminized by fetal stress.
In addition, since NE also serves a neural protective function, if NE levels are reduced--such as due to chronic or severe stress--the continuing hypersecretion of corticosteroids can profoundly effect the HPA axis (Rots et al., 2015; Suchecki, et . 1993), and neurons located in the hypothalamus, amygdala, and hippocampus (Lupien & McEwen, 1997; Sapolsky, 1996; Uno, Tarara, Else, & Sapolsky, 1989). If the hypothalamus and the HPA axis is injured, the result may be chronic depression and a host of related emotional abnormalities (see Carrol et al., 1976; Sachar et al. 1973; Swann et al. 2014), including a tendency to forever hyper secrete glucocosteroids and to maintain high levels of cortisol even under neutral conditions. Stereotypically, individuals with high cortisol levels tend to become easily depressed, as well as easily overwhelmed by even minimal stress (e.g., Johnson et al., 1996; Kagan, Reznick, & Snidman, 1988). If sufficiently stressed, they may also die (Uno et al. 1989).
In the case of the fetus, they may aborted. If not aborted, they may suffer neurological damage and may be born with a host of congenital abnormalities. In the less extreme, they may instead suffer from a variety of emotional and sexual disturbances, due in large part to the effects of stress on the hypothalamus and amygdala, and in consequence, may later experience considerable confusion over their sexual orientation.
FETAL STRESS, LEARNING DISABILITIES, PSYCHOSIS,
The fetus may experience stress if the mother is stressed, or if she is abusing drugs, alcohol, or eating poorly. Mothers may be stressed due to poverty, poor living conditions, and/or because she is in an abusive relationship. Moreover, the fetus may be stressed simply because it is a "foreign" object, and thus subject to attack by the mother's immune system. Women who spontaneously and repeatedly abort their fetus may well be having these difficulties because of her immune system.
In general, however, if the immune system does react, it is the male fetus which is more likely to be attacked as "foreign" due to its different genetic makeup. In fact, attack by the mother's immune system may well be a factor in the high incidence of congenital and development disorders in males vs famales, and their higher death rate at birth and for the ensuing post-natal months, including dyslexia (4.3 boys/1 girl) autism (4/1), speech and language impairments (2.6/1), learning disabilities (2.2/1), and mental retardation (1.3/1). Moreover, the building up of the mother's immune system over successive pregnancies may explain why late born males are more likely to have developmental disorders.
PRIMARY CONGENITAL ABNORMALITIES IN BRAIN DEVELOPMENT
As noted, congenital abnormalities in brain development have been loosely characterized as being due to primary or secondary factors. However, it is often difficult to distinguish between the two, particularly if secondary factors came into play prior to the 17th week of fetal development.
Primary malformations are generally related to genetic or chromosomal abnormalitie which result in massive neuronal migration failure, such that the forebrain and even the brainstem may only partially develop (Encha-Razavi 2015). About 1% to 6% of congenital disturbances in brain formation are due to major chromosomal abnormalities (Norman & Ludwin 2008).
By contrast, secondary malformation are non-genetic in origin, and may be due to fetal stress, hypoxia, anoxia, lowered pH, cyanosis, and excessive levels of carbon dioxide, toxins, maternal infections, drugs and malnutrition. These disturbances typically result in massive neural migration failures, the premature arrest of migrating neurons (heterotopias), their migration to inappropriate substrates, and/or the establishment of inappropriate interconnections, neural atrophy, and the development of seizure activity.
DEFECTS OF BRAINSTEM, SPINAL & FOREBRAIN DEVELOPMENT
Interference with neuronal maturation or the mass failure of dendrites and axons to form synaptic connections can result in cell death which in turn will effect the formation of the forebrain including gyri and sulci development. In general, the more severe forms of defective neuronal development and migration result in anencephaly, whereas less severe forms allow the forebrain to develop although the convolutional pattern is exceedingly simplified, with only a few primary sulci and fissures present, conditions referred to as lissencephaly and agyria-pachygyria (Norman & Ludwin 2008).
In addition, infection, ischemia, or anoxia may result in cell death and necrosis, which again would effect forebrain and gyral formation; conditions which also result in severe intellectual deficits and/or death. In cases where synaptic failure or necrosis is more subtle or restricted, behavioral, emotional or intellectual abnormalities may also be more subtle.
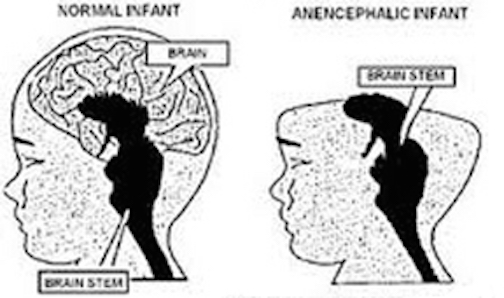
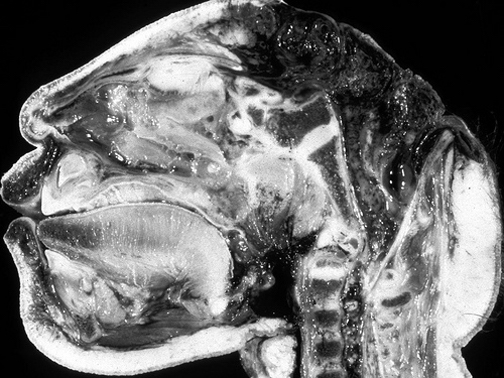
Anencephaly.
