Rhawn Gabriel Joseph, Ph.D.
BrainMind.com
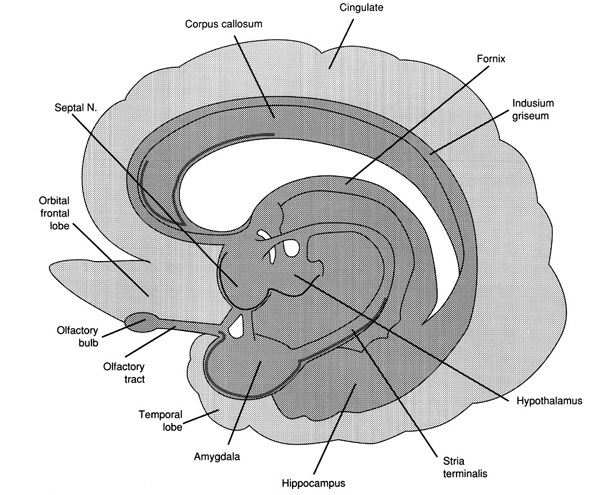
The Amygdala
The amygdala is concerned with all aspects of emotion, including fighting, fleeing, feeding, and fornicating. Thus the amygdala is greatly concerned with emotional behavior and stereotyped emotional movements.
Early in the course of vertebrate evolution, motor functioning was the province of the spinal cord, brainstem, cerebellum, and forebrain (see chapter 5), just as it is in modern species including humans. Much of the forebrain, however, consisted of the olfactory bulbs and tracts, and the amygdalostriatal gray which was both dorsal and venterally located and contained at its central-posterior-dorsal core, the hippocampus. The amygdalostriatal gray was dominated by the olfactory system, and reacted to olfactory impulses by feeding, fighting, fleeing, or engaging in sexual behavior. Although the amygdalostriatal gray became separate structures once terrestrial vertebrates had evolved (Gloor, 1997, MacLean, 2010; Stephans, 2014; Ulinsky, 2010), the striatum and related structures collectively referred to as the basal ganglia, continued to govern all aspects of affectively triggered gross motor behavior, just as they do in modern humans, albeit in conjunction with the brainstem, cerebellum, spinal cord and cranial nerve nuclei, as well as the thalamus, and the primary, secondary and supplementary motor areas of the frontal lobes.
Structural Overview
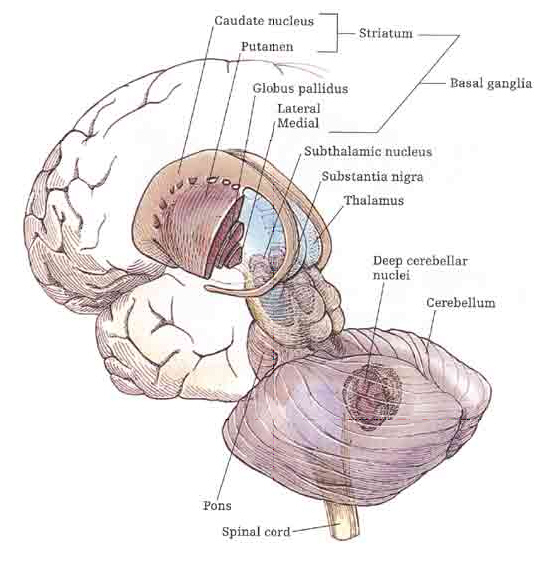
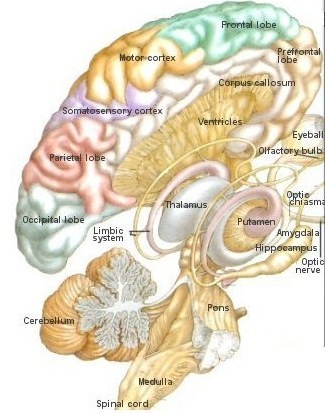
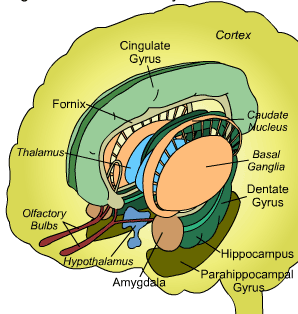
Thus the amygdala is part of the basal ganglia which is composed of several major nuclei which include the corpus (or dorsal) striatum ("striped bodies") i.e., the caudate and putamen which are extensively interconnected and which project to a variety of brain areas including the immediately adjacent globus pallidus ("pale globe"). The dorsal globus pallidus although related to the hypothalamus and midbrain, is in many respects coextensive and appears to merge with the putamen giving the entire structure the appearance of a camera lens.
