Neuroscience
Rhawn Gabriel Joseph, Ph.D.
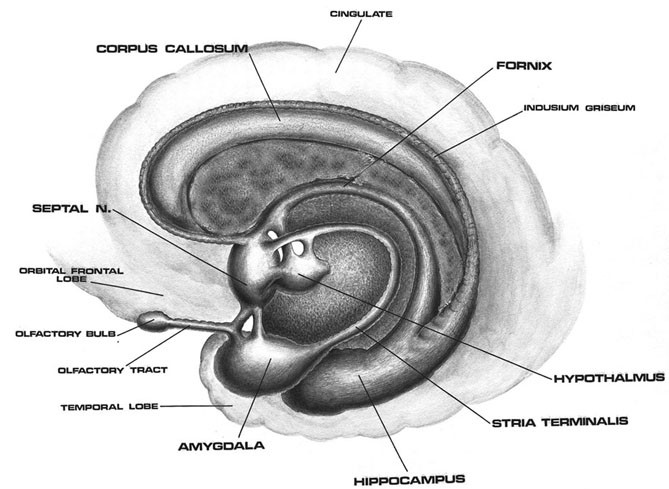
AMYGDALA
In contrast to the primitive hypothalamus, the more recently developed amygdala (the "almond") is preeminent in the control and mediation of all higher order emotional and motivational activities. Via it's rich interconnections with various neocortical and subcortical regions, amygdaloid neurons are able to monitor and abstract from the sensory array stimuli that are of motivational significance to the organism (Gaffan 1992; Gloor 1960, 1992, 2014; LeDoux 1992; Morris et al., 2013; Rolls, 2007, 1992 Steklis & Kling, 1985; Kling & Brothers 1992; Ursin & Kaada 1960). This includes the ability to discern and express even subtle social-emotional nuances such as friendliness, fear, love, affection, distruct, anger, etc., and at a more basic level, determine if something might be good to eat.
In fact, amygdaloid neurons respond selectively to the flavor of certain preferred foods, as well as to the sight or sound of something that might be especially desirable to eat (Fukuda et al. 2009; Gaffan et al. 1992; O'Keefe & Bouma, 1969; Ono et al. 1980; Ono & Nishijo, 1992) including even the sight of drugs that induce extreme pleasure.
For example, it has been shown, using positron emission tomography, that detoxified cocaine users not only respond to a cocaine video with cocaine craving, but with increased amygdala (and anterior cingulate) activity (Childress, et al., 2015).
Belying its involvement in emotion, including the pleasure associated with cocaine usage, is the unique chemical anatomy of the amygdala, which is rich in a variety of neuropetides including enkephalins and beta-endorphins as well as opiate receptors (Atweh & Kuhar, 1977; Fallon & Ciofi, 1992; Uhl et al. 2002). In fact, of all brain regions, the greatest concentration of opiate receptors is found within the human amygdala. Other chemical systems include leutenizing hormone, vasopressin, somatostatin, and corticotropin releasing factor (Fallon & Ciofi, 1992) --indications of its involvement in stress and sexuality, especially female sexuality. The primate amygdala is sexually differentiated with male and female patterns of dendritic organization and steroid activity (Bubenik & Brown, 1973; Nishizuka & Arai, 2005; see also Simerly, 1990).
The amygdala is exceedingly responsive to social and emotional stimuli as conveyed vocally, through touch, and via the face (Gloor, 1992; Halgren, 1992; Kling & Brothers 1992; Morris et al., 2013; Rolls, 2007, 1992). In fact, the amygdala, as well as the overlying (and partly coextensive) temporal lobe, contains neurons which respond selectively to smiles and to the eyes, and which can differentiate between male and female faces and the emotions they convey (Hasselmo, Rolls, & Baylis, 1989, Heit et al., 1988; Kawashima, et al., 2015; Rolls, 2007). For example, the left amygdala acts to discriminate the direction of another person's gaze, whereas the right amygdala becomes activated while making eye-to-eye contact (Kawashima, et al., 2015).
Moreover, the normal human amygdala typically responds to frightened faces by altering its activity (Morris et al., 2013), whereas injury to the amygdala disrupts the ability to recognize faces (Young, Aggleton, & Hellawell,1995). With bilateral destruction, emotional speech production and the capacity to respond appropriately to social emotionally stimuli is abolished (Lilly, Cummings, Benson, & Frankel, 1983; LeDoux, 2013; Marlowe, Mancall, Thomas,1975; Scott, Young, Calder, Hellawell, Aggleton, & Johnson, 2014; Terzian & Ore, 1955).
Single amygdaloid neurons receive a considerable degree of topographic input, and are predominately polymodal, responding to a variety of stimuli from different modalities simultaneously (Amaral et al. 1992; O'Keefe & Bouma, 1969; Ono & Nishijo, 1992; Perryman, Kling, & Lloyd, 2009; Rolls 1992; Sawa & Delgado, 1963; Schutze et al. 2009; Turner et al. 1980; Ursin & Kaasa, 1960; Van Hoesen, 2005). The amygdala is also very sensitive to somesthetic input and physical contact such that even a slight touch in a very circumscribed area of the body can produce amygdaloid excitation. Overall, because emotional, motivational, and multimodal assimilation of various sensory impressions occurs in this region, it is also involved in attention, learning, and memory (Gloor, 2014; Halgren, 1992; LeDoux, 2013).
Moreover, through the massive interconnections maintained with the lateral and medial (ventromedial) hypothalamus, the amygdala is able to act directly on this structure, driving the hypothalamus, so to speak, and thus tapping into its emotional reserviour so that its ends may be met. Indeed, it is able to modulate hypothalamic activity through inhibitory and excitatory projections to this structure (Dreifuss, et al., 1968).
Direct stimulation of the basolateral amygdala and the ventral amydalofugal pathway excites the principle neurons of the medial hypothalamus (Dreifuss, et al., 1968). By contrast, stimulation of the medial (ventro-medial) amygdala and the stria terminalis pathway, inhibits the these same hypothalamic neurons (Dreifuss, et al., 1968). Hence, whereas the lateral amygdala exerts excitatory influences on the hypothalamus, the medial amygdala exerts inhibitory influences, and can thus control, or at least exert excitatory/inhibitory and thus modulatory influences on hunger, thirst, sexual arousal, rage, etc., as well as hormonal, endocrine, and other functions associated with the hypothalamic nucleus (Dreifuss, et al., 1968; Joseph, 1992a; Gloor, 2014). Indeed, the amygdala can be likened to the chief executive of the limbic system and wields enormous power via its control over the hypothalamus.
For example, in the cat and monkey, stimulation of the border area between the lateral and medial hypothalamus can trigger aggressive defensive reactions (De vito & Smith, 1982; Hess, 1949). As indicated by radioactive tracers, both the lateral and medial amygdala projection to this area (De vito & Smith, 1982). And, when the amygdala is electrically activated, the hypothalamus becomes activated (Dreifuss, et al., 1968), and defensive and aggressive reactions can be triggered.
However, this system is also interactional, especially in regard to sexual activity, fear, anger, hunger, and stress. For example, the hypothalamus can stimulate the amygdala which may then survey the environment so that internal needs may be met, and/or they may act in concert regarding sexual behavior, the stress response, and so on.
OVERVIEW: AMYGDALA STRUCTURAL FUNCTIONAL ORGANIZATION
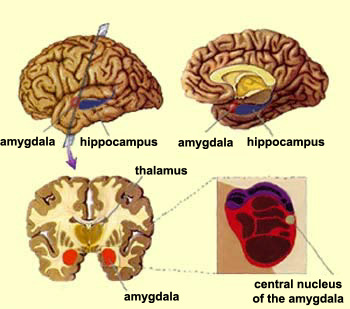
The amygdala is buried within the depths of the anterior-inferior temporal lobe and consists of several major nuclear groups including what has been referred to as the "extended amygdala." These include the cortical-medial, central, paralaminar, lateral, basal, and acessory basal nucleus (Amaral et al., 1992; Stephan & Andy, 1977). Different authors propose different divisions and link them differently. For example, Stephan and Andy (1977) assign the cortical division to the basolateral amydala, and the central division to the medial division. Price et al., (1977) subdivided the amygdala into basolateral, corticomedial and central amygdaloid nuclei. Others propose yet different schemes.
For our purposes we will primarily focus on the "medial" and the basolateral subdivisions. The phylogenetically ancient medial group (or cortico-medial amygdala) is involved in olfaction, sexual, and motor activity (via its interconnections with the striatum), and the relatively newer basolateral division (lateral amygdala) is most fully developed in primates and humans (Amaral et al. 1992; Herrick, 1925; Humphrey, 1972; McDonald 1992; Stephan & Andy, 1977). Of its various subdivisions, the basolateral amygdala is the most "cortex-like. However, being allocortex it contains three layers (vs 7 for the neocortex) with layer 2 containing pyramidal neurons which rely on excitatory neurotransmitters, e.g., glutamate--whereas the local-circuit (interneurons) rely on the inhibitory transmitters, e.g., GABA (Fallon & Ciofi, 1992).
The lateral amydala utilizes the ventral amygdalofugal pathway and less so the stria terminalis to influence the hypothalamus. The lateral amygdala also relies on the medial forebrain bundle which is the pathway subserving the pleasure circuit (Olds & Forbes, 2005). By contrast, the medial amygdala relies on the stria terminalis, and less so the amygdalofugal pathway to influences the hypothalamus. Through these pathways, these different subdivisions of the amygdala can act to modulate activity in the hypothalamus, septal nuclei, and other subcortical structures (Amaral et al. 1992; Stephan & Andy, 1977).
Evolution & Embryology
In humans, the right amygdala is also larger than the left amygdala, with the basolateral portion contributing to most of this asymmetry (Murphy et al., 2009). Moreover, over the course of evolution, and in the transition from amphibians to reptiles, to mammals and then humans, the basolateral amygdala appears to have grown the most in size as compared to other amygdaloid nuclei (Stephan & Andy, 1977)--that is, when considered only in regard to its subtemporal diminsions. However, it also appears that the medial amygdala may have contributed to the evolution of the piriform cortex, and then the evolution of the 4 to 5 layered mesocortex, which when layered upon the 3-layered allocortical piriform cortex, gave rise to the neocortex of the temporal lobe. Hence, the expansion of the medial amygdala may not be apparent as that expansion is represented as mesocortex and neocortex.
Moreover, it appears that the medial group was broken up over the course of evolution such that structures such as the claustrum (Gilles et al., 1983), became separated and is now situated beneath the auditory cortex of the superior temporal lobe. Indeed, the claustrum which is very cortical in organization, may well act as an interface between the auditory cortex and the amygdala, processing and relaying auditory impulses to and fro, and may have, in addition to the medial amygdala, contributed to the evolution of the auditory cortex.
The amygdala, therefore, has definitely increased in size over the course of evolution (Stephan & Andy, 1977), and has become increasingly cortical, has contributed to the evolution of mesocortex and neocortex (which is thus a cortical extension of the neocortex, maintaining extensive interconnections). In addition, the right amygdala is larger than the left (Murphy et al., 2009) which in turn may contribute to right hemisphere dominance for emotion (chapter 10).
Intrinsic & Extrinsic Organization: The Flow of Information
Like the lateral and medial hypothalamus, the medial and basolateral (hereafter referred to as the lateral) amygdaloid nuclei subserve different functions and maintain different anatomical interconnections (Amaral et al., 1992; Stephan & Andy, 1977). And, they can be subdivided into additional subnuclei. As noted, they also contain pyramidal neurons which are excitatory (Rolls, 1992) and use glutamate (Fallon & Ciofi, 1992) and which project throughout the neocortex as well as to the hippocampus (Amaral et al., 1992).