Anencephaly occurs in almost one third of all major CNS malformation found at birth (Roessman 2015). Commonly, no CNS tissue is formed or found above the foramen magnum, although the lower brainstem and spinal cord are usually intact. Because the deficit involves the midbrain and brainstem as well as the forebrain, anencephaly cannot be considered a true prosencephaly. Anencephaly is believed to be due to a malformation that has its onset by the 28th day of gestation, and is due to a defect in the closure of the neural tube (Roessman 2015).
There are several subtypes of ancencephaly, such as "cranioschisis" which are characterized by cranial openings and scalp defects, and "rachischisis" which refers to an opening in the spine which leads to destruction and exposure of the spinal cord. These conditions may also occur together. Encephalocele
Encephalocele is characterized by brain tissue herniation outside the cranial vault and is analogous to spina bifida. However, in the vast majority of cases the blow out occurs in the occipital region (Roessman 2015).
Spina Bifida
The most common (i.e. obvious) congenital malformations are spinal tube defects, the least severe of which is spinal bifida occulta. In spina bifida occulta, the underlying spinal cord is essentially normal as instead this condition is due to a failure of the bone to fuse which in turn requires surgical intervention (Roessman 2015).
In general, the majority of lesions occur in the lumbar-thoracic area and is usually due to dysraphias involving the ectoderm and mesoderm. Symptoms may range from mild gait abnormalities to profound sensory and motor deficits.
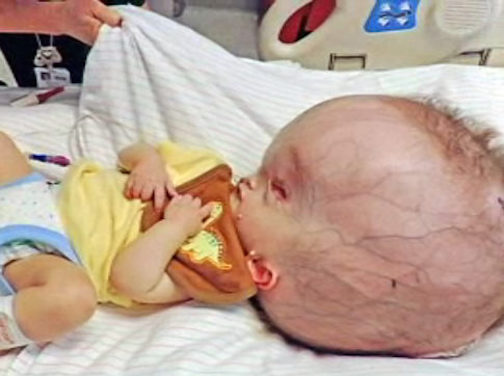
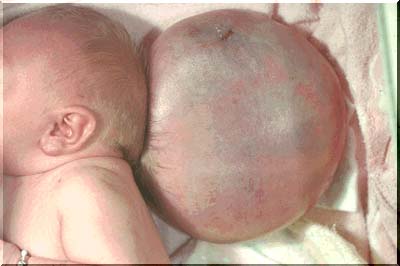
FOREBRAIN GROWTH FAILURE
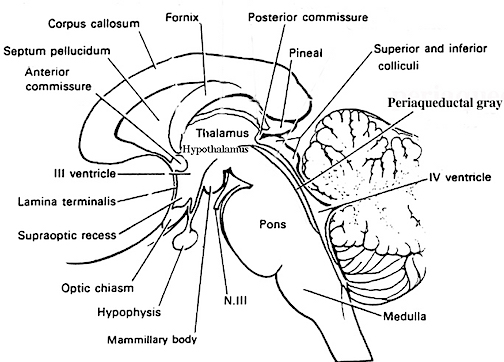
As noted, the forebrain (prosencephalon) first begins to develop around the early part of the 5th week of gestation, and then continues beyond birth. Abnormalities or a failure of the forebrain to develop characteristically result in a wide spectrum of malformations referred to as prosencephalies (Encha-Razavi 2015). The mildest forms of this disorder are associated with an isolated absence of the olfactory bulbs. In fact, agenesis of the olfactory bulbs and olfactory tracts is common to all forms of prosecephalon malformation. Given that much of the forebrain evolved from the olfactory system (see chapter 12), and as originally proposed by this author, it is likely that the olfactory system exerts a trophic influences which promotes brain development and that abnormalities in the olfactory system results in brain atrophy.
In support of this theory, this author found that when neonatal olfactory tissue was injected into lesions of the dorsal hippocampus, that there was evidence of modest neurological and behavioral recovery--as demonstrated on tests of maze learning and retention (Joseph, 1998d). These findings also raised the possibility that hippocampal neurons could be induced to regenerate--which is also a capability demonstrated by olfactory neurons every 4 to 8 weeks (Costanzo & Graziadei, 1987)--and/or that neonatal-olfactory stem cells had been transformed into hippocampal neurons.
Moreover, after removing the olfactory bulbs of adult rats and rat pups between 5 and 10 days of age, and when these animals were sacrificed approximately 45 to 60 days later, both groups (that is, including the pups that survived) demonstrated profound cerebral atrophy in the frontal and temporal-lateral regions. These findings were interpreted as indicating that the olfactory bulbs exert a trophic influence promoting brain development and the functional maintenance of the brain, and that olfactory removals eliminated this growth promoting and maintaining influences, thus resulting in cerebral atrophy (Joseph, 1998d), or, in the case of the developing fetus, prosencephaly.