The dorsal striatum is also intimately related to the amgydala, receiving massive input from this structure but providing little in return.
As noted the amydalostriatal gray consisted of both dorsal and ventral components, and so too does the human striatum--also referred to as the limbic (ventral) striatum. The substantia innominata, nucleus accumbens, olfactory tubercle constitute the limbic striatum. The inferior ventral globus pallidus (also referred to as the ventral pallidum or substantia innominata) is also part of the limbic (ventral) striatum and in fact eventually merges with the centro-medial amygdala and receives extensive projections from the lateral amygdala, the olfactory tubercle, and nucleus accumbens.
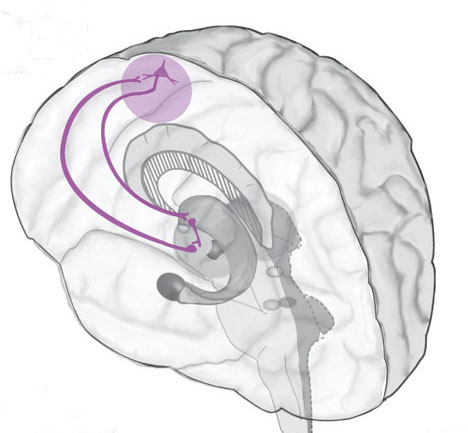
In addition, the "motor thalamus," the orbital and medial frontal lobes and medial supplementary motor area, hippocampus (as well as the central and medial amygdala), are richly interconnected with and constitute major components of the basal ganglia. However, as noted in chapter 12, the basal ganglia (i.e. the corpus and limbic striatum and globus pallidus) evolved out of the olfactory-amygdala and in many respects it could be considered part of the limbic system (Heimer & Alheid, 2011; MacLean, 2010).
Intimately linked and thus part of the basal ganglia is the substantia nigra and the midbrain tegmentum, which feed dopamine to the corpus and limbic striatum. Specifically, the nigrostriatal DA (A-9) cell group projects from the substantia nigra to the dorsal caudate and putamen and the medial frontal lobes (Ellison, 1994; Fibiger & Phillips, 1986; Parent & Hazrati 2013), although some fibers also innervate the limbic striatum. It is the nigro-(dorsal)-striatal system which is thought to be related to motor functions including the production of stereotyped and routine actions.
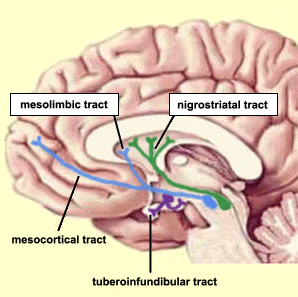
The mesolimbic DA systems originates in the ventral midbrain tegmentum (A10 DA cell group) and sends fibers to the amygdala, septal area, hippocampus, frontal cortical areas, including the ventral caudate-putamen, nucleus accumbens and substantia innominata (Le Moal & Simon 2011; Olton et al. 2011; Zaborszky et al. 2011), although some fibers also innervate the dorsal striatum. The mesolimbic DA system is believed to be related to emotion, mood, memory, and reward, including locomotor survival related activities such as running and even "galloping" (Ellison, 1994; Fibiger & Phillips, 1986; Le Moal & Simon 2011).
THE AMYGDALA, EMOTION, MEMORY, PSYCHOSIS, & THE BASAL GANGLIA
As detailed in chapter 13, early in the course of animal evolution, the amydala and striatum formed a composite dorsal/ventral amygdala-striatum, these structures being pushed apart and become distinct one animals began to live on land. For example, it can be surmised, based on the brain of a "living fossil" the shark, that 450 millions B.P., that the majority of the forebrain in cartilaginous animals, consisted of a dorsal and ventral amygdala-striatum, the striatoamygdaloid gray, such that the amygdala and striatum were enmeshed forming a dorsal lateral/ventral lobe, an organization pattern that is also evident in cyclostromes, fish, and (to a much lesser degree) in tailed amphibians (urodela). The dorsal/ventral amygdala/striatum formed a composite allocortical structure that was dominated by the olfactory system (Haberly, 2010; Smeets, 2010; Ulinksi, 2010).