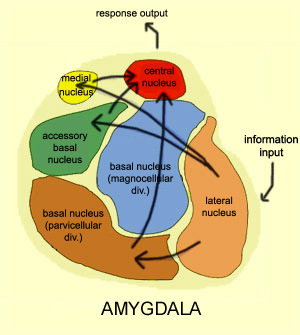
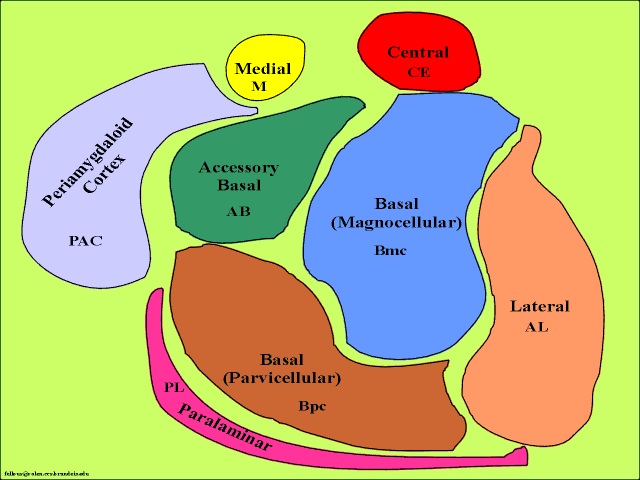
Local circuit neurons are mostly stellate-like and chandelier cells, which account for about 30% of the amygdala's neurons and which use the inhibitory transmitter GABA (Fallon & Ciofi, 1992). Considered rather broadly and simplistically, these local circuit neurons are organized in such a fashion that they appear to project information from the the lateral to the basal amygdala, and from the lateral basal to the medial and central amygdala which transmits, via pyramid and local-circuit neurons to the uncus, piriform cortex, medial temporal cortex, entorhinal cortex, anterior hippocampus, and via pyramidal neurons to the striatum, the septal nuclei, hypothalamus, cingulate, medial dorsal nucleus of the thalamus, brainstem, and throughout the frontal and temporal lobes (Amarral et al., 1992; Krettek & Price, 2002; Stephan & Andy, 1977; van Hoesen, et al., 2005). However, the lateral amygdala also projects to the septal nuclei, hypothalamus, corpus striatum, dorsal medial thalamus, brainstem, and throughout the neocortex via pyramidal axons (Amaral et al, 1992; Carlsen et al. 1982; Gloor, 1955; McDonald 1992; Russchen, 1982; Swanson & Cowan, 1979).
It appears that much of the input from the neocortex is directed at the lateral amygdala (the exception being auditory cortex which also projects to the medial amygdala). Hence, in certain respects, at least at the level of the neocortex, it appears that there is an almost circular stream of activity, from lateral/basal to medial/central to neocortex to lateral/basal. However, subcortically, both the lateral and medial project to many of the same exact structures, often providing counterbalancing excitatory/inhibitory influences.
Moreover, as detailed in chapter 12, the amygdala receives significant projections directly from the olfactory bulb. In act, "the olfactory system is the only sensory system in which first- and second-order central sensory neurons project directly to the amygdala" (Goor, 2014). Moreover, it receives projections from the gustatory system. Smell and taste thus converge in the amygdala, which may explain why some patients with temporal lobe epilepsy experience terrible odors and tasts as part of the aura which announces the onset of a seizure.
In addition, the secondary and in particular the association and multi-modal assimilation areas, including the orbital frontal lobe, project directly to the amygdala (Amaral et al, 1992; Carlsen et al. 1982; Gloor, 1955; Krettek & Price, 2002; McDonald 1992; Russchen, 1982; Swanson & Cowan, 1979; Stephan & Andy, 1977). In addition, as is evident from dissecting the human brain, the amygdala maintains significant reciprocal connections with the primary auditory area and Wernicke's area--and similar projections are evident in primates (Amaral et al., 1992). Hence, this structure receives simple and complex auditory, as well as fully formed perceptions from the neocortex which feeds the amygdala this information which it then analyzes for social, sexual, gustatory, and emotional significance.
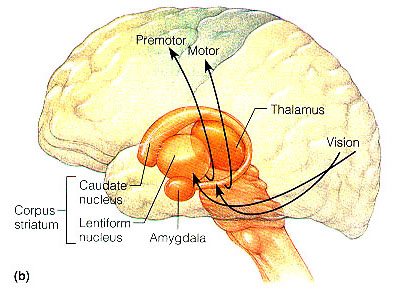
The Amygdala-Striatum
Embryologically, the medial amygdala is the first portion of the basal ganglia (limbic) striatal complex to appear during development, being formed via neuroblast migration from the epithilium of the lateral ventricle (Humphrey, 1972). Specifically, around the sixth week of fetal development immature neuroblasts migrate in massive numbers from the ventricular lining, and congregate in the more caudal portion of the emerging forebrain, thus forming an arc shaped "striatal ridge" from which the primordial amygdala and striatum will emerge (Gilles et al., 1983; Humphrey, 1968).
Approximately one week after the formation of the amygdala, this primordial amygdala-striatum begins to differentiate and balloon outward to also create the striatum. That is, both the striatum and amygdala are derived from the arc shaped "striatal ridge," the caudal portion giving rise to the primordial amgydala at about the 6th week of gestation, and the basal portion later giving rise to the primordial striatum which initially overlies and is contiguous with the amygdala (Gilles et al., 1983; Humphrey, 1968).
Thus initially these structures are contiguous. However, over the ensuing weeks, these structures are pushed apart as the forebrain and its interconnections form. However, although they are pushed apart and may even break up into semi-separate islands (or rather, penninsulas) the amygdala maintains connections with what is referred to as the "extended amygdala," i.e., the limbic striatum (Heimer & Alheid, 1991) and through what is called the "tail of the caudate" maintains massive interconnections with the corpus striatum
Specifically, the tail of the caudate nucleus (as it circles in an arc from the frontal to temporal lobe) terminates and merges with the medial and in particular, the lateral amygdala. Hence, the lateral amygdala has also been referred to the "striatum limitans, and the "striatum accessorium" (Gloor, 2014).
By contrast,the medial amygdala (or rather, the central division of the medial amygdala, central-medial amygdala) extends almost imperceptibly around the fundus of the entorhinal sulcus, and merges with the substantia innominata of the limbic striatum. The amygdala, therefore, is in fact part of the basal ganglia and is heavily involved in motivating and coordinating gross, or whole body motor activity via the striatum (Heimer & Alheid, 1991; Mogenson & Yang, 1991).
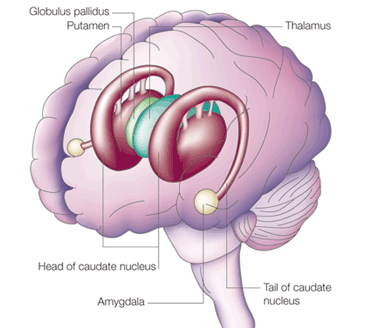
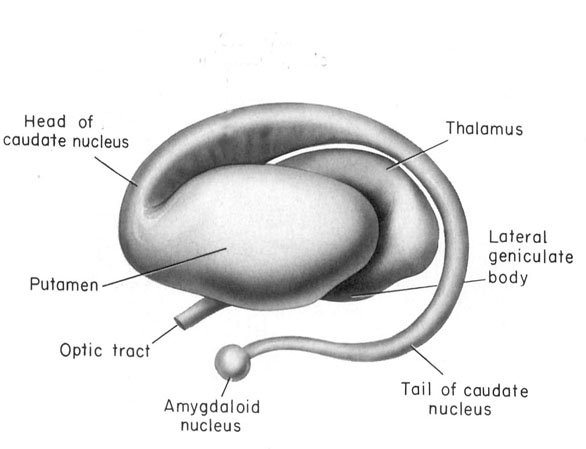
THE MEDIAL AMYGDALA
The medial amygdala receives fibers from the olfactory tract, and via a rope of fibers called the stria terminalis projects directly to and receives fibers from the medial hypothalamus (via which it exerts inhibitory influences) as well as the septal nucleus (Amaral et al, 1992; Carlsen et al. 1982; Gloor, 1955; McDonald 1992; Russchen, 1982; Swanson & Cowan, 1979). The stria terminals is significantly larger and thicker in males vs females (Allen & Gorksi 1992) which suggests that information and impulse exchange (or inhibition) between the hypothalamus and amygdala is different in men vs women. Moreover, in humans, the amygdala in general is large in males than in females, and in primates, the medial amygdala is sexually differentiated (Nishizuka & Arai, 2005; see also Simerly, 1990), such that the male amygdala contains a greater number of synaptic connections and shows different patterns of steroidal activity (Nishizuka & Arai, 2005; Simerly, 1990). In fact, the human amygdala is 16% larger in the male in total volume (Filipek, et al., 2012) whereas in male rats, the medial amygdala is 65% larger than the female amygdala and grows or shrinks in the presence of testosterone (Breedlove & Cooke, 2015).
The female medial amygdala is a principle site for uptake of the female sex hormone, estrogen, and contains a high concentration of lutenizing hormones (Stopa et al., 1991) which are important during pregnancy and nursing. In fact, the female medial amygdala fluctuates immunoreactive activity during estrus cycle, being highest during proestrus (Simerly, 1990). Moreover, the medial amygdala projects directly to the ventromedial hypothalamus and the preoptic area of the hypothalamus which, as noted above, are sexually differentiated (e.g. Allen et al., 1989; Gorski, et al., 2002; Le Vay, 1991; Raisman & Field, 1971), and which when activated produce sex specific behaviors (Hart et al., 1985; Lisk, 1967, 1971; MacLean, 1973) and, in primates, even maternal behavior (Numan, 1985). These amygdala to hypothalamic synapses are excitatory.
Because the medial amygdala is sexually differentiated, and through its massive connections with the hypothalamus and preoptic area, as well as the striatum which controls gross motor and limb movements, when activated, male vs female sexual behavior can be triggered. These amygdala-induced sexual behariors include sexual posturing, penile erection and clitoral tumenence (Kling and Brothers, 1992; MacLean, 1990; Robinson and Mishkin, 1968; Stoffels et al., 1980), thrusing, sexual moaning, ejaculation, as well as ovulation, uterine contractions, lactogenetic responses, and orgasm (Backman and Rossel, 2007; Currier, Little, Suess and Andy, 1971; Freemon and Nevis,1969; Warneke, 2001; Remillard et al., 1983; Shealy and Peel, 1957).
In addition, the medial (and lateral) regions are rich in cells containing enkephalins, and opiate receptors can be found throughout the amygdala (Atweh & Kuhar, 1977; Fallon & Ciofi, 1992; Uhl et al. 2002) and the amygdala becomes exceedingly active when experiencing a craving for pleasure inducing drgus, such as cocaine (Childress et al., 2015). In this regard, the amygdala is capable of inducing extreme feelings of pleasure as well as motivating the individual to engage in pleasure-seeking behaviors such as sexual activity.
LATERAL AMYGDALA
With the evolutionary ascent of primates the lateral division of the amygdala progressively expands and differentiates. The lateral amygdala contributes fibers to the stria terminalis and gives rise to the amygdalofugal pathway via which it projects to the lateral and medial hypothalamus (upon which it exerts inhibitory and excitatory influences respectively), the dorsal medial thalamus (which is involved in memory, attention and arousal), the limbic and corpus striatum, as well as other subcortical regions including the brainstem (Aggleton et al. 1980; Amaral et al. 1992; Carlsen et al. 1982; Dreifuss et al., 1968; Gloor, 1955, 1960, 2014; Klinger & Gloor, 1960; McDonald 1992; Mehler, 1980; Russchen, 1982). Lateral amygdala brainstem projection pathways include the dopamine producing substantia nigra, the vocalizing periaqueductal gray, the pontine tegmentum which includes and area that triggers the startle response (Amaral et al., 1992; Davis et al., 2014) as well as visceral nuclei such as those controlling blood pressure, respiration, vosodilation and constriction and so on. It also sends some fibers into the spinal cord, where they travel along with those of the pyramidal tract (Amaral et al., 1992). It also receives fibers from the medial forebrain bundle which in turn has it's site of origin in the lateral hypothalamus (Mehler, 1980).
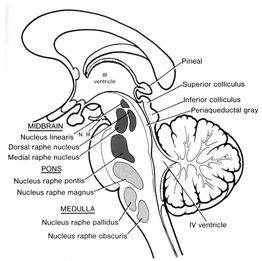
In general, whereas the medial amygdala is highly involved in motor, olfactory and sexual functioning, the lateral division is intimately involved in all aspects of emotional activity. Hence, it's rich interconnections with the lateral and medial hypothalamus, and the neocortex and those brainstem centers controlling the visceral aspects of affective-motor behavior.