As noted, the most severe prosencephalies are characterized by anencephaly (in which case most of the brainstem also fails to develop) or profound microencephaly. Related or intermediate disturbances include agenesis of the corpus callosum and anomalies of the septal nuclei and septum pellucidum. These disorders are also associated with a failure of the telencephalic vesicles to form which results in a single anterior ventricle (Roessman 2015); i.e. holoprosencephaly.
Prosencephalies are also commonly associated with facial abnormalities such as cyclopia or cleft lip. This is because the prechordal mesoderm which induces forebrain and olfactory bulb development (via influences on the ectoderm) is also associated with facial development. Hence, abnormalities involving this tissue effects both the forebrain and the face. Lissencephaly, Agryi & Micropolygyria.
Individuals suffering from severe congenital malformations of early onset tend to have small brains which in turn are due to an arrest in development and neural migration defects (Roessman 2015). These disturbances occur as early as the 7th week of gestation and may be related to cell multiplication failure, early cell death, and/or dendritic atrophy such as occurs with Down's syndrome (Norman & Ludwin 2008).
Characteristic disturbances include, "agryi," i.e. a defect that occurs around 11-15 weeks of gestation and is characterized by a smooth brain devoid of gyri (also referred to as lissencephaly). Micropolygyria is characterized by multiple smooth and/or small gyri, a condition that is due to developmental arrest at a somewhat later period.
Megalencephaly.
Megalencephaly refers to a wide range of abnormalities which are characterized by increased brain size and excessive brain weight. This condition is usually associated with disorders of neuronal migration. Typically both the white matter and cortex are increased in thickness.
Microencephaly
Microencephaly refers to a smallness in brain size and is usually associated with disorders of cell migration, or with holoposencephaly. Microencephaly may also be due to destructive, toxic, or degenerative lesions. Gyral patterns may range from the exceedingly simplified to hyper-gyral complexity. Microcephaly Microcephaly refers to smallness in head size. However, in determining if the size is abnormal, the most important variable may well be head proportion in relation to other body parts, including overall body length.
In neonates and infants (as well as young children), the cranium adjusts to the brain as it grows. Hence, there is a relationship between brain weight and volume and head circumference in neonates and infants. In consequence, congenital malformations of the brain can significantly effect the size, shape, and growth of the skull (Gilles 2008).
For example, if the child suffers from microencephaly, the overall size of the brain and the skull will be dramatically reduced. If the malformations are predominantly unilateral, then the skull may grow more slowly on that side. If the malformation or growth failure involves the frontal lobes, the frontal cranium will not properly develop and may take on a crumpled or sloped appearance and the frontal lobe will be missing. In addition, the skull may begin to thicken around the areas of malformation.
With lesions acquired later in develoment, due to subsequent atrophy, the close fit between the brain and skull will be lost, and there may be empty spaces, including widened subarachnoids spaces due to the loss of brain tissue (Gilles 2008).
HEMORRHAGE
Hemorrhage is not uncommon in the fetal or neonatal brain (reviewed in Gilles 2008). However, even hemorrhages at the same site and of the same size may have completely different etiologies even if they occur around the same time period. Unfortunately, only a few significant risk factors have been determined: trauma, intrapartum hypertension, and abnormal uterine environment (e.g. prior fetal waste, intrapartum hypertension, maternal gonococcal infection). Hemorrhages in premature infants is not uncommon and is associated with low birth rate (Leestma 2015).
Fetal and neonatal cerebral hemorrhage commonly occur in the leptomeninges, ventricular systems, the germinal matrix overlying the caudate nucleus and its tail, and in the molecular layers of the cerebrum and cerebellum, and in the neocortex of the temporal and parietal lobes (Gilles 2008). In some cases the hemorrhage is secondary to capillary leakage. In consequence, the lateral, third, or fourth ventricles may be abnormally impacted, including the rhombic lip from which will spring the cerebellum.
Given that the germinal matrix of the ganglionic eminence is the embryological source for the development of the striatum, whereas the temporal lobe contains the amygdala, early onset and even microscopic hemorrhages in these areas can induce profound or subtle disturbances in related emotional, cognitive, and motor functioning, and/or place the individual at risk for later developing such disturbances if later environmental insults are experienced.
As noted in chapter 31, the caudate, amygdala/temporal lobe, cerebellum, and increased ventricle size have all been implicated in the pathogenesis of "schizophrenia." Indeed, whereas the amygdala is susceptible to kindling, the striatum (as well as the thalamus) appears to be a common fetal site of glial fibrillary scarring, acute neuronal necrosis, as well as neuronal mineralization. If this occurs, information transfer to the neocortex may be disrupted, whereas cognitive-motor activities may become and remain abnormal.
Hence, neonates can suffer brain injuries which may be quite subtle and which may be later manifested as learning, behavioral, intellectual, or emotional disturbances. In most cases, however, microscopic hemorrhages normally never come to the attention of a physician unless the infant or neonate dies due to some other cause. By contrast, hemorrhages deep in the white matter, particularly those which lie close to the ventricular linings are usually associated with profound cerebral abnormalities and usually result in death (Gilles 2008).