The striatoamygdaloid gray (or "dorsal pallium") performed motor functions that in some respects mirrored those of the brainstem which was dominated by the vestibular system and to a lesser extent, the visual system; one of the major differences being that the olfactory-forebrain was provided with a crucial "pregnant interval" before it need respond, which enabled it to beocme a thinking machine whereas the brainstem remained reflexive.
As animals emerged from the sea and began to live on dry land, the amygdala and striatum were pushed apart due to the increased important of motor functioning and as the olactory bulb expanded as did its forebrain pathways, so as to adapt to living in a world of smell. With the evolution of amphibians (the tailless anura) the amygdala and the striatum (and much later the hippocampus and striatum) became semi-separate structures (Herrick, 1925; Nieuwenhuys & Meek, 2010ab; Stephan & Andy, 2017; Ulinksi, 2010) which nevertheless remained tightly linked, the striatum responding to amygdala impulses which were directed to the brainstem. As these structures expanded and were pushed further apart, the dorsal aspect became a rudimentary dorsal striatum, and the ventral aspect became the ventral (limbic) striatum.
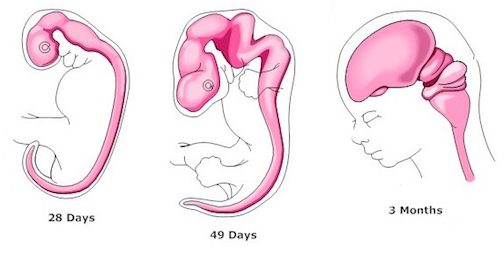
Ontogeny often replicates phylogeny and this amygdala-striatal separation is also repeated over the course of embrylogical development. For example, around the sixth week of fetal development immature neuroblasts migrate in massive numbers from the ventricular lining, and congregate in the more caudal portion of the emerging forebrain, thus forming an arc shaped "striatal ridge" from which the primordial amygdala will emerge (Gilles et al., 1983; Humphrey, 1968). Approximately one week after the formation of the amygdala, this primordial striatum begins to differentiate and balloon outward. Thus both the striatum and amygdala are derived from the arc shaped "striatal ridge," the caudal portion giving rise to the primordial amgydala at about the 6th week of gestation, and the basal portion later giving rise to the primordial striatum which initially overlies and is contiguous with the amygdala (Gilles et al., 1983; Humphrey, 1968). Over the ensuing weeks, these structures are pushed further apart thus again, replicating phylogeny.
Nevertheless, they do not completely separate, and the medial amygdala remains extensively interconnected with the limbic (ventral) striatum. As noted, the substantia innominata (ventral globus pallidus/nucleus basalis) merges with the centro-medial amygdala and receives extension projections from the lateral amygdala. In addition, the lateral amygdala id directly connected with the corpus striatum, via the so called "tail of the caudate." However, as these connnections are predominantly from the amygdala to the striatum and not vice versa, the caudate could be considered the bulbous "tail" end of the amygdala.
In fact the human corpus and limbic striatum are sandwiched between the posterior/ventral medial/lateral "tail" and the extended centro-medial "nose" (or internal shoulder) of this nucleus. And, these structures, being derivatives of the amygdala, respond to amygdala impulses, as they receive extensive amygdala projections (Gloor, 1955; Klingler & Gloor, 1960; Krettek & Price, 1978)--projections which are not reciprocated. The the right and left amygdala povides ipsilateral connections.
The amygdala (as well as the anterior cingulate, lateral hypothalamus, and hippocampus), therefore is able to exert considerable influence on the basal ganglia which appears to have evolved out of the amygdala in order to serve as an emotional-motor interface so that amygdala needs and impulses may be acted on in a flexible manner These are functions the basal ganglia (the limbic striatum in particular) continues to perform in humans as well as other creatures (e.g., MacLean, 2010; Mogenson 2011).