The lateral amygdala maintains rich interconnections with the inferior, middle, and superior temporal lobes, as well as the insular temporal region, which in turn allows it to sample and influence the auditory, somesthetic, and visual information being received and processed in these areas, as well as scrutinize this information for motivational and emotional significance (Gloor 1992; Herzog & Van Hoesen 2001; Kling et al., 2009; Machne & Segundo, 1956; Mesulam & Mufson, 1982; O'Keefe & Bouma, 1969; Rolls 1992; Steklis & Kling, 1985; Turner et al., 1980; Van Hoesen, 2005). Gustatory and respiratory sense are also re-represented in this vicinity (Amaral et al. 1992; Fukuda et al., 2009; Maclean, 1949; Ono et al., 1980) as is the capacity to influence (via sensory analysis) food and water intake. The lateral division also maintains rich interconnections with cingulate gyrus, orbital and medial frontal lobes and the parietal cortex (Amaral et al. 1992; McDonald 1992; O'Keefe & Bouma, 1969; Pandya et al. 1973) through which it able to influence emotional expression and receive complex somesthetic information.
The lateral amygdala is highly important in analyzing information received and transferring information back to the neocortex so that further elaboration may be carried out at the neocortical level. It is through the lateral division that emotional meaning and significance can be assigned to as well as extracted from that which is experienced.
The amygdala, overall, maintains a functionally interdependent relationship with the hypothalamus. It is able to modulate and even control rudimentary emotional forces governed by the hypothalamic nucleus. However, it also acts as the behest of hypothalamically induced drives. For example, if certain nutritional requirements need to be meet, the hypothalamus signals the amygdala which then surveys the external environment for something good to eat or drink.
On the other hand, if the amygdala via environmental surveillance were to discover a potentially threatening stimulus, it acts to excite and drive the hypothalamus so that the organism is mobilized to take appropriate action. as noted, direct stimulation of the basolateral amygdala and the ventral amydalofugal pathway excites the principle neurons of the ventromedial hypothalamus (Dreifuss, et al., 1968). When the hypothalamus is activated by the amygdala, instead of responding in an on/off manner, cellular activity continues for an appreciably longer time period (Dreifuss et. al. 1968; Rolls 1992). The amygdala can tap into the reservoir of emotional energy mediated by the hypothalamus so that certain ends may be attained.
ATTENTION
The amygdala acts to perform environmental surveillance and can trigger orienting responses as well as mediate the maintanence of attention if something of interest or importance were to appear (Gloor, 1955, 1960, 1992; Kaada, 1951; Kapp et al., 1992; Rolls 1992; Ursin & Kaasa, 1960).
In part, the attention response can be triggered by amygdala activation of the brainstem, frontal lobes, and the dorsal medial nucleus (DMN) of the thalamus, each of which is implicated in arousal (see chapter 19). The DMN, for example, in conjunction with the frontal lobe, acts to gate and regulate the flow of information destined for the neocortex (Joseph, 2015a). The amydala, being provided thalamic, brainstem, as well as neocortical input (as well as projecting to these nuclei), is therefore able to directly influence the DMN so that attention can be directed to particular percepts (and emotional significance attached). In fact, the projections of the amygdala to extend well beyond the DMN, but extends throughout the thalamus (Aggleton et al., 1980; LeDoux, 2013; McDonald, 1992; Russchen, 1982), as well as throughout the neocortex .
Electrical stimulation of the lateral amygdala, therefore, can initiate quick and/or anxious glancing and searching movements of the eyes and head such that the organism appears aroused and highly alert as if in expectation of something that is going to happen (Halgren 1992; Kapp et al., 1992; Ursin & Kaasa, 1960). The EEG becomes desynchronized (indicating arousal), heart rate becomes depressed, respiration patterns change, and the galvanic skin response significantly alters (Bagshaw & Benzies, 1968; Kapp et al. 2012; Ursin & Kaada, 1960) and the animal may freeze (Gloor, 1960; Kapp et al., 1992) -- reactions which characteristically accompany the orienting response of most species.
Once a stimulus of potential interest is detected, the amygdala then acts to analyze its emotional-motivational importance and will act to alert other nuclei such as the hypothalamus, brainstem, and striatum, so that appropriate action may take place.
FEAR, RAGE & AGGRESSION
Initially, electrical stimulation of the amygdala produces sustained attention and orienting reactions. If the stimulation continues the subject may begin to experience, wariness, fear and/or rage (Cendes et al. 2012; Davis et al., 2014; Gloor 1992; Halgren 1992; LeDoux, 2013; Rosen & Schulkin, 1998; Ursin & Kaada, 1960). When fear follows the attention response, the pupils dilate and the subject will cringe, withdraw, and cower. This cowering reaction in turn may give way to extreme fear and/or panic such that the animal will attempt to take flight.
Among humans, the fear response is one of the most common manifestations of amygdaloid electrical stimulation and abnormal activation (Davis et al., 2014; Gloor, 1992, Halgren, 1992; LeDoux, 2013; Rosen & Schulkin, 1998). Moreover, unlike hypothalamic on/off emotional reactions, attention and fear reactions can last up to several minutes after the stimulation is withdrawn.
In addition to behavioral manifestations of heightened emotionality, amygdaloid stimulation can result in intense changes in emotional facial expression. This includes crying and facial contortions such as baring of the teeth, dilation of the pupils, widening or narrowing of the eye-lids, flaring of the nostrils, as well as sniffing, licking, and chewing (Anand & Dua, 1955; Ursin & Kaada, 1960). Indeed, some of the behavioral manifestations of a seizure in this vicinity (i.e. temporal lobe epilepsy) typically include throat and mouth movements, including chewing, smacking of the lips, licking, and swallowing--a consequence, perhaps of amygdala activation of the brainstem periaqueductal gray and nuclei subserving mastication.
In many instances patients or animals will react defensively and with anger, irritation, and rage which seems to gradually build up until finally the animal or human will attack (Egger & Flynn, 1963; Gunne & Lewander, 1966; Mark et al., 1972 Ursin & Kaada, 1960; Zbrozyna, 1963). Unlike hypothalamic "sham rage", amygdaloid activation results in attacks directed at something real, or, in the absence of an actual stimulus, at something imaginary. There have been reported instances of patient's suddenly lashing out and even attempting to attack those close by, while in the midst of a temporal lobe seizure (Saint-Hilaire et al., 1980), and/or attacking, kicking, and destroying furniture and other objects (Ashford et al., 1980).
Moreover, rage and attack will persist well beyond the termination of the electrical stimulation of the amygdala. In fact, the amygdala remains electrophysiologically active for long time periods even after a stimulus has been removed (be it external-perceptual, or internal-electrical) such that is appears to continue to process--in the abstract--information even when that information is no longer observable (O'Keefe & Bouma, 1969).
The amygdala, in addition to sustained electrophysiological activity, has been shown to be heavily involved in the maintenance of behavioral responsiveness even in the absence of an immediately tangible or visible objective or stimulus (O'Keefe & Bouma, 1969). This includes motivating the organism to engage in the seeking of hidden objects or continuing a certain activity in anticipation of achieving some particular long term goal. At a more immediate level, the amygdala is probably very important in object permanence (i.e. the keeping of an object in mind when it is no longer visible) and concrete or abstract anticipation. Anticipation is, of course, very important in the prolongation of emotional states such as fear or anger, as well as the generation of more complex emotions such as anxiety. In this regard, the amygdala is probably important not only in regard to emotion, but in the maintenance of mood states.
Fear and rage reactions have also been triggered in humans following depth electrode stimulation of the amygdala (Chapman, 1960; Chapman et al., 1954; Heath et al. 1955; Mark et al. 1972). Mark et al. (1972) describe one female patient who following amygdaloid stimulation became irritable and angry, and then enraged. Her lips retracted, there was extreme facial grimacing, threatening behavior, and then rage and attack--all of which persisted well beyond stimulus termination.
Similarly, Schiff et al. (1982) describe a man who developed intractable aggression following a head injury and damage (determined via depth electrode) to the amygdala (i.e. abnormal electrical activity). Subsequently, he became easily enraged, sexually preoccupied (although sexually hypoactive), and developed hyper-religiosity and psuedo-mystical ideas. Tumors invading the amygdala have been reported to trigger rage attacks (Sweet et al. 1960; Vonderache, 1940).
The amygdals appears capable of not only triggering and steering hypothalamic activity but acting on higher level neocortical processes so that individuals form emotional ideas . Indeed, the amygdala is able to overwhelm the neocortex and the rest of the brain so so that the person not only forms emotional ideas but responds to them, sometimes with vicious, horrifying results. A famous example of this is Charles Whitman, who in 1966 climbed a tower at the University of Texas and began to indiscriminantly kill people with a rifle (Whitman Case File # M968150. Austin Police Department, Texas, The Texas Department of Public Safety, File #4-38).
Case Study in Amygdala-Aggression: Charles Whitman
Charles Whitman was born on June 24, 1941 and even before entering grade school had shown exceptional intellectual promise, was well liked by neighbors and had already shown some mastery of the piano, which he "loved to play." At the age of six he was administered the Stanford Binet tests of intellectual ability and obtained an IQ of 138; thus scoring at the 99.9% rank. He also became enamored by guns; his father being described as a gun fanatic. According to his father, "Charlie could plug a squirrel in the eye by the time he was sixteen." However, Charlie loved animals, was somewhat religiously oriented as a child, was very athletic, was described as "handsome" and "fun" and "high spirited" and was in many respects the "all American boy." He became an Eagle Scout at age 12, and receiving national recognition as being the youngest Eagle Scout in the world. Within 15 months he had earned 21 merit badges. While in high school he continued these activities, also pitching for the baseball team and managing the football team. After high school he joined the Marines and was described as "the kind of guy you would want around if you went into combat." It was while in the Marines that he got married, and it was during this period that began to show the first subtle signs that something might be amiss.
He began having ocassional bursts of anger. He threatened to "kick the teeth out" of another Marine, was court marshalled, consigned to the brig for 30 days, and reduced in rank. He also began taking copious notes, and developed what is referred to as "hypergraphia" excessive writing--a disturbance associated with the amygdala (Joseph, 2015b).
Incessantly he began to write and leave himself notes, ranging from the mundane, to the tremendous love he felt for his wife. "Received a call from Kathy... it was fabulous, she sounds so wonderful. I love her so much... I will love her to the day I die. She is the best thing I have in life. My Most Precious Possession."
Increasingly, however, he was having trouble with his temper and composed notes offering self-advice as to how to control his growing temper and rage attacks. "CONTROL your anger" he wrote, "Don't let it prove you the fool. SMILE--Its contagious. DON'T be belligerent. STOP cursing. CONTROL your passion; DON'T LET IT lead YOU."
On February 4, 1964, he purchased a diary. According to Charles: "I opened this diary of my daily events as a result of the peace of mind or release of feelings that I experienced when I started making notes of my daily events...."
Nevertheless, he also continued to excel and although he had been Court marshaled, he also won a scholarship to attend the University of Texas and to attend classes while still in the Marines. He also became increasingly religious and would often have discussions with his school mates about the nature of God--hyperreligiousness also being associated with an abnormality involving the amygdala (see chapter 9). And, although he was attending classes, he also began to perform volunteer work, while simultaneously holding a part time job, and at times felt overwhelmed with energy, almost manic--mania also being associated with the amygdala (Strakowski et al., 2015) as well as the frontal lobes (Joseph, 1986a, 1988a, 2015a). And, he continued to be well liked and admired. His supervisor at the bank, E. R. Hendricks, described Charles "as a truly outstanding person. Very likeable. Neat. Nice looking... A great guy."
However, Charles also began suffering terrible headaches, and one day lost his temper in class, pulling a male student bodily from his chair and tossing him from the classroom. Apparently he felt considerable remorse. He also continued to have frequent bouts of anger and on occasion, difficulty concentrating, and was beginning to over eat--increased food consumption being associated with a disturbance of the hypothalamus. Moreover, he began having periods where he couldn't sleep for days at a time--yet another disturbance associated with the hypothalamus, a major sleep center. Charles also realized that something was wrong, and continued writing copious notes to himself, reminding himself to be nice, to control his appetite, and especially to control temper. But his temper was getting out of control and Charles was gaining weight.