NECROSIS
Usually major cerebral trauma experienced during the early months of gestation result in neuronal death and/or gross cerebral malformations. During these early months, however, there is not the usual buildup of glial fibrils, macrophages, and hypertrophic astrocytes that characteristically follow cerebral injury.
It is only after the sixth fetal month that the brains response to injury begins to increasingly become similar to that of the adult cerebrum.
Therefore, it is generally during the later stages of fetal development (e.g. late second trimester, early third trimester) that trauma may induce cellular and tissue necrosis. Moreover, because different regions mature and develop at different times, the location of the necrosis may indicate the age at which the injury was sustained. This is because mature neurons are more vulnerable than those which are immature to becoming necrotic.
In that the brainstem matures more rapidly than most forebrain nuclei, brainstem (including midbrain and thalamic) neurons, and to a lesser extent, the striatum, tend to show the earliest vulnerabilities. It is not until the end of gestation that cortical and white matter necrosis may be induced in which case etiology can also be deduced.
For example, areas of necrosis which may develop in the white matter are often caused by hypoxia as well as hypotension or infarcts in which case multiple lesions may be discovered, particularly in the frontal and the parietal-occipital-temporal junction (reviewed in Gilles 2008). Lesions of this sort, even if quite mild, would place those infants at later risk for developing learning disabilities involving reading and writing.
CYSTS
Cysts may develop at any time during gestation are are usually located along the gyri and the distribution of the major arteries. Presumably these are a consequence of impaired blood flow and the development of massive necrosis, though in some cases they are due to developmental abnormalities and migration errors involving mantle formation or repair. In general, cysts are frequently found in the brains of children and infants with marked intellectual and neurological disturbances, though some children may be only mildly effected.
WHITE MATTER TEARS
In contrast to the adult brain, the white matter of an infants brain is highly susceptible to tearing, usually in a symmetrical and linear fashion, such as in response to head trauma (Gilles 2008) and whiplash injuries; e.g. shaken baby syndrome (see below). In instances of white matter tears, wide ranging areas of the neocortex, thalamus, and other subcortical structures can be completely disconnected due to the shearing of their axonal interconnections. In consequence, neurons may die, abnormal connections may form, glia scarring may develop, and a host of abnormalities ranging from the mild to the profound may ensue. However, it is usually on those dramatic cases that come to a physicians attention, and those with mild abnormalities may simply go through life suffering, in turn, a variety of mild (to severe) disturbances involving various aspects of behavior, intellect, emotional functioning and cognition.
HYDROCEPHALUS
Congenital hydrocephalus is always secondary to another disease or injury to the brain, including obstruction of the aqueduct of Sylvius which prevents fluid that has formed in the third and lateral ventricular system from passing and flowing to the cerebral subarachnoid space where it is absorbed. This condition is usually discovered within the first few months of life and may be due to congenital malformations, cysts, tumors, space occupying lesions, or failure of the foramina of Luschka and Magendie to develop (the Dandy-Walker malformation), which normally occurs during the 4th to 5th months of gestation.
DANDY-WALKER SYNDROME
Migration errors involving the cerebellum include the "Dandy-Walker Syndrome," which refers to malformation and hypoplasia of the vermis, as well as other hindbrain abnormalities involving the sinuses and ventricles. This conditions is characteristically associated with hydrocephalus with variable degrees of ventricular dilation (Norman & Ludwin 2008). Hypoplasia is often found in trisomy 18 as well as with other malformations (Encha-Razavi 2015).
In brief, the Dandy-Walker syndrome complex includes a wide spectrum of disorders that include hydrocephalus, posterior fossa cysts which are continuous with the fourth ventricle and cystic dilation of the fourth ventricle with absence of the foramina of Luschka and Magendie, and abnormal or partial agenesis of the the cerebellar vermis.
Secondary Congenital Cerebral Disturbances
Environmental agents are major factors in the development of congenital abnormalities. Environmental trauma may trigger a genetic susceptibility which results in congenital disturbances, or it may create discrete or widespread structural lesions.
For example, alcohol (e.g. fetal alcohol syndrome) can induce cerebral, cerebellar and brainstem dysraphia and disorganization, microencephaly, microcephaly, and hydrocelphaly. Similarly, rubella may induce microencephaly, although most viral infections induce destructive lesions including leptomeningeal inflammation, mineralization, and necrosis.
The capacity of the brain is to respond to infections invaders is only slowly acquired and by different regions at different times. Hence, rubella is most destructive when acquired early during development and can induce germinolysis due to infection of the germinal matrix (Gilles 2008).
HUMAN IMMUNODEFICIENCY VIRUS: HIV
Neonates can become infected with the human immunodeficiency virus (HIV) via vertical transmission from the mother. In completed pregnancies, approximately 30 to 70% of infected women are found to have infected their newborns during the fetal period. HIV does not appear to significantly effect pregnancy, however (Kozolowski 2015). Nevertheless, approximately 50% of children with congenital HIV die by 18 months of age (Kozolowski 2015). Unfortunately, only a few neuropathological studies have been conducted on such fetuses and HIV infected infants.