For example, the basal ganglia is exceedingly important in the stereotyped and species specific motoric expression of social and emotional states such as running away in fear, biting defensively, or via ballistic movements (hitting, kicking) or as manifested through facial expression, posture, muscle tone, or gesture (Mogenson & Yang, 2011; MacLean, 2010; Rapoport 2011). Because humans possess basically the same basal ganglia and limbic system, when happy, sad, angry, and so on, the facial and body musculature assumes the same readily identifiable emotional postures and expression regardless of culture or racial orgins (Ekman 1993; Eibl-Ebesfedlt, 2010; However, see Russell 1994).
Because of this similarity in basal ganglia functional architecture, regardless of culture or race (and in many respects, mammalian species) if frightened, angry, or in the process of being assaulted, animals or humans may similarly engage in (ballistic) hitting and kicking, or biting, and/or all of the above, depending on context, mood and situational variables (including play). However, in contrast to the brainstem and cerebellum which provides reflexive and stereotyped motor programs that can be performed without thinking, the basal ganglia is capable of considerable flexibility in regard to motor-emotional expression, and is exceedingly responsive to the organisms motivational and emotional state -via its extensive interconnections with the limbic system.
Thus, the striatum contains cells which selectively respond to motivationally significant stimuli, including novel and familiar variables that are rewarding or punishing (Rolls & Williams, 1987; Schneider & Lidsky, 1981). Striatal neurons can also react differently to familiar stimuli depending on their reinforcement properties, and many neurons will in fact increase their responsiveness as stimuli approach the mouth, and/or touch the face and mouth (Rolls & Williams, 1987; Schneider & Lidsky, 1981). Some striatal neurons also respond when making tongue and lip movements (e.g. licking) and during arm movements toward a food item (Rolls & Williams, 1987).
These findings suggest the striatum is involved in orienting and guiding movements toward the mouth presumably so that a desired object can be licked, sucked on, chewed, and consumed. In this regard the striatum could be considered a primary motor center which enables various limbic desires, needs, and impulses such as hunger, to be satisfied.
The limbic striatum (e.g. the nucleus accumbens) and the mesolimbic DA system which projects to these nuclei also appear to be highly involved in mediating feelings of pleasure including the rewarding effects of amphetamine, cocaine and opiates (see Ellison, 1994; Hakan et al. 1994; Koob et al. 2011). These effects (including the aversiveness of opiate withdrawal) are probably due not only to the presence of opiate, DA, and related receptors, but the rich interconnections maintained with the amygdala as well as the lateral hypothalamus (Kelsey & Arnold 1994; Olton et al. 2011; Zaborszky et al. 2011) which constitute part of the "pleasure circuit" maintained by the medial forebrain bundle (Olds & Forbes, 1981; see also chapter 13).
However, animals will also work in order to self-administer opiates directly into the accumbens (Koob et al. 2011; Olds & Forbes 1981). Conversely, lesions to the nucleus accumbens may disrupt the capacity to experience pleasure or the rewarding effects of opiates and cocaine (Koob et al. 2011); or to engage in complex coordinated defensive acts (see below).
Catatonia, Parkinson's Disease, & Psychosis.
Lesions to the corpus striatum and lenticular nucleus (putamen and globus pallidus) can attenuate one's capacity to motorically express their emotions via the musculature; e.g. the face may become frozen and mask-like. These latter motor disturbances are well known symptoms associated with Parkinson's disease, a disturbance directly linked to dopamine deficiency (Fahn, 2009) and neuronal degeneration not only in the putamen (Goto, et al. 2010; Kish, et al. 1988; see also Hauser et al., 2009), but within the limbic striatum, i.e. the nucleus accumbens (see Rolls & Williams, 1987), as well as in the supplementary motor areas and medial frontal lobe -which maintains rich interconnections with the striatum.
However, when chemical or structural lesions extend beyond the basal ganglia and come to include the medial frontal lobe, not only might an individual suffer motor rigidity, they may become catatonic and experience extreme difficulty responding to external or internally mediated impulses (Joseph, 2009a). Various aspects of this symptom complex also characterize those with Parkinson's disease (see below).