A close friend, Elaine Fuess, also noticed that something was amiss. "Even when he looked perfectly normal, he gave you the feeling of trying to control something in himself. He knew he had a temper, and he hated this in himself. He hated the idea of cruelty in himself and tried to suppress it."
Charles Whitman finally sought professional help and consulted a staff psychiatrist, at the University of Texas Health Center about his periodic and uncontrollable violent impulses. Charles was referred to Dr. Heatly. According to the report written by Dr. Heatly about his session with Whitman, a report which was distributed to the media: "This massive, muscular youth seemed to be oozing with hostility as he initiated the hour with the statement that something was happening to him and he didn't seem to be himself...." Whitman "could talk for long periods of time and develop overt hostility while talking, and then during the same narration show signs of weeping.... Past history revealed a youth who... grew up in Florida where his father was a very successful plumbing contractor... who achieved considerable wealth. He identified his father as being brutal, domineering, and extremely demanding of the other three members of the family." Whitman "married four or five years ago, and served a hitch in the Marines.... He referred to several commendable achievements during his Marine service, but also made reference to a court martial for fighting which resulted in being reduced several grades to private. In spite of this he received a scholarship to attend the University for two years and remained a Marine at the same time... He expressed himself as being very fond of his wife, but admitted that he had on two occasions assaulted his wife physically. He said he has made an intense effort to avoid losing his temper with her... His real concern is with himself at the present moment. He readily admits having overwhelming periods of hostility with a very minimum of provocation... he... also... made vivid reference to thinking about going up on the tower with a deer rifle and start shooting people. ....He was told to make an appointment for the same day next week."
Instead, Charles apparently decided to climb the tower and to begin killing people. But not before first contacting the police and asking to be arrested. As Charles had not committed a crime, the desk sergeant instead suggested that he see a psychiatrist.
Several days prior to climbing the tower, Charles Whitman wrote himself a letter:
"I don't quite understand what it is that compels me to type this letter.... I don't really understand myself these days... Lately I have been a victim of many unusual and irrational thoughts. These thoughts constantly recur, and it requires a tremendous mental effort to concentrate. I consulted Dr. Cochrum at the University Health Center and asked him to recommend someone that I could consult with about some psychiatric disorders I felt I had.... I talked to a doctor once for about two hours and tried to convey to him my fears that I felt overcome by overwhelming violent impulses. After one session I never saw the Doctor again, and since then I have been fighting my mental turmoil alone, and seemingly to no avail. After my death I wish that an autopsy would be performed to see if there is any visible physical disorder. I have had tremendous headaches in the past and have consumed two large bottles of Excedrin in the past three months."
On August 1, 1966, one day before climbing the tower at the University of Texas, Charles Whitman paid a visit to his mother, who greeted him outside her penthouse and introduced him to the night watchman who noticed that Charles was carrying a big black attache case. According to police reports, Charles must have immediately attacked his mother after they entered the penthouse, and then brutally beat, strangled, and stabbed her to death, crushing the back of her head, smashing her hands, and stabbing her in the chest with a huge hunting knife.
Later, neighbors told police that the only sounds they heard were that of a "child crying and whimpering," which they found puzzling as no child lived in the penthouse.
After brutally murdering his mother, Charles cleaned up the mess, and placed her in bed with a notepad laying across and covering up the massive wound in her chest. Charles had left a note. It read: "To Whom It May Concern: I have just taken my mother's life. I am very upset over having done it. However, I feel that if there is a heaven she is definitely there now... I am truly sorry... Let there be no doubt in your mind that I loved this woman with all my heart."
After killing his mother, Charles returned home, planning on killing his wife "as painlessly as possible.," as he explained in yet another note:
"It was after much thought that I decided to kill my wife, Kathy, tonight....I love her dearly, and she has been a fine wife to me as any man could ever hope to have. I cannot rationally pinpoint any specific reason for doing this..."
Apparently she was sleeping, and after removing the blankets to expose her nude body, he viciously stabbed her repeatedly with his huge hunting knife, leaving five gaping holes in her chest. She died instantly.
Charles wrote another note which he left with the body:
"I imagine it appears that I brutally killed both of my loved ones. I was only trying to do a quick thorough job... If my life insurance policy is valid please pay off my debts... donate the rest anonymously to a mental health foundation. Maybe research can prevent further tragedies of this type."
And then he added a post script beneath his signature: "Give our dog to my in-laws. Tell them Kathy loved "Schoci" very much."
The next morning Charles Whitman climbed the University tower carrying several guns, a sawed off shotgun, and a high powered hunting rifle, and for the next 90 minutes he shot at everything that moved, killing 14, wounding 38.
Post-mortem autopsy of his brain revealed a glioblastoma multiforme tumor the size of a walnut, erupting from beneath the thalamus, impacting the hypothalamus, extending into the temporal lobe and compressing the amygdaloid nucleus (Charles J. Whitman Catastrophe, Medical Aspects. Report to Governor, 9/8/66).


DOCILITY & AMYGDALOID DESTRUCTION
Bilateral destruction of the amygdala usually results in increased tameness, docility, and reduced aggressiveness in cats, monkeys and other animals (Schreiner & Kling, 1956; Weiskrantz, 1956; Vochteloo & Koolhaas, 2009), including purportedly ferocious creatures such as the agoutie and lynxe (Schreiner & Kling, 1956). In man, bilateral amygdala destruction (via neurosurgery) has been reported to reduce and/or eliminate paroxysmal aggressive and violent behavior (Terzian & Ore, 1955).
In some creatures, however, bilateral ablation of the amygdala has been reported to al least initially result in increased aggressive responding (Bard & Mountcastle, 1948), and if sufficiently aroused or irritated, even the most placid of amygdalectomized animals can be induced to fiercely fight (Fuller et al. 1957).
However, these aggressive responses are very short-lived and appear to be reflexively mediated by the hypothalamus. Hence, these findings (and the data reviewed above) suggests that true aggressive feelings including violent moods, are dependent upon activation of the amygdala.
SOCIAL-EMOTIONAL AGNOSIA
Among primates and mammals, bilateral destruction of the amygdala significantly disturbs the ability to determine and identify the motivational and emotional significance of externally occuring events, to discern social-emotional nuances conveyed by others, or to select what behavior is appropriate given a specific social context (Bunnel, 1966; Fuller et al. 1957; Gloor, 1960; Kling & Brothers 1992; Kluver & Bucy, 1939; Lilly et al., 1983; Marlowe et al., 1975; Scott et al., 2014; Terzian & Ore, 1955; Weiskrantz, 1956). Bilateral lesions lower responsiveness to aversive and social stimuli, reduce aggressiveness, fearfullness, competitiveness, dominance, and social interest (Rosvold et al. 1954). This condition is so pervasive that subjects have tremendous diffficulty discerning the meaning or recognizing the significance of even common objects -- a condition sometimes referred to as "psychic blindness", or, the "Kluver-Bucy syndrome" (Lilly et al., 1983; Marlowe et al., 1975; Terzian & Ore, 1955).
Thus, animals with bilateral amygdaloid destruction, although able to see and interact with their environment, may respond in an emotionally blunted manner, and seem unable to recognize what they see, feel, and experience. Things seem stripped of meaning. Like an infant (who similarly is without a fully functional amygdala), individuals with this condition engage in extreme orality and will indiscriminately pick up various objects and place them in their mouth regardless of its appropriateness. There is a repetitive quality to this behavior, for once they put it down they seem to have forgotten that they had just explored it, and will immediately pick it up and place it again in their mouth as if it were a completely unfamiliar object.
Although ostensibly exploratory, there is thus a failure to learn, to remember, to discern motivational significance, to habituate with repeated contact, or to discriminate between appropriate vs inappropriate stimuli. Rather, when the amygdala has been removed bilaterally the organism reverts to the most basic and primitive modes of object and social-emotional interaction (Brown & Schaffer, 1888; Gloor, 1960; Kluver & Bucy, 1939; Weiskrantz, 1956) such that even the ability to appropriately interact with loved ones is impaired (Lilly et al., 1983; Marlowe et al., 1975; Terzian & Ore, 1955).
For example, Terzian & Ore (1955) described a young man who following bilateral removal of the amygdala subsequently demonstrated an inability to recognize anyone, including close friends, relatives and his mother. He ceased to respond in an emotional manner to his environment and seemed unable to recognize feelings expressed by others. He also demonstrated many features of the Kluver-Bucy syndrome (perseverative oral "exploratory" behavior and psychic blindness), as well as an insatiable appetite. In addition, he became extremely socially unresponsive such that he preferred to sit in isolation, well away from others.
Among primates who have undergone bilateral amygdaloid removal, once they are released from captivity and allowed to return to their social group, a social-emotional agnosia becomes readily apparent as they no longer respond to or seem able to appreciate or understand emotional or social nuances. Indeed, they appear to have little or no interest in social activity and persistently attempt to avoid contact with others (Dicks et al. 1969; Jonason & Enloe, 1971; Kling & Brothers 1992; Jonason et al. 1973). If approached they withdraw, and if followed they flee.
Indeed, they behave as if they have no understanding of what is expected of them or what others intend or are attempting to convey, even when the behavior is quite friendly and concerned. Among adults with bilateral lesions, total isolation seems to be preferred.
In addition, they no longer display appropriate social or emotional behaviors, and if kept in captivity will fall in dominance in a group or competitive situation -- even when formerly dominant (Bunnel, 1966; Dicks et al., 1969; Fuller et al., 1957; Jonason & Enloe, 1971; Jonason et al., 1973; Rosvold et al. 1954).
As might be expected, maternal behavior is severely affected. According to Kling (1972), mothers will behave as if their "infant were a strange object be be mouthed, bitten and tossed around as though it were a rubber ball".
EMOTIONAL LANGUAGE & THE AMYGDALA
Although cries and vocalizations indicative of rage or pleasure have been elicited via hypothalamic stimulation, of all limbic nuclei the amygdala is the most vocally active--particularly the lateral division (Robinson, 1967). In humans and animals a wide range of emotional sounds have been evoked through amygdala activation, such as sounds indicative of pleasure, sadness, happiness, and anger (Robinson, 1967; Ursin & Kaada, 1960). The human amygdala can produce as well as perceive emotional vocalizations (Halgren, 1992; Heit, Smith, & Halgren, 1988).
Conversely, in humans, destruction limited to the amygdala (Freeman & Williams 1952, 1963), the right amygdala in particular, has abolished the ability to sing, convey melodic information or to properly enunciate via vocal inflection. Similar disturbances occur with right hemisphere damage (chapter 10). Indeed, when the right temporal region (including the amygdala) has been grossly damaged or surgically removed, the ability to perceive, process, or even vocally reproduce most aspects of musical and emotional auditory input is significantly curtailed .
EMOTION & TEMPORAL LOBE SEIZURES
The amygdala is buried within the depths of the anterior-inferior temporal lobe and maintains rich interconnections with areas throughout the temporal neocortex. Because of their intimate association, damage to the temporal lobe, particularly the anterior regions, often involves and distrupts amygdaloid functioning. In fact, because the amygdala and inferior-anterior temporal lobe have, of all brain regions, the lowest seizure threshold, and are minimally resistant, and thus maximally vulnerable to developing abnormal seizure activity, even mild injuries may result in kindling (i.e. abnormal activation), and thus disruption of their functional integrity. Indeed, damage to adjacent tissue has been know to spread, via kindling, to the amygdala and inferior regions (Cain 1992). One consequence is temporal lobe epilepsy (Gloor, 2014; Wolf & Blumcke, 2015).
Personality, emotional, and sexual disturbances are a frequent complication of temporal lobe seizures in a significant minority of patients (Gloor, 2014; Trimble, 1991, 2013). Such individuals may develop paranoid, hysterical, or depressive tendencies, deepening of mood, hyposexuality and other characteristics suggestive of affective disorders (Bear et al. 1982; Gibbs, 1951; Gloor 1986, 1992; Herman & Chambria, 1980; Strauss et al. 1982; Williams, 1956). Immediately following or during the course of a seizure 10% or more of such individuals experience a change in emotionality (Herman & Chambria, 1980; Strauss et al., 1982; Trimble, 1991; Williams, 1956).