In general, head circumference of the newborn, as well as height and birth weight do not appear to be significantly effected and infected infants appear little different from "normal" infants. Nevertheless, a wide spectrum of abnormalities are associated with fetal HIV infection and these include progressive encephalopathy, significant loss of neurons, abnormal microglia cell proliferation, nonspecific necrosis and hypoxic-ischemic necrosis, myelin abnormalities, vascular lesions, calcification of the basal ganglia, reductions in brain mass, and microencephaly (Encha-Razavi 2015; Kozolowski 2015).
Approximately 38% of HIV infected children suffer from HIV encephalitis and multinucleated giant cells are frequently found in the white matter and deeper layers of the neocortex. These giant cells are also believed to be replication cites for the virus (Kozolowski 2015).
MATERNAL MALNUTRITION
As noted above, the neural tube is devoid of vasculature and thus a blood supply. Its main source of nutrition is amniotic fluid including ependymal cell secretions. It is only around the 5th week, at the time the forebrain begins to be established, that the anterior choroidal artery penetrates the medial ventricular wall and begins to form the vascularized choroid plexus which then begins secreting CSF.
The choroid plexus remains the primary source of nutrition for the developing brain until about the 12th to 16th week of gestation at which point the cerebral vasculature becomes established and takes over the role of providing nutrtion (reviewed in Duckett & Winick 2015; Marin-Padilla 1988b). Hence, there are some periods of overlapping nutritional sources during these various developmental stages.
Fetal nutrition is completely dependent on the mother --her diet is the fetal diet and the fetus has no way of regulating or selecting intake. There is no protective system in the mother or fetus that controls or regulates the amounts and proportions of proteins, carbohydrates and fats necessary for fetal growth (Duckett & Winick 2015). In consequence, deficiencies in the mother's diet can affect fetal development such as by slowing or retarding cellular division, growth, migration, and maturation -a secondary consequence of protein deficiency.
For example, in rats it has been shown that maternal malnourishment can result in a 20% reduction in the synapse to neuron ratio, as well as stunting of dendrites, decreases in cortical density, and abnormal cortical srtatification (reviewed in Duckett & Winick 2015). Myelin formation can also be reduced and significant associations between maternal malnutrition and CNS malformations such as encephalopathy, hydrocephalus, and even spina bifida have been established.
Although there have been authors who have argued or presented evidence indicating otherwise, malnutrition is the primary killer of children and is one of the more important causes of congenital neurological and cognitive abnormalities (Duckett & Winick 2015).
MATERNAL DRUG ABUSE
The ingestion of a wide variety of drugs and neurotoxicants is a common practice among "modern" pregnant women (Bates et al.2015), and this includes both illicit and therapeutic substances. According to Bates et al. (2015), "as many as 90% of pregnant women take one drug and 60% of women use multiple drugs."
Of course, when a pregnant woman takes drugs, her fetus is also exposed to these substances. Indeed, although the placenta is impermeable to compounds in excess of 1000 in molecular weight, most medications have a molecular weight that is less than 500. Hence, the placenta does not act as a barrier to most drugs and neurotoxicants. In addition, the "blood brain barrier" does not develop or mature until after the 28th week of gestation.
Not surprisingly, a small percentage of babies (3%) are born with gross congenital malformations which are secondary to the mother's drug use (Bates et al. 2015). The proportion of those with mild to moderate deficits is currently unknown.
COCAINE
Complications secondary to maternal cocaine abuse include premature labor, fetal death, and spontaneous abortion. Infants born to such mothers tend to suffer major disturbances, including small head size, cerebral infarcts, and abnormal EEGs in up to 40% of those exposed. Severe emotional disturbances, including postnatal irritability have been reported in over 90% of those afflicted (Bates et al. 2015).
HEROIN
Complications secondary to maternal heroin abuse include premature birth and fetal complications of pregnancy, small-for-gestational-age babies, sudden infant death, fetal addiction and withdrawal, and reduced head size. Abnormal sleep patterns and excessive wakefulness, irritability, hyperactivity, and seizures are also common (Bates et al. 2015).
MATERNAL ALCOHOL ABUSE
Drinking as little as 30 ml of absolute alcohol per day is associated with the development of fetal alcohol syndrome (reviewed in Bates et al. 2015). Characteristic features include microcephaly and developmental retardation, hyperactivity, congenital heart defects, restrictions in mobility of hand and elbow joints with a tendency to dislocate the hip, as well as short palperable fissures.
Approximately 32% of infants born to heavy drinkers suffer gross congenital abnormalities, and up to 14% of infants born to moderate drinkers suffer similar disturbances. Of course, it is likely that factors such as poor maternal diet, smoking, frequent falls, etc., are contributory.
MATERNAL TRAUMA
It is not uncommon for pregnant women to fall and/or bump their swollen bellies and thus their fetus against sinks and other objects, sometimes repeatedly on a daily basis; e.g. as they wash or brush their teeth in the morning. In addition, women involved in abusive relationships usually continue to be struck, beat, kicked, etc., while pregnant. Obviously, the fetus would be also affected by these traumas.