Other disturbances associated with striatal abnormalities include Huntington's chorea, ballismus, restless leg syndrome, sensory neglect and apathy, obsessive compulsive disorders, mania, depression, "schizophrenia" and related psychotic states (Aylward et al. 1994; Baxter et al. 1992; Caplan, et al. 2010; Castellanos et al. 1994; Chakos et al. 1994; Davis, 1958; Deicken et al. 2013; Ellison, 1994; Rauch et al. 1994; Richfield, et al. 1987; Turjanski, et al., 2009). Severe memory loss and social-emotional agnosia and an inability to recongize friends or loved ones is also characteristic of striatal abnormalities, particularly disturbances involving the limbic striatum.
Thus although the basal ganglia is often viewed and described as a major motor center (detailed below), the functional capacities and symptoms associated with this group of nuclei are quite diverse, and vary depending on the nuclei and chemical neurotransmitters involved as well as the laterality, location, and extent of any lesion.
THE AMYGDALA, STRIATUM, SMA, & LIFE THREATENING FEAR & AROUSAL
In situations involving exceedingly high levels of arousal coupled with extreme fear, the individual may simply freeze and attentional functioning may become so exceedingly narrow that little or nothing is perceived and cognitive activity may be almost completely (albeit temporarily) abolished (see chapter 30). These behaviors are apparently under the control of the amygdala which can trigger a "freezing" reaction and a complete arrest of ongoing behavior (Gloor, 1960; Kapp et al. 1992; Ursin & Kaada, 1960) via these brainstem/striatal interconnections. This is part of the amygdala attention response, which at lower levels of excitation may be followed by anxious glancing about, an increase in respiration and heart rate, pupil dilation, and perhaps cringing and cowering or flight (Gloor, 1960; Ursin & Kaada, 1960).
Among humans, the fear response is one of the most common manifestations of amygdaloid stimulation (Gloor, 2010; Halgren, 1992; Williams, 1956). However, if arousal levels continue to increase, subjects do not merely freeze in response to increased fear, they may become catatonic; a condition which may be secondary to dopamine and serotonin depletion and amygdaloid influences on the SMA as well as the striatum; nuclei which are intimately interconnected.
For example, in response to extreme fear, "one tendency is to remain motionless, which reaches its extreme form in death-feigning in certain animals and sometimes produces the waxy flexibility of catatonics" (Miller, 1951). The affected individual becomes psychologically and emotionally numb and unresponsive which is coupled with a complete blocking off of cognition. Moreover, the individual may resist and fail to respond to attempts at assistance (Krystal, 1988; Miller, 1951; Stern, 1951).
The airline industry has referred to this as "frozen panic states" (Krystal, 1988), a condition sometimes seen in air and sea disasters. For example, in mass disasters, 10-25% of the victims will become frozen, stunned, and immobile, and will fail to take any action to save their lives, such as attempting to evacuate a burning or sinking craft even though they have been uninjured (see Krystal, 1988).
According to Krystal (1988) with increasing fear "there is also a progressive loss of the ability to adjust, to take the initiative or defensive action, or act on one's own behalf... that starts with a virtual complete blocking of the ability to feel emotions and pain, and progresses to inhibition of other mental functions" (Krystal, 1988, p. 151).
Evolutionary Significance of Rigidity.
Catatonic panic states are prevalent in the animal kingdom, and constitute a life preserving reaction that is apparently mediated by the amygdala and striatum. That is, by freezing and not moving, predators may fail to take note of their presence.
Catatonia, coupled with emotional and "psychological" numbing, also represents a total surrender reaction, usually as a prelude (and hopeful guarantee) of a painless death when attacked by predators or invaders. That is, the prey may cease to run or fight and simply stand still or lie down and allow predators to literally eat them alive. Or, in the case of humans as sometimes occurs during war and genocidal mass murders, passively allow themselves to be marched into a ditch and shot.
According to Krystal (1988, p. 144), "thousands of European Jews obeyed orders in an automatonlike fashion, took off their clothes, and together with their children decended into a pit, lay down on top of the last layers of corpses, and waited to be machine-gunned," all the while seemingly almost petrified with fear and/or completely numb as to what was going on around them.