In part, because the highest incidence of psychiatric disorder occurs in cases where the EEG spike focus is in the anterior temporal area (Gibbs, 1951), and because limbic nuclei such as the amygdala are frequently involved (e.g. Gloor, 2014; Strakowski et al., 2015), it has been postulated that seizure activity sometimes hyperactivates these nuclei (Bear, 1979) which in turn distorts the affective meaning applied to afferent streams of visual, auditory, and somesthetic information (Gibbs, 1951; Gloor, 2014).
Thus, during a seizure, an individual may be temporarily overwhelmed by feelings such as fear. Or things they see or hear seem to become abnormally invested with emotional significance (Bear et al., 1979; Gibbs, 1951; Gloor 1992, 2014; Gloor et al., 1982), presumably due to abnormal amygdala activation. Interestingly, one common symptom of temporal lobe epilepsy is an aura of tastes and more often odors which are usually quite unpleasant (e.g. like "burning wire", "burning shit", "burning rubber or tires", etc.).
Seizure induced emotional changes tend to predominantly involve feelings of depression, pleasure, displeasure, or fear -- with fear being one of the most common emotional experiences (Gloor 1992; Gloor et al., 1982; Halgren 1992; Williams, 1956). Patients may also have seizures involving sexual behavior, crying, laughing, or rage-like responses have been associated with temporal lobe epilepsy. Moreover, many of these same exact feelings and behaviors can be triggered by direct stimulation of the amygdala (Chapman, 1960; Chapman et al., 1954; Gloor, 2014; Halgren, 1992; Kaada, 1951, 1972). These include fear, coupled with the remembrance of a fearful or traumatic memory, rising epigastric sensations, chest sensations, nausea, heart palpations, feelings of cold or warmth, shivering, pallor or flushing of the face, respiratory changes including apnea, salivation, belching, farting, sweating, and vaginal secretions in women which may be accompanied by sexual feelings or behaviors.
There is evidence which suggests that certain emotional changes are more frequently associated with seizure originating in the right temporal lobe and/or amygdala, whereas disturbances involving thought (e.g. psychosis, schizophrenia) characterize left temporal lobe abnormalities (Flor-Henry, 1969, 1983; Offen, Davidoff, Troost, & Richey, 2001; Joseph, 1988a; Sherwin, 2005; Schiff et al., 1982; Taylor, 1975; Velakooulis, et al., 2015; Weil, 1956). For example, Gloor et al. (1982) in their depth electrode study of five individuals with seizure disorders, found that all feelings of fear and displeasure were associated with right temporal, right amygdala, or right hippocampal activation. These findings are consistent with the observations of Slater, Beard and Glithero (1969), Bear and Fedio (1977), Flor-Henry (1969) and others who have noted an association between right temporal seizures and affective disorders.
Other reports, however, have been less clear cut -- presumably due to the fact that seizure originating in the amygdala/temporal lobe can quickly spread from one hemisphere to another (via the anterior commissure), and the failure to employ depth electrodes to pinpoint the seizure foci. Hence, in some instances, emotions such as fear seem to arise regardless of which hemisphere is involved (Strauss et al., 1982).
Nevertheless, even in these cases an emotional dichotomy is apparent. For example, Herman and Chambria (1980) have described causes with right temporal foci who developed free floating fears that were not tied to something specific but encompassed terrifying, "death-like" and "nightmarish" feelings, whereas an individual with a left temporal foci developed specific fears of certain individuals and situations. Similarly, Weil (1956) reports that the majority of his patients with right sided foci developed intense fears but were unable to describe what they were afraid of.
As noted, stimulation of the amygdala can significantly alter facial emotional expression, including tearing. In a small minority of cases, right temporal seizures have been reported to cause paroxysmal attacks of weeping, with lacrimation, the making of mournful sounds, including sobbing and crying (Offen et al., 2001). However, crying as well as laughing seizures have also been noted to occur with left sided involvement (Chen & Forster, 1973; Sethi & Rao, 2001). Nevertheless, without the benefit of depth electrodes, such as employed by Gloor and colleagues (1982) it is difficult to determine in which amygdala and/or temporal lobe a seizure actually originates.
SEXUAL SEIZURES
The primate amygdala is sexually differentiated (Nishizuka & Arai, 2005; see also Simerly, 1990). As noted, the male amygdala is larger than the female amygdala (Breedlove & Cooke, 2015; Filipek, et al., 2012), contains a greater number of synaptic connections and shows different patterns of steroidal activity (Breedlove & Cooke, 2015; Nishizuka & Arai, 2005; Simerly, 1990). These sex differences are particularly evident in the medial amygdala, which is also a principle site for steroidal uptake, including the female sex hormone, estrogen, and contains a high concentration of leutenizing hormones (Stopa et al., 1991). The number of immunoreactive cells in the female amygdala also fluctuates during the estrus cycle, being highest during proestrus (SImerly, 1990), and thus presumably acts so that if pregant, the fetus will not be attacked as foreign, and/or so as to coordinate, with the hypothalamus, the appropriate neuroendocrine responses during pregnancy and following birth.
In conjunction with the overlying temporal lobe, the male and female amygdala is capable of detecting sexually significant stimuli, and can determine and detect gender differences, which in turn presumably enables the male amygdala to respond to female-visual and olfactury cues and vice versa.
Because the amygdala is involved in sexuality and is sexually differentiated, activation of the amygdala can produce penile erection (Kling and Brothers, 1992; MacLean, 1990; Robinson and Mishkin, 1968; Stoffels et al., 1980) sexual feelings (Bancaud et al., 1970; Remillard et al., 1983), sensations of extreme pleasure (Olds and Forbes, 2005), memories of sexual intercourse (Gloor, 1986), as well as ovulation, uterine contractions, lactogenetic responses, and orgasm (Backman and Rossel, 2007; Currier, Little, Suess and Andy, 1971; Freemon and Nevis,1969; Warneke, 2001; Remillard et al., 1983; Shealy and Peel, 1957).
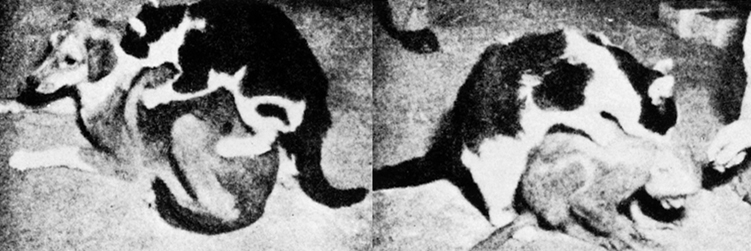
By contrast, injuries to and/or seizure activity within the amygdala/temporal lobe may result in bizarre sexual changes, such as continuous masturbation and indiscriminate, often hypersexual hetero- and homosexual behaviors including attempts at sex with inanimate objects (Kling and Brothers, 1992; Kluver and Bucy, 1939; Pribram and Bagshaw 1953; Terzian and Ore, 1955). Hypersexuality following amygdala injury has been documented among numerous species, including cats and dogs (Blumer 1970; Kling and Brothers, 1992).
Humans with an abnormally activated or severely injured amygdala/temporal lobe may expose and manipulate their genitals (Leutmezer et al., 2015), masturbate in public, and attempt to have sex with family members or individuals of the same sex (Blumer, 1970; Kolarsky, Freund, Macheck, and Polak, 1967; Terzian and Ore, 1955). Moreover, abnormal activity involving the amygdala and overlying temporal lobe has been associated with the the development of hyposexuality (Taylor, 1971; Heirons and Saunders, 1966; Toon, Edem, Nanjee, and Wheeler, 1989), hypersexuality (Blumer, 1970) as well as homosexuality, transvestism, and thus confusion over sexual orientation (Davies and Morgenstern, 1960; Kolarsky et al., 1967).
In fact, abnormal- or seizure activity within the amygdala or overlying temporal lobe may induce an individual to engage in "sexual intercourse" even in the absence of a partner. For example, Currier and colleagues (1971, p. 260) described a female temporal lobe seizure patient who was "sitting at the kitchen table with her daughter making out a shopping list" when she suffered a seizure. "She appeared dazed, slumped to the floor on her back, lifted her skirt, spread her knees and elevated her pelvis rhythmically. She made appropriate vocalizations for sexual intercourse such as: It feels so good...further, further."
FRONTAL LOBE SEXUAL SEIZURES
The frontal lobes, the orbital region in particular, maintain rich interconnections with the amygdala (Gloor, 2014; Nauta, 1964), and receives olfactory projections as well (Tanabe, 1975). Like the limbic system, the frontal lobes, are also sexually differentiated and have "male" vs "female" patterns of dendritic branching which are due to the presence of absence of gonadal hormonal (Kolb & Stewart 1991).
Thus, as is the case with the limbic system, epileptiform activity arising in the deep frontal (orbital) regions have also been associated with the development of "sexual seizures", including exhibitionism, genital manipulation, and masturbatory activity (Spencer et al. 1983).
Similar to that described regarding right temporal emotional and sexual disturbances, Spencer et al., (1983) found that 3 of their 4 patients with sexual automatism had seizures originating in the right frontal area whereas the remaining patient had bifrontal disturbances. Other investigators have also noted that peculiar disturbances in emotion and personality are far more likely to arise following right versus left frontal damage (Joseph, 1986a, 1988a, 2015a; Hillbom, 1960; Lishman, 1968).
Presumably, at least in part, emotional and sexual disturbances associated with right temporal and right frontal (orbital) dysfunction are due to activation of the limbic structures with which they are intimately interconnected. However, as discussed in chapter 12, the inferior temporal and orbital frontal lobes are outgrowths o
AMYGDALA/TEMPORAL LOBE HOMICIDAL HYPERSEXUALITY: A CASE STUDY
In rare instances, temporal lobe and amygdala abnormalities can trigger uncontrolled sexual behavior and violent rage reactions simultaneously; i.e. homicidal hypersexuality. As detailed below, "TL" developed temporal lobe epileptic seizures following a severe head injury and right temporal basilar skull fracture which he suffered at age 15. Immediately after he was released from the hospital, his family noted that TL had undergone a drastic personality change. He became indiscriminately hypersexual, exposing himself and making aggressive sexual overtures toward his mothers and sisters, and suffered attacks of explosive rage.
Over the next several years he continued to deteriorate, his behavior becoming more abnormal. Although attempts were made to treat these aberrant behaviors medically, this was to no avail, and he finally committed an act of extreme sexual savagery. TL brutally beat and strangled his mother to death, in the middle of the day, in between music lessons as one of her students waited outside, and then sexually assaulted her body.
TL readily admitted guilt when arrested, and tried to joke with the interviewing officers, repeatedly spoke of sex, made sexual advances to a nurse, but also frequently apologized for what he had done, repeating in an embarrassed, sometimes silly yet chastised voice: "I feel like such an asshole." TL admitted being sexually attracted to his mother, and further admitted that he attacked her, knocked her to the floor, and throttled her with left hand while savagely beating her in the face and mouth with his fist (spraining his hand, and destroying her face). TL reported he removed her clothes, and then sexually molested the body by kissing and licking her breasts and performing cunnilingus. However, he states he was unable to engage in sexual intercourse because he had become impotent. When the interviewing officers informed him that his mother was dead, he was surprised, and then stated in a dissociative manner: "Oh, what are her music students going to do now?" When the officers accused him of stealing money from his mother's purse, he suddenly became enraged.
After his arrest, TL was housed in the main jail and almost continually behaved in a labile, irrational, enraged, hypersexual, and sometimes silly, childlike fashion. He frequently cursed, threatened and screamed at officers and staff, and would kick and pound on the doors and walls. However, immediately following these episodes he would suddenly become apologetic and childlike, and/or hypersexual and would proposition female staff as well as make up grandiose delusions regarding his accomplishments, wealth or prowess as a lover.