In fact, given that it has been demonstrated that newborn infants can recognize (e.g. prefer) the voice of their mother over that of other females, and also demonstrate other "memories" and reactions to auditory stimuli experienced during the late fetal period, even non-physical abuse (yelling, screaming, etc.) and associated maternal stress would likely exert negative influences on development and the functional integrity of the maturing nervous system (see also chapter 28).
There are, however, no statistics as to the incidence of fetal injury and/or subsequent developmental disorders secondary to these physical (and emotional) traumas. On the other hand, it has been reported that approximately 7% to 8% of pregnancies are disrupted or complicated by maternal trauma (Leestma 2015). That is, if sufficient trauma has been received, the result is usually fetal death.
BIRTH TRAUMA
It could well be argued that even under the best and most desirable of conditions, that birth is a traumatic experience. According to Leestma (2015, p. 255), "numerous studies of presumably normal births have revealed a remarkable incidence of sequelae in neonates, which include subarachnoid and retinal hemorrhages, cranial hematomas, and other injuries," e.g. skull fractures, intracranial and extra calvarial hemorrhages, brain contusions, brainstem and spinal cord injuries, and peripheral nerve damage. Skull fractures and temporal lobe injuries are also not uncommon with forceps delivery.
POSTNATAL DISTURBANCES OF BRAIN DEVELOPMENT & FUNCTION
FALLS, HEAD INJURIES & ABUSE
Children and infants may be subject to multiple forms of abuse which range from and include neglect, sexual molestation, beatings, and head injuries (e.g. Courtois, 1988; Kempe & Helfer 1980). Indeed, it is not uncommon for children and even infants to be struck with fists and heavy objects, or thrown to the floor or across rooms, and even swung by the legs so that their head will strike walls and furniture. Of course those infants and children who suffer head injuries also suffer cerebral injuries, including infarcts and hemorrhage (Leestma 2015).
Children and infants who are reared in homes where drug and alcohol abuse or domestic violence is common, are at obvious risk for physical abuse. Moreover, children and infants who are abused, are generally repeatedly abused (Courtois, 2015; Kempe & Helfer 1980; see chapter 28), and thus suffer repeated cerebral and emotional traumas.
Abuse, however, does not occur exclusively in "abnormal" homes. Even a presumably "normal" mother or father may become frustrated by their crying infant and in consequence, strike, violently shake, or even toss it against a wall or floor -inducing significant brain damage in the process; brain damage that may never come to the attention of a physician and which may not be obvious to the parent although years later they may notice their child is intellectually "a little slow."
Head injuries secondary to non-abusive factors are also common in healthy as well as abnormal environments. It is very common for children to fall and strike their head. Falls (or at least alleged falls), in fact, account for up to 50% of all emergency room visits (reviewed in Leestma 2015).
Although it is not uncommon for children in urban setting to fall from buildings, falls down stairways are far more frequent. In more suburban and rural areas, falls are also common regardless of the quality of home life. These include head injuries secondary to infants toppling over while in walkers, or when momentarily left on a table or bed (while being changed).
As stressed by Leestma (2015, p. 250), however, "severe injuries, especially those involving major or multiple skull fractures and/or multiple truncal, or proximal extremity injuries attributed to a fall... demands further investigation and point toward a willful injury and child abuse."
THE BATTERED INFANT
Characteristically, the "classic" battered child is often less than one year of age and in the vast majority of cases the abuse occurs on a regular basis sometimes beginning the very day the infant in brought home from the hospital (e.g. Kempe & Helfer 1980). The range of abuse may include neglect, malnourishment, and sexual and physical trauma resulting in skull fractures, hemorrhages, as well as cuts, lacerations, burns, bites, and tissue damage involving the viscera, genitals and anus. It is not at all uncommon to find bite marks on the bodies of abused infants. Head injuries, however, account for 75% of those cases that come to the attention of authorities (Leestma 2015), and up to 80% of all instances of fatal Unfortunately, even those infants who were initially spared abuse may be subject to repeat physical traumas once they reach the "terrible twos" and/or begin toilet training. Even those who pass through this difficult period unscathed may be increasingly subject to abuse, particularly of a sexual nature as they approach puberty (e.g. Courtois 2015; Kempe & Helfer 1980). As detailed in chapter 28, sexual abuse as well as emotional trauma can induce profound neurological abnormalities involving the hippocampus and temporal lobes.
THE SHAKEN BABY
Not all infants who are abused and suffer significant brain damage receive head injuries. Instead, they may be shaken to death, or shaken sufficiently so as to induce axonal shearing injuries (see chapter 32) as well as midline subdural hematomas. Direct blows to the head tend to produce lateral and basilar hematomas (Leestma 2015). Unfortunately, not uncommonly, a crying, screaming infant may be shaken by a frustrated, immature, enraged caretaker, or even a "wonderful" and devoted mother (or father) who may not even realize the damage they are inducing in their attempt to quiet their baby.