Presumably this numbing is made possible via the massive secretion of opiates within the amygdala and basal ganglia, whereas the rigidity and loss of the will to resist is a consequence of overwhelming fear and hyper-amygdala influences on the medial frontal lobe and corpus and limbic striatum.
Once prey have sighted a predator, some animals, however, instead of running in fear, will simply "freeze," fall to the ground, and lie stiff, rigid, and motionless as if dead. Unless exceedingly hungry, many predators will avoid eating creatures which appear to be already dead (i.e. unresponsive).
As sometimes occurs to potential victims during mass killings (e.g. the 1994 Rwanda civil war between the Hutus and the Tutus) humans too, will sometimes fall down as if dead and may remain frozen, stiff and and unmoving for long time periods even though they may not have been harmed (Krystal 1988). Indeed, sometimes these individuals are believed to be dead even by rescuers or those who are clearing away and burying bodies.
Presumably it is via connections with the basal ganglia and medial frontal lobes that the amygdala is able to induce these catatonic states, which in part is also dependent on dopamine. For example, it has been demonstrated that under extremely stressful conditions the striatal and frontal lobe DA system is adversely affected (see Le Moal & Simon 2011).
IMPLICATIONS REGARDING PARKINSON'S DISEASE
As noted, many of the functions originally associated with the basal ganglia have been subsumed by the neocortex, and damage to the frontal lobes can produce essentially similar symptoms as seen following caudate destruction; including stiffness, rigidity, and difficulty initiating movement (see chapter 19).
Given that the medial frontal lobes, corpus and limbic striatum, and amygdala are extensively interconnected, and given the powerful influences of the limbic system on all aspects of behavior, it thus appears that when exceedingly aroused or emotionally stressed, the amygdala is able to inhibit (or overactivate) the frontal-striatal motor centers which (in addition to the amygdala) are simultaneously undergoing DA depletion (which in turn results in hyperactivation of these nuclei including the amygdala; see Le Moal & Simon 2011). When this occurs, the organism may fall and cease to move, blink or even breathe (or breathe only shallowly and slowly). The creature therefore appears to be in a state of rigor mortis and thus dead (i.e. catatonic).
Overall, these amygdala-basal ganglia-medial frontal lobe-fear induced frozen and catatonic states are exceedingly adaptive; that is, unless the hapless victim is in a burning airplane or sinking ship.
Although not as dramatic or severe, similar states occur with biochemical abnormalities involving the dopamine pathways from the substantia nigra which not only feed the caudate, but the medial frontal lobes and the amygdala. Specifically, loss of DA (or excessive amygdaloid arousal) results in motor neuron hyperactivity and tonic EMG activity and thus limb and facial rigidity; conditions which also afflict those with Parkinson's disease.
In that some of these same exact "semi-frozen" and akinetic states are present in many of those with Parkinson's disease, and given that affected individuals are sometimes described as excessively aroused and/or unable to relax, and to suffer from heightened autonomic nervous system activity (Stacy & Jankovic, 1992) this raises the possibility that the amygdala and related limbic nuclei may significantly contribute to the development of this disorder. Indeed, as noted, destruction of the amygdala prior to chemically lesioning the corpus striatum prevents the development of Parkinsonian symptoms.
PARKINSON'S DISEASE
The basal ganglia plays a major role in controlling and facilitating specific movements, as well as inhibiting unwanted movements (Marsden & Obesco 1994; Turjanski, et al., 2009). The basal ganglia is also directly implicated in the generation of a variety of movement disorders, including chorea, ballismus, restless leg syndrome, rigidity, stiffness, and Parkinson's disease.