This examiner was appointed by the Santa Clara County Superior Courts, at the request of his attorney, to perform a forensic neuropsychological evaluation of TL for the purposes of determining competency and sanity at the time of offense. During the course of our initial interview TL repeatedly made sexual comments including graphic descriptions of sex acts he claimed to have performed on family members and female guards. He also manifested a hightened startle reaction and appeared to frequently get irritable and angry. When I asked him why he had been arrested, he put his hand over his mouth and giggled: "I killed mom." When I asked if he had removed his mother's clothes after beating her to death, he quite suddenly flew into an uncontrolled rage, and tried to leap across the interview table as he screamed: "I'll rip your fucking head off...." Fortunately, he was chained to his chair. He quickly calmed down, underwent a remarkable personality change, and became meek and apologetic, stating: "I'm sorry. I'm a gentleman. I'll be good... I'm a gentleman." Other than behaving in a silly, labile, hypersexual, and sometimes hostile manner, and making, on occasion, grandiose confabulatory remarks, he generally cooperated with this examiner.
During our second interview, TL claimed he had no memory of having met me. He even commented about having lost his temper with "the other doctor." It is noteworthy that his two sisters independently reported instances in which he failed to recognize them when he approached them on the street.
History Post Head Injury
"TL" suffered a severe head injury at age 15 when he was thrown from a horse and slammed his head against a tree. He immediately lost consciousness and remained in a coma for two months during which he twice developed "spiking fevers." X-Rays indicated a basilar skull fracture and fracture of the right temporal bone. Upon awakening he was partially paralyzed on his right side and demonstrated severe memory loss, right sided visual and hearing problems as well as speech and language difficulties; the latter of which gradually resolved. In addition, he developed partial complex, temporal lobe epilepsy, and suffered occasional grand mal seizures. His temporal lobe epilepsy was controlled with Dilantin.
Upon his return home family noted a remarkable personality change and stated "he was no longer the same person." TL became emotionally labile, easily irritated, and sometimes childlike, and consistently displayed disinhibited and inappropriate sexuality as well as difficulty controlling his temper. Over the next several years he increasingly suffered explosive rage reactions that frequently resulted in physical violence against adults, children, and pets. He also began exposing himself and masturbating, and sexually propositioning or sexually touching his sisters and mother as well as female strangers.
Hypersexuality
According to family and medical records, TL became preoccupied with sex and repeatedly and incessantly made overt verbal and physical sexual advances to female strangers, friends and family members and would attempt to fondle, kiss, and even bite them passionately. He would also intrude into the bedrooms of female family members, proposition them for sex, and would expose himself and masturbate. He allegedly sexually molested a 12 year old girl and was no longer allowed to ride the bus to school due to his propensity for sliding his hands beneath the buttocks or between the legs of female passengers. He later began having frequent sex with prostitutes and would bring them to his place of employment (his father's used car lot) where he would have intercourse in the parking lot. This incessant sexual activity, coupled with his rage ractions, led to his dismissal.
Hyper-Aggressiveness
TL was described as increasingly suffering explosive rage reactions "over nothing." He would shake and "vibrate" with rage and anger. These fits of rage eventually escalated into physical violence, and he would slap, push, and strike friends, family and their children, or engage in acts of animal cruelty; after which he would often panic and run away and then undergo a remarkable personality change and behave as if he had no memory of his outbursts, or he would become childlike and apologetic.
As his family no longer felt safe, he was forced to move into his own apartment. However he was soon evicted due to his inappropriate sexual behavior and as he physically assaulted a security guard. Several weeks later he attacked a supermarket security guard, beating and wrestling him to the ground. He was arrested for battery but bail was posted and he was later released. Several evenings later he confronted, began screaming at, and then assaulted several police officers who had stopped his car which he had been driving erratically and with his automobile lights off. He was admitted for a 72 hour psychiatric observation. Six weeks later, he attacked his mother during the middle of the day, between music lessons, as a student waited outside. He shoved his mother to the floor, and strangled her as he beat her to death with his fist. He then removed her clothes and sexually assaulted the body.
EEG, MRI, SPECT, Neuropsychological Exam By history, temporal lobe seizure activity was repeatedly documented. Following his arrest, and upon the request of this examiner, TL was administered an EEG, MRI, and a brain SPECT scan. Several EEG's were administered, and revealed theta slowing over the left temporal lobe and intermittent spiking over the right temporal lobe. The SPECT revealed decreased perfusion and severe, extensive, and widespread atrophy and abnormal activity of the right lateral and inferior-anterior-medial temporal lobe, including the amygdala. The MRI indicated bilateral temporal lobe as well as frontal lobe atrophy. Neuropsychological testing indicated right frontal, right temporal lobe, and right hippocampal and right amygdala injury, as well as severe disturbances of immediate and short-term verbal narrative memory and especially visual memory, including an inabilty to recognize pictures of faces that he had been shown just minutes before, or to discriminate them from pictures he had not seen. Moreover, during the second test session, he failed to recognize this examiner (though he remembered being examined) and even failed to recognize pictures of his own face--prosopagnosia.
Hence, based on the above, it appears that his hypersexual homocidal behavior was a direct consequence of the injuries to the temporal lobe, amygdala, and frontal lobe, coupled with seizure activity involving these structures.
THE AMYGDALA, THE ANTERIOR COMMISSURE, SEXUALITY & EMOTION,
When the amygdala or the bed nuclei for the anterior commissure of both cerebral hemispheres are damaged, hyperactivated, or completely inhibited a striking disturbance in sexual and social behavior is evident (Brown & Schaffer, 1888; Gloor, 1960; Kluver & Bucy, 1939; Terzian & Ore, 1955; Schriner & Kling, 1953). Specifically, humans, non-human primates, and felines who have undergone bilateral amygdalectomies will engage in prolonged, repeated, and inappropriate sexual behavior and masturbation including repeated sexual acts with members of different species (e.g. a cat with a dog, a dog with a turtle, etc.). When activated from seizures, patients may involuntarily behave in a sexual manner and even engage in what appears to be intercourse with an imaginary partner. This abnormality is one aspect of a complex of symptoms sometimes referred to as the Kluver-Bucy syndrome.
As noted, portions of the hypothalamus and amygdala are sexually dimorphic; i.e. there are male and female amygdaloid nuclei (Bubenik & Brown, 1973; Nishizuka & Arai, 2005). In humans the male amygdala is 16% larger (Filipek, et al., 2012), and in male rats the medial amygdala is 65% larger than the female amygdala (Breedlove & Cooke, 2015), and the male amygdala grows or shrinks in the presence of testosterone--findings which may be related to sex differences in sexuality and aggression. Moreover, female amygdala neurons are smaller and more numerous, and densely packed than those of the male (Bubenik & Brown, 1973; Nishizuka & Arai, 2005), and smaller, densely packed neurons fire more easily and frequently than larger ones--which may contribute to the fact that females are more emotional and more easily frightened than males (chapters 7,13,15), as the amygdala is a principle structure involved in evoking feelings of fear (Davis et al., 2014; Gloor, 2014; LeDoux, 2013).
Moreover, despite myths to the contrary, females, regardless of species, are more sexually active than males, on average (see chapter 8)--that is, when they are in estrus-- and the human female is capable of experiencing multiple orgasms of increasing intensity--which may also be a function of sex differences in the amygdala. That is, since female primate amygdala neurons are more numerous and packed more closely together (Bubenik & Brown, 1973; Nishizuka & Arai, 2005), and as smaller, tightly packed neurons demonstrate enhanced electrical excitability, lower response thresholds, and increase susceptibility to kindling and thus hyper-excitation, the amygdala therefore is likely largely responsible for sex differences in emotionality and sexuality.
Indeed, electrical stimulation of the medial amygdala results in sex related behavior and activity. In females this includes ovulation, uterine contractions and lactogenetic responses, and in males penile erections (Robinson & Mishkin, 1968; Shealy & Peele, 1957). Moreover, in rats and other animals, kindling induced in the amygdala can trigger estrus and produce prolonged female sexual behavior.
Moreover, the anterior commissure, the band of axonal fibers which interconnects the right and left amygdala/temporal lobe is sexually differentiated. Like the corpus callosum, the anterior commissure is responsible for information transfer as well as inhibition within the limbic system. Specifically, the female anterior commissure is 18% larger than in the male (Allen & Gorski 1992). It has been argued that the increased capacity of the right and left female amygdala to communicate (via the anterior commissure) coupled with the more numerous and more densely packed neurons within the female amygdala (which in turn would decrease firing thresholds and enhance communication), and the sex diffferences in the hypothalamus, would also predispose females to be more emotionally and socially sensitive, perceptive, and expressive (Joseph 1993). Hence, these limbic sex differences induces her to be less aggressive and more compassionate and maternal, and affects her sexuality, feelings of dependency and nurturance, and desire to maintain and form attachments in a manner different than males.
In contrast, whereas the right and left female amygdala are provided a communication advantage not shared by males, the "male" amygdala in turn may be more greatly influenced by the (medial) hypothalamus via the stria terminalis which is larger in men than women (Allen & Gorski 1992). As noted, the male medial amygdala is larger than its female counterpart (Breedlove & Cooke, 2015) and changes in size in response to testosterone, which is significant as the medial nuclei (and testosterone) is directly implicated in negative and aggressive behaviors (see above).
Although environmental influences can shape and sculpt behavior and the functional organization of the brain (chapter 28), most sex differences are innate and shared by other species (see chapters 7 & 8); a direct consequence of the presence or absence of testosterone during adulthood and fetal development (see Gerall et al. 1992; Joseph 1993, Joseph et al. 2002) and the sexual differentiation of the limbic system.
FEMALES ARE MORE EMOTIONAL, RELIGIOUS, NURTURING, etc.
To recapitulate, since the limbic system is concerned with emotion, and since the female amygdala appears to be more emotional, more maternal, and more sexual whereas the male limbic system is more greatly influenced by the activating and aggression inducing hormone, testosterone, sex differences in these and other limbic structures may account for why women and females of many species are more nurturant, empathetic, sympathetic, self-sacrificing, and emotionally sensitive and compassionate (Bakan, 1966; Belle, 1982; Blakemore, 1990; Berman, 1983; Eagly & Steffen, 2007; Fresbach, 1982; Graham, 1988; Hoffman, 1977). Human females have a richer inner emotional life and are better able to understand, perceive, express social-emotional nuances as compared to males (Burton & Levy, 1989; Brody, 1985; Buck, 1977, 2007; Buck et al. 1974, 1982; Card et al. 1986; Eisenberg, et al. 1989; Fuchs & Thelan, 1988; Harackiewicz, 1982; Kemper, 2002; Lewis, 1983, Rubin, 1983; Shennum & Bugental, 1982; Soloman & Ali, 1972; Strayer, 1985). Females are also more willing to express emotional issues and confide and discuss personal problems with others (Gilbert, 1969; Gilligan, 1982; Demos, 1975; Lutz, 1980; Pratt, 1985; Walker, et al. 2009; Parelman, 1980; Lombardo & Levine, 2005), and are much more likely than a male to cry (de Beauvoir 1952; Thomas, 1993), or to freeze, panic, or run away in fear -functions and behaviors directly associated with amygdala activation.
Even female memories are more emotional and more inclined toward social and interpersonal concerns (Pratt, 1985; Walker, et al. 2009; Friedman & Pines, 1991). Hence, women are superior to men in recalling emotional memories (Pratt, 1985; Walker, et al. 2009; Friedman & Pines, 1991) and can recall events that many men swear did not even occur. In this regard, it is noteworthy that dendritic spine density in the female (rat) hippocampus increases and decreases by as much as 30% during each estrus cycle (Woolley, et al., 1990) which in turn may influence memory. This may explain why estrogen replacement therapy slows memory loss in women and results in a 54% lower chance of developing Alzheimers (see chapter 16).
In contrast, males have difficulty discussing personal difficulties or expressing their emotions other than anger and happiness (Balswick, 1982, 1988; Campbell, 2000; Goldberg, 2001; O'Neil, 1982; Joseph, 1992b, 1993; Sattel, 1989) and are much more inclined to develop psychopathological conditions such as sociopathy (Draper & Harpending, 1988). Many male criminals are sociopaths and sociopaths in general have very little human regard, empathy, compassion, or concern for others.