Nevertheless, due to immature muscular development and coordination, and the disproportionate size of the infants head relative to the body, the infant has little control over their head. In consequence when they are shaken the head will whip back and forth which results in rapidly alternating decelerating and accelerating forces such that the brain is subject to shearing forces, axonal injury, subdural hemorrhages, edema, and in some cases, brainstem and cervical spinal injuries (see chapter 32).

For example, when the infant is shaken, due to the various rotational forces and the swishing and swirling of the brain within the hard inner surface of the cranium, the upper layers of the neocortex may be sheared from the brain surface. In addition, long distance axons as well as blood vessels may be stretched, torn, and severed.
Moreover, when the infant is shaken and the head is subject to oppositional and rapidly changing rotational forces, the cerebral hemispheres, brainstem and different parts of the brain will actually move in somewhat different directions as well as at different speeds thus inducing widespread stretching, straining and snapping of axons running throughout the white matter including the corpus callosum (Adams et al., 1981, 1989; Gennarelli, 1986; Zimmerman et al. 2008). Because these strains are not uniform but effect various parts of the cerebrum differently, such that one part of the brain is moving in one direction whereas another portion is sliding at a somewhat different angle, certain regions can literaly snap and break loose.
Not surprisingly, rotational injuries are also associated with venous tears, arterial shearing stresses, and hemorrhagic lesions in the midline region and throughout the brain. If the vasculature is injured, the consequences may include hemorrhage, cerebral edema, and herniation.
In addition, rotational acceleration and decelerations injuries in infants secondary to being shaken can create extensive brainstem damage. For example, when subject to severe rotational forces there sometimes is tearing at the pontine-medullary junctions (where the brainstem meets the midbrain) and the brainstem may be partly torn loose and disconnected from the rest of the brain (Hardman, 2009; Jennett & Plum, 1992). Because the cerebral hemispheres sit atop the thin brainstem, there can result torque in the pontine-midbrain junction thus disconnecting these lower from higher regions. When this occurs consciousness is lost and the patient either dies or remains in a prolonged coma.
Although the shaken infant may suffer obvious cognitive and physical impairment, it is usually only when they are near death or severely injured that medical treatment is sought. Moreover, some parents may see their behavior as normal and "normally" relie on shaking to quiet their babies. This is because they are rewarded by the infants ensuing silence and what appears to be sleep. Indeed, because infants may be rendered unconscious or stuporous when they are repeatedly shaken, they cease to cry, and the "relieved" parent may then put them to bed where they may slip into a coma; though insofar as the parent is concerned, the baby is in fact "sleeping peacefully."
In some cases, however, due to the development of an expanding hemorrhage, the infant may eventually cease to breath and die. More commonly, however, they will later awaken (perhaps 24 or 48 hours later) and then interact in a lethargic or stuporous manner, or, play and eat "normally." Chronic lethargy and drowsiness are common symptoms in this population; disorders which may never resolve (Leestma 2015).
It is also not uncommon for crying infants to be smothered, which in turn results in oxygen deprivation and cell death. Some crying infants are in fact smothered to death. If there is no gross evidence of physical trauma the death may be erroneously attributed to "crib death" or "sudden infant death syndrome" (see Leestma 2015). Those who do not die, however, like their shaken or battered counterparts, simply go through life with varying degrees of brain damage. Even when the child may be shaken only once for a brief time period, the brain is subject to these rotational and shearing forces.
REPETITIVE TRAUMA & ABERRANT SYNAPTIC RECOVERY
When an infant is shaken, not uncommonly this results in cell death (necrosis), usually within the first 24 hours after injury --a process which may contininue for up to a week. In addition, a few days after axons and nerve fibers have been torn, the nerve cell often dies and the glial cellular reactions around these damaged, dying, and dead cells, creates microglia scars and cavities within the cortex and white matter which can also prevent functional recovery and reinnervation of normal neurons which have been disconnected. Often these scars can become epileptogenic and a source of abnormal activity.
Therefore, due to the immaturity of the cerebrum, infants who are shaken (and/or who receive direct blows to the head) not only suffer brain damage, but the maturational process itself is disrupted such that normal interconnections may not form, and abnormal pathways may develop in their place.
For example, following white matter or cortical injury, axons may sprout, grow, and reinnervate tissues that have lost their interconnections (Goldberger & Murray 2015; Raisman 1969), such as due to disconnection. This is not just a property of the damaged axon, however, for the deinnervated tissue is acting in some manner to attract these processes. For example, if an axon is severed not only the severed axon but other axons may spout, including those which might not normally synapse or form connections with these regions. One synaptic reconnection has been established, growth influences cease and migrating axons may shrink, die, or become rerouted (Joseph 1982).
Nevertheless, those axons which establish these new connections may be wholly inappropriate. That is, neurons and their axons which subserve completely different functions and which normally innervate completely different tissues and nuclei, may be attracted to the deinnervated region. Abnormal interconnections may therefore be established.