As will be discussed in further detail in chapter 9, activation of the amygdala-temporal lobes are also associated with the capacity to have religious experiences. Sex differences in this structure may explain why women are not just more emotional, but have more intense religious experiences, attend church more often, are more involved in religious activities, involve their children more in religious studies, hold more orthodox religious views, incorporate religious beliefs more often in their daily lives and activities, and pray more often as well (Argyle & Beit-Hallahami, 1975; Batason & Ventis, 1982; De Vaus & McAllister Glock et al. 1967' Lazerwitz, 1961; Lindsey, 1990' Sapiro, 1990). Presumably this sex difference too is a consequence of sex differences in the structure and function of the limbic system, the amygdala and anterior commissure in particular.
It is tempting to speculate that these limbic system sex differences may also predispose females to become more easily emotionally stressed and upset -which in turn might affect the functioning of the hypothalamus and other nuclei thereby giving rise to a greater incidence of depression (including SADs), as well as hysteria, and related disorders including "chronic fatigue syndrome;" conditions which are more common among women than men. Indeed, in almost all surveys of developed countries, females suffer from two to three times as many mood disorders as men (Mollica 1989).
MALE AGGRESSION IS A SOURCE OF MALE PLEASURE
Due to sex differences in the size and functioning of the hypothalamus and medial amygdala, the effects of testosterone on these structures, and given that these limbic nuclei control the ability to behave violently or experience pleasure or displeasure as well as sexual orgasm, some behaviors, like nurturance or aggression, are rewarding in themselves depending if one possess a male vs a female limbic system (Joseph 1992a, 1993). That is, males and females experience pleasure in response to different experiences; e.g. holding and nurturing a baby vs watching a boxing match or playing a violent "video game" (e.g. Campbell, 2015). For example, male humans, chimpanzees, birds, fowl, and fish, will look for fights and opportunities to behave antagonistically or destructively because they experience pleasure when they behave aggressively. Males frequently seek aggressive competitive situations and the opportunity to behave combatively even in otherwise neutral environments, and this is true of male rats, mice, fish, chimpanzees, and humans (Calhoun 1950; Goodall, 1986; Hamburg, 1971; Johnson 1972; Lorenz, 1966; Manning, 1972; McGrew & McGrew, 1970; Mitchell, 1979; Moyer 1974).
In learning situations where subjects are able to control their number of antagonistic encounters, males tend to chose situations where they can behave belligerently much of the time (Jonhson 1972). Even male mice will learn a maze when the only reward is the opportunity to attack another mouse (Tellegen et al. 1969). Females tend to have no such desire except under certain conditions, e.g. threat to her brood, or the presence of another sexually receptive (attractive) female.
It is not uncommon for gangs of young human males to break and destroy things just for the pleasure of the destruction or to simply look for a fight or the opportunity to attack someone (McGrew & McGrew, 1970; Sears & Beach, 1965). Male chimpanzees often look for opportunities to attack and put much energy into creating encounters with strangers from neighboring troops. In fact, they often make raids into the core areas of neighboring groups just to fight and even kill one another. According to Goodall (1986), "these encounters are highly attractive to the participants. To some extent all adult males show a keenness to participate in these exciting incidents." In fact, male chimps will become extremely aroused by violence and fights occurring among members of their own troop and may charge in and attack innocent bystanders as well as join the general melee simply for the sake of fighting (Goodall, 1986). Human males responding likewise have been the fare of media depictions of bar room brawls since the inception of motion pictures.
Indeed, almost regardless of species, be it male deer, oxen, seals, walruses, wolves, sperm whales, hippopotamus, or even Australian marsupials, males not only derive more enjoyment from aggression, but they are far more aggressive and violent than females in general (Elia, 1988; Fedigan, 1992; Goodall 1986; Hamburg, 1971; Johnson 1972; Lorenz, 1966; Manning, 1972; Mitchell, 1979; Moyer 1974). These differences are also characteristic of the majority of social primates including humans.
Over 80% of all violent crimes and murders are committed by males (Elliott et al. 1986; Uniformed Crime Reports, 1990-2014). Even when considering violence prone repeat criminal offenders, males continue to commit almost twice the amount of violent crimes as compared to violent females (Elliott et al. 1986).
Similarly, male chimpanzees initiate attack five times as frequently as females (Van Lawick-Goodall, 1968) and adult male chimps engage in 90% of all aggressive behaviors (Bygott, 1979). Moreover, these differences are are found among male baboons, gorillas, rhesus monkeys, (Mitchell, 1979; Fedigan 1992; Elia 1988; Jolly 1972) as well as chimps who are reared in a nursery with peers only (Nadler & Braggio, 1974) and those raised in social isolation (Sackett, 1974). Indeed, sex differences in violence and belligerence are apparent even in infancy and appear as early as the first two months of life even in chimps and other primates (Ransom & Rowell, 1972; Mitchell, 1979). Hence, male aggression is not learned, per se, but is an innate, limbic characteristic that is common among most species. However, this is not to say that females are not aggressive for this is not the case (Thomas, 1993).
THE LIMBIC SYSTEM & TESTOSTERONE
In large part these and related sex differences in aggressiveness are also a consequence of the relatively higher concentrations of the activating hormone, testosterone flowing through male bodies and brains. The overarching influence of neurological and hormonal predispositions are also indicated by studies which have shown that females who have been prenatally exposed to high levels of masculinizing hormones (i.e. androgens) behave similar to males even in regard to spatial abilities (Joseph et al. 2002; see Gerall et al. 1992). They are also more aggressive and engage in more rough and tumble play as compared to normal females (Money & Ehrhardt, 1972; Ehrhardt & Baker, 1974; Reinisch, 1974) and this is also true of other species such as dogs, wolves, gorillas, baboons, and chimpanzees.
Similarly, female primates and mammals who have been exposed to testosterone during neonatal development display an altered sexual orientation, as well as significantly higher levels of activity, competitiveness, combativeness and belligerence (Mitchell, 1979). Nevertheless, it is important to re-emphasize that it is generally the presence or absence of testosterone during the critical period of neuronal differentiation which determines if one is in possession of a "male" vs "female" limbic system.
SEXUAL ORIENTATION & HETEROSEXUAL DESIRE
As noted, the amygdala surveys the environment searching out stimuli, events, or individuals which are emotionally, sexually or motivationally significant. Moreover, it contains facial recognition neurons which are sensitive to different facial expressions and which are capable of determining the sex of the individual being viewed and which become excited when looking at a male vs female face (Leonard et al. 1985; Rolls 2007). In this regard, the amygdala can act to discern and detect potential sexual partners and then motivate sex-appropriate behavior culminating in sexual intercourse and orgasm.
That is, an individual who possess a "male" limbic system is likely to view the female face, body and genitalia as sexually arousing because the amygdala and limbic system responds with pleasure when stimulated by these particular features. Conversely, male physical features are likely to excite and sexually stimulate the limbic systems possessed by heterosexual females and homosexual males (Joseph, 1993). This is because, at a very basic level emotional, sexual, and motivational perceptual/behavioral functioning becomes influenced and guided by the anatomical sexual bias of the host.
THE HOMOSEXUAL LIMBIC SYSTEM
Homophobia
Homosexuality, and "homo-phobia" are not uncommon among males of many different species. Often, however, what appears to be "homosexuality" is in fact a dominance display, with the dominant animal mounting the submissive. In some instances, such as involving multi-male/female primates societies, subordinate males and females will present their derriere to a superior, for mounting, in which case the dominant may simulated sexual intercourse. In some instances, however, an animal will attempt to mount another, so as to establish dominance, and in consequence is violently assaulted, and may be mounted in turn. Among humans this is sometimes referred to as "homophobia." That is, a heterosexual male may respond with a violent attack in reaction to a homosexual overture, so as to establish that it is HE who is dominant--and this "homophobic" male may then force the "victim" to perform fellatio, or the victim's genitals may be attacked.
As I have noted elsewhere (Joseph, 1992b), in some instances of "homophobic" violence, the attacker is in fact beating upon his own unknown face. Homosexuals are often beat up and attacked by other homosexuals--including those males who do not know, but who fear that they, themselves may be "queer." These latter males may in fact go out in search of a "queer" to beat up, and may force the "queer" to perform fellatio, or even perform anal sex on the victim.
A great deal of "gay" violence, is in fact perpetrated by homosexuals, including those who know they are homosexual, and who are in homosexual relationships. As an illustrative example, a San Francisco homosexual organization, "Community United Against Violence" and which tracks "anti-gay violence" has repeatedly reported, beginning in 1995 when they first began tracking these behaviors, that same-sex domestic violence exceeded "anti-gay violence." Same-sex domestic violence, and this and other homosexual organizations admit, is significantly underreported. Moreover, whereas homosexual domestic violence actually involves violent physical assaults and frequent battering (Lobel, 1988), the above and other homosexual groups, such as the National Coalition of Anti-Violence Programs, considers "slurs" or "yelling out a car windo" to be a form of "anti-gay" violence. Hence, actual physical violence against homosexual is predominantly committed by homosexuals.
In some instances, homosexual violence against homosexuals is due to the abusive childhoods some of these individuals experienced. Indeed, in some instances, severe abuse and emotional trauma, may in fact have mitigating factor in the development of homosexuality in these and other "homosexuals" and bi-sexuals (Finkelhor, 1979; Johnson and Shrier, 1985; Simari & Baskin, 2007).
Stress, Abuse, and Homosexuality
A significant number of males with a history of childhood sexual abuse (CSA) report sexual disturbances (Finkelhor, 1979; Johnson & Shrier, 1985; Rogers & Terry, 2007), including hypo or hypersexuality, compulsive masturbation, and difficulty forming sexual relationships or performing adequately with a female sex partner (Duncan & Williams, 1998; Elliott & Briere, 1992; Hunter, 1991). Some sexually abused males experience considerable confusion over their sexual identity and engage in homosexuality (Johnson and Shrier, 1985; Simari & Baskin, 2007). Finkelhor (1979) reported that males with a history of CSA are four times more likely than non-abused males to engage in homosexual activities. In addition, a significant relationship between CSA and teenage- or adult-onset pedophilia and violent criminal behavior has been reported (Haaspasalo & Kankonon, 2014; Knight & Prentky, 1993; Rubinstein et al., 1993; Watkins and Bentovim, 1992; Widom and Ames, 2012), including violence toward loved ones and intimate partners (Duncan and Williams 1998). As is also the case with women, men who were sexually abused suffer significant emotional problems, including depression, fear, anger, and anxiety, and not infrequently experience homicidal, suicidal, and self-destructive feelings (Mendel 1995; Urquiza and Capra, 1990; Watkins and Bentovim, 1992); emotions, feelings, and behaviors which approximately 30% of those who are "homosexual" also report (see below). Indeed, approximately 30% of male and female homosexuals have significantly alcohol, drug, and serious mental disorders (Family Research Counsel, 1998; Lewis, et al., 1982; Zehner & Lewis, 1985; Ziebold & Mogenson, 1982), and frequently batter their sex partners (Lobel, 1988).
Perhaps not surprisingly, men with a history of CSA, that is, those who have been abused by men rather then women, often become exceedingly homophobic (Urquiza and Capra, 1990). Of those who claim that the "abuse" was a positive experience, these males may well have been homosexual in orientation to begin with (e.g. Johnson and Shrier, 1985).
Homosexuality probably does not have a single "cause." That is, some individuals may become homosexual because of a genetic predisposition (Bailey et al., 1993; Byne & Parsons, 1993), whereas others may engage in these behaviors because they were sexually abused, whereas yet others may have become homosexual due to the profound effects of stress and stress hormones on the limbic system. That is, in these latter instances, the sexual organization and thus the "sexual orientation" of the limbic system may have become altered, and may have assumed the female pattern of sexual differentiation due to profound fetal, neonatal, or childhood stress (e.g., Meyer-Bahlburg, 1993; Reinisch & Sanders, 1992). In fact, because of limbic system sensitivity to circulating stereoids, chronic stress may in fact induce steroidal alterations in the functional organization of the hypothalamus and amygdala, thereby, possibly inducing alterations in sexuality, including changes in sexual orientation and related cognitive activities (e.g., Joseph et al., 2002; Meyer-Bahlburg, 1993; Reinisch & Sanders, 1992).
Specifically, chronic stress can alter the binding of testosterone, and can prevent testosterone from binding with hypothalamic and amygdala neurons responsible for sexual behavior (Raab & Haedenkamp, 2005). If the fetus is subject to considerable stress, for example, if "mother" is in an abusive relationship or subject to other profound stress, the secretion of stress-steroids, may effect the sexual differentiation and the development of the limbic system and induce the female pattern of neural development. That is, as these stress-steroids, e.g., cortisol and aldosterone, are not true androgens, rather than inducing a male pattern, they may block the reception and binding of fetal testosterone, thereby inducing the female developmental pattern. In fact, the secretion of cortisol and aldosterone--particularly if prolonged--can reduce the secretion of gonadropins (Moberg, 1985), which would also interfere with male sexual behavior and the development of the male pattern of neural development.
As noted, it has been shown that the ventromedial and anterior nuclei of the hypothalamus of male homosexuals demonstrate the female pattern of development (Levay, 1991; Swaab, 1990); sex-specific patterns which may have been induced by stress. If these stress induced steroidal changes also negatively impact the amygdala, affected individuals may not only become homosexual, or sexually and emotionally abnormal, but later engage in self-destructive sexual activities including prostitution and indiscriminate homosexuality (see chapter 28).
Corticosteroids when secreted at high levels, can in fact induce wide spread neural injury and abnormal neuroplastic changes throughout the brain. Pyramidal neurons in particular are especially vulnerable to the deleterious effects of stress and high levels of corticosteroids, especially those located in limbic system structures due the abundance of Type II adrenal steroid receptors which abound within this tissue (Lupien & McEwen, 2014; Pugh, Fleshner, & Rudy, 2014). Corticosteroids at high levels can exert a suppressive influences on membrane receptor proteins, thereby altering excitability and information transmission between neurons (Hua & Chen, 1989; Majewska, Harrison, Schwartz, Barker, & Paul, 1986), and can detach the cellular receptor from its attached protein (Beaulieu, 2009); a condition which interferes with messenger RNA protein transcription and thus the genetics of neural differentiation and neural plasticity, including, perhaps, those related to the sexual differentiation of the brain.
Moreover, prolonged, repetitive, profound stress, can induce abnormal activity involving the amygdala (see chapters 28, 29, 30), including seizure-like activity referred to as kindling. Abnormal amygdala (and overlying temporal lobe) activity has been associated with the development of hyposexuality (Taylor, 1971; Heirons and Saunders, 1966; Toon, Edem, Nanjee, and Wheeler, 1989), hypersexuality (Blumer, 1970) as well as homosexuality, transvestism, and thus confusion over sexual orientation (Davies and Morgenstern, 1960; Kolarsky et al., 1967). In fact, abnormal- or seizure activity within the amygdala or overlying temporal lobe may induce an individual to engage in "sexual intercourse" even in the absence of a partner.
Coupled with animal studies which demonstrate that sex specific behaviors and cognitive activities can be enhanced or altered by steroids (Joseph et al., 2002; Reinisch & Sanders, 1992) or suppressed due to early environmental influences, including the stress of deprivation (Joseph, 1979; Joseph and Gallagher, 1980), and given that the sexual differentiation of the hypothalamus may be altered by steroidal manipulations (Raisman and Field, 1973), it can also be assumed that traumatic stress and the massive secretion of steroids, particularly during early sexual development, may alter the neural organization of these same structures. In consequence, sexuality, sexual orientation, as well as emotion, personality, including the ability to cope with stress may be disrupted and become abnormal among those who have been severely or repetitively sexually abused and traumatized.
The Homosexual Limbic System.
As noted above, some men become homosexual or bisexual due to sexual abuse experienced as children (Finkelhor, 1979; Johnson and Shrier, 1985; Simari & Baskin, 2007), whereas in others there is a suggestion of a genetic contribution (Bailey et al., 1993; Byne & Parsons, 1993). Nevertheless, it has been reported that the hypothalamus (i.e. the ventromedial and anterior nuclei) in male homosexuals is organized in a manner similar to the female pattern of development (Levay, 1991; Swaab, 1990). Moreover, the anterior commissure is larger not only in females, but is 35% larger in homosexual males vs male heterosexuals (Allen & Gorski, 1992). Coupled with the evidence reviewed above, this raises the possibility that sex differences in male vs female and male vs homosexual male sexual orientation and thus the capacity to experience sexual pleasure when with a heterosexual or homosexual partner, may be determined by these nuclei.
For example, it could be assumed that homosexual males respond to males with feelings of sexual attraction and desire, as do heterosexual females, i.e. because they are in possession of a "female" limbic system which responds to male physical and facial features with sexual arousal; i.e. the amygdala contains neurons which respond to faces, facial expressions, and which can determine the sex of the individual viewed (e.g. Leonard et al. 1985; Rolls 2007). Conversely, heterosexual males, being in possession of a "male" hypothalamus and amygdala, not only respond to females with sexual arousal, but they behave and act more aggressively than females and homosexual males.
Presumably, it is because homosexuals have a "female" limbic system they they are thus more inclined to behave in a manner similar to women than to men. Indeed, homosexual males (in general) and females tend to be more alike than different in regard to social-emotional reactions and tendencies (Tripp 2009). In some cases these feminine tendencies are grossly exaggerated (Tripp, 2009); i.e. the "swishy" male with the exaggerated high pitched voise.
A significant number of homosexuals, in fact, are psychologically similar to females in a number of ways, including having a high interest in fashion and wearing apparel, a pronounced tendency to employ feminine body language and vocal tones, to shun sports and avoid fights, and to have a fear of physical injury, particularly during childhood (Bell et al. 2005; Bieber et al. 1962; Van Den Aardweg, 2007; Tripp, 2009). Many also tend to maintain intense dependency relations with their mothers and to remain distant from strong male figures including their fathers (Green, 2009); heterosexual males (including male primates; e.g. Fedigan, 1992; Goodall, 1986) tend to behave in a completely different fashion and are far more likely to seek the company of males and men and to engage in rough and tumble activities.
As children homosexual males tended to prefer female companions and friends, girls toys, activities, and often girls clothes, and behaved in an effeminate manner (Bell, et al. 2005; Saghir & Robins, 1973; Grellet et al. 1982; Green, 2009). Indeed, from 67% to 75% of homosexuals vs 2%-3% of heterosexual males reported being "feminine" and more like girls than boys as children (Saghir & Robins, 1973; Green, 2009). Moreover, homosexual males, like heterosexual females, demonstrate comparatively inferior spatial perceptual capabilities as compared to heterosexual males (Gladue et al. 1990; Sanders & Ross-Field, 1986; Wilmot & Brierley, 2007). Homosexuals tend to perform similarly to females on these spatial tests.
It is tempting to speculate that because some homosexuals demonstrate an almost hyper-developed pattern in the structure of the anterior commissure (and thus presumably the amygdala, as well as the hypothalamus), that this may account for why a significant minority of these individuals engage in excessive, dangerous and sometimes "bizarre" (e.g. "fist fucking") sexual behaviors, including indiscriminate promiscuity, "orality," "anality," and a proclivity to engage in group oral/anal sex, or to "cruise" and repeatedly have sex with strangers (sometimes in a single evening). It has been reported that between 24% to 30% of this population have repeatedly engaged in truly reckless and self-destructive sexual behaviors during their youth (Aaron, 1973; Gans, 1993; Pollak, 1993; Symons 1979; Tripp, 2009) --manifestations of the Kluver-Bucy syndrome?
Even at its most benign, male-homosexual sexual-encounters often involve strangers having unprotected sex with strangers. And among younger homosexuals "gay love" is stereotypically often little more than one stranger ejaculating into the anus or the mouth of another stranger, who then swallows or spits, and then goes on his way. Of course many heterosexual males would be happy to ejaculate into the mouth or vagina or a beautiful female stranger. Moreover, like heterosexuals many homosexuals form "long-term" male-male relationships. However, even in so called "committed" homosexual relationships, promiscuity (at least among younger homosexuals) is common (Tripp, 2009).
The hyper-developed pattern of limbic system development, coupled perhaps with abuse/stress-induced abnormalities may also account for the high incidence of emotional disorders including suicide in homosexuals. It has been estimated that approximately 30% of the homosexual population suffer from significant alcohol and drug addiction and/or psychopathology (Family Research Counsel, 1998; Lewis, et al., 1982; Zehner & Lewis, 1985; Ziebold & Mogenson, 1982). Of course, a significant number of homosexuals are exceedingly high functioning, earning six and seven figure incomes, and are devoid of any evidence of pathology or severe emotional disturbances.
Nevertheless, as compared to the general population, severe and profound psychological problems are over represented among homosexuals as a group (Family Research Counsel, 1998; Lewis, et al., 1982; Lobel, 1988; Zehner & Lewis, 1985; Ziebold & Mogenson, 1982), which in turn may be related to possible limbic system abnormalities. This is not to negate the impact of social-environmental influences on the development of these "abnormalities" as the environment can have a significant impact on one's self concept (Joseph 1992b). That is, feeling different, being victimized, rejected, taunted, tormented, isolated, and so on, is in-itself exceedingly stressful and debilitating, and it would be expected that those victimized in this fashion would experience considerable self-hate, anger, depression, and so on, and would perhaps be inclined to engage in self-destructive behaviors.
OVERVIEW: THE AMYGDALA
Over the course of early evolutionary development, the hypothalamus reigned supreme in the control and expression of raw and reflexive emotionality, i.e., pleasure, displeasure, aversion, and rage. Largely, however, it has acted as an eye turned inward, monitoring internal homeostasis and concerned with basic needs. With the development of the amygdala, the organism was now equipped with an eye turned outward so that the external emotional features of reality could be tested and ascertained. When signaled by the hypothalamus the amygdala begins to search the sensory array for appropriate emotional-motivational stimuli until what is desired is discovered and attended to.
However, with the differentiation of the amygdala, emotional functioning also became differentiated and highly refined. The amygdala hierarchically wrested control of emotion from the hypothalamus.
The amygdala is primary in regard to the perception and expression of most aspects of emotionality, including fear, aggression, pleasure, happiness, sadness, etc., and in fact assigns emotional or motivational significance to that which is experienced. It can thus induce the organism to act on something seen, felt, heard, or anticipated. The integrity of the amygdala is essential in regard to the analysis of social-emotional nuances, the organization and mobilization of the persons internal motivational status regarding these cues, as well as the mediation of higher order emotional expression and impulse control. When damaged or functionally compromised, social-emotional functioning becomes grossly disturbed.
The amygdaloid nucleus via its rich interconnections with other brain regions is able to sample and influence activity occurring in other parts of the cerebrum and add emotional color to ones perceptions. As such it is highly involved in the assimilation and association of divergent emotional, motivational, somesthetic, visceral, auditory, visual, motor, olfactory and gustatory stimuli. Thus it is very concerned with learning, memory, and attention, and can generate reinforcement for certain behaviors. Moreover, via reward or punishment it can promote the encoding, storage and later retrieval of particular types of information. That is, learning often involved reward and it is via the amygdala (in concert with other nuclei) that emotional consequences can be attributed to certain events, actions, or experiences, as well as extracted from the world of possibility so that it can be attended to and remembered.
Lastly, as is evident from studies of individuals with abnormal activity or seizures originating in or involving this nuclei, the amygdala is able to overwhelm the neocortex and thus gain control over behavior. As based on electrophysiological studies, the amygdala seems capable of literally turning off the neocortex (such as occurs during a seizure) at least for brief time periods. That is, the amygdala can induce electrophysiological slow wave theta activity in the neocortex which indicates low levels of arousal (see below) as well as high voltage fast activity. In the normal brain it probably exerts similar influences such that at times individuals (i.e. their neocortex) "lose control" over themselves and respond in a highly emotionally charged manner.
In consequence, after they "explode" or respond "irrationally" they (that is, the neocortex of the left hemisphere) are likely to wonder aloud: "I don't know what came over me."
But we know the answer: The Limbic System.