Prenatal complications, or trauma experienced during infancy or the fetal period is commonly associated with inapropriate neurons terminating in and establish interconnections in inappropriate layers of the neocortex (e.g. Bruton 1988; Akbarian et al. 1993; Cannon et al. 2014; Fish et al. 2012; Mednick, 2008; Taylor 1972). However, infants who are shaken or struck in the head not only may develop aberrant neural interconnections, but associated disturbances involving intellectual, emotional, and cognitive functioning. Affected children may seem slow, dull, overly aggressive, withdrawn, easily frightened and startled, etc.. Unfortunately, as repeatedly stressed above, the etiology (e.g. child abuse, head injury, shaken baby) may never even be suspected or detected.
GLIA, NE, FUNCTIONAL RECOVERY & REORGANIZATION
As detailed above, in many regions of the developing fetal brain radial glia fibers act to guide migrating neurons to their terminal substrate. Similarly, when the brain has been injured, glia cells near the site of injury begin to secrete various peptides, proteins, and related neurotrophic substances (e.g. nerve growth factor) and laminin. These substances induce or stimulate neuronal repair as well as promote neuronal growth such as by attracting axonal terminals to newly vacated cortical areas. Indeed, glia cells continue to secret these substances for up to 15 days post injury (reviewed in Stein & Glasier 2012).
Moreover, glia begin to secrete trophic substances which also share a feedback relationship with NE terminals. That is, NE-neurons respond to brain injures by secreting NE which in turn can influence the glia, some of which also have NE receptors (reviewed in Kolb 2012). When these glia are stimulated they can release more trophic substances which again act on NE neurons.
NE levels are differentially effected depending on the site of injury. For example, right cerebral injuries can induce widespread and bilateral alterations in NE levels, whereas left cerebral damage is characterized by local changes in NE (Robinson 1979).
Anamolous cerebral "recovery" even at distant and inappropriate sites may ensue due to these widespread NE changes. Because some glia have NE receptors, glia far distant from the site of injury may be induced to secrete trophic factors which in turn induce axonal (Foerester 1988) and dendritic growth (Coleman & Flood 1988) and possible reorganization even at sites far from the original injury (Pons et al. 2008). Thus injury to the brain can promote the establishment of abnormal and appropriate interconnections between adjacent injured tissues and even in normal tissue far from the site of the original injury.
As detailed above, following injury or deinnervation, often axons from adjoining tissue are stimulated to sprout terminals which may compete with severed and recovering axons for undamaged, albeit temporarily vacated synaptic space. That is, since lesions not only destroy tissue, but destroy fibers of passage, when those axons begin to regenerate their original target may be occupied by a different axon, which may in fact subserve and transmit information quite different from that of the original neural circuit.
Misdirected axonal interconnections, even when terminating in completely unrelated areas subserving completely different functions, may in turn become functional. For example, when retinal axons were experimentally redirected to the medial (rather than the lateral) geniculate or the somesthetic areas of the thalamus, the auditory and somesthetic neocortex was found to subsequently respond to visual stimuli (reviewed in O'Leary 1989). That is, neurons which normally process auditory input, instead began to process visual stimuli.
Similarly, Dunnett et al. (1985) performed neonatal lesions of the substantia innominata and destroyed cholinergic neurons. In consequence, the neocortex was deprived of cholinergic innervation. Dunnett et al. (1985) then transplanted ventral neuropeptide Y-immunoreactive neurons into the frontal cortex whcih were now deprived of cholinergic input. In consequence, these neuropeptide Y-immunoreactive neurons innervated the cholinergic deinnervated frontal neurons (see Lescaudron & Stein 2011).
Hence, depending on the timing and extent of the lesion, there may result functional substitution as neurons which normally do not directly communicate form synaptic interconnections. In consequence, they will begin performing functions that differ from those normally performed. However, in some cases, as adjacent neurons which perform similar functions may establish connections, there may be a restitution of lost functions, such that the original behavior and pattern of action is recovered. However, as detailed above, sometimes wholly inappropriate and abnormal interconnections are established, which in turn results in perceptual limitations and a host of behavioral abnormalities.
GLIA, AMINO ACIDS, REPETITIVE INJURIES & LIMITATIONS ON RECOVERY
Glia not only promotes recovery but acts as a limiting factor as well as a possible source of epileptic activity due to the presence of the buildup of a glia scar. Glia scarring may generate abnormal activity which results in epilepsy, or it may creates a barrier, due to the buildup of glia, which in turn prevents severe axons from growing back and reforming connections with damaged tissue.
Similar disturbances and functional limitations may be induced by the release of various amino acids; substances which can also act as neurotransmitters and which assist in the regulation and control of neurotransmission. For example, following brain injury, large quantities of excitatory amino acids such as glutamate are released (reviewed in Stein & Glasier 2012). However, when released in large amounts, glutamate can induce neuronal degeneration. For example, ischemia can produce a "glutamate cascade" which causes an increased and excessive flow of calcium ions into effected neurons thereby producing cell death (Stein & Glasier 2012).
Given that children and infants who are abused are usually repeatedly abused and thus suffer multiple and repeated brain injuries, the buildup of glia as well as the secretion amino acids such as glutamate can therefore act to interfere and disrupt subsequent attempts by the CNS to repair itself. Children who are repeatedly physically abused, therefore, not only suffer a high incidence of head trauma and brain damage, but their ability to recover from these injuries is significantly diminished.