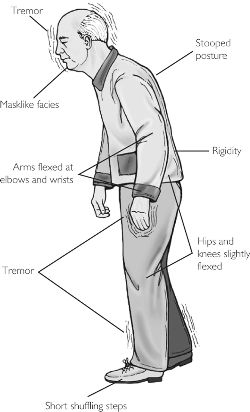
Parkinson's disease is a progressive degenerative disorder characterized by rigidity and shuffling gait, stooped posture, generalized slowness and stiffness of movement, and a loss of facial emotional expression, as well as a loss of spontaneity and flexibility in making postural adjustments when eating, going to the toilet, or having sex. Moreover, many Parkinson's patients experience not only rigidity and akinesia, but suffer from episodic freezing of movement (Dietz, et al. 2010; Pascual-Leone, et al. 1994), a tendency to easily fall (Stacy & Jankovic 1992) an impairment of "righting reflexes" (Calne 1994), and a reduced capacity to blink (Freedman 1992) and even to breathe (Stacy & Jankovic, 1992); similar to the frozen panic states and induced catatonia.
Hypophonia (reduced voice volume) dysarthria and a tendency to speak in a monotone, micrographia (small handwriting), and a 4-8 c/sec. ("pill rolling") resting tremor (exacerbated by stress) involving antagonistic muscles are common (Freedman, 1992; Stacy & Jankovic, 1992). Depression, changes in personality and slowed thought processes are also not unusual among Parkinson's patients (Walters, 2009). Parkinsonism can result following neurological trauma or vascular abnormalities (Koller, 1987; Murrow et al. 2010) or isolated lesions to the substantia nigra (Stern, 1966) and can therefore arise from a number of different causes including infection and toxic exposure. Parkinson's disease may also overlap with other striatal disturbances such as Alzheimers disease (Calne 1994). Indeed, sigificant cognitive deficits are not unusual among those with Parkinson's disease (Freedman, 1992; Rajput 1992); i.e. subcortical dementia.
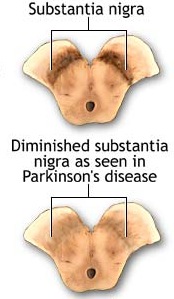
Parkinson's disease usually begins after age 60 (though the first signs may appear during the early 40's), effects about 2% of the population (Koller, 1987) and is usually characterized by a massive loss of up to 80-85% of the dopamine neurons in the substantia nigra and an 80% decrease in striatal DA, with DA depletion greatest in the putamen (Goto, et al. 2010; Kish, et al. 1988). In fact, transplanting fetal nigral tissue into the putamen, can result in increased dlurodopa intake and some long-term clinical improvement (Hauser et al., 2009).
Hence, whereas the substantia nigra DA system is directly implicated, the mesolimbic pathways and limbic striatum are only a mild factor and are only mildly effected in this disorder.
STRIATAL IMBALANCE & PARKINSON'S DISEASE
The differential involvement of the nigrostriatal vs mesolimbic DA system raises the possibility that whereas the corpus striatum and medial frontal lobes are negatively impacted, the amygdala and limbic striatum may continue to function normally -due to preservation of the mesolimbic DA system. However, because the normal balance between these various nuclei is disrupted, amygdala influences received within the SMA and corpus striatum may in fact overwhelm and massively inhibit (or over activate) these nuclei thereby giving rise to Parkinsonian symptoms ; i.e. rigidity, a tendency to fall coupled with reduced blinking and disturbed righting reflexes, etc.
In addition to DA in the genesis of Parkinsonian symptoms, significant reductions in opiate receptors within the putamen as well as the globus pallidus have also been reported (Goto et al. 2010), such that neurons involved in the experience of "reward" are effected. This selective loss of "reward" neurons suggests a a reduction in the capacity of the basal ganglia to receive pleasurable or positive emotional input as might be provided not only by opiates, but via the lateral amygdala and lateral hypothalamus.
Possibly, because striatal neurons associated with negative feelings states may be selectively preserved in Parkinson's disease, the basal ganglia (but not the amygdala, hypothalamus, or neocortex) may respond as if in a highly aroused negative state (e.g. fearful, stressed) even when that is not the case. Adding to this imbalance would be the relative preservation of the mesolimbic DA system, and the continued input from the medial amygdala into the corpus and limbic striatum -the medial amygdala being more involved in the generation of unpleasant including fearful mood states (chapter 13)
Indeed, loss of corpus striatal DA and/or excessive amygdaloid arousal is directly associated with the development of tonic EMG activity, motor neuron hyperactivity and thus excessive tonic excitation of the musculature and limb rigidity; i.e. Parkinson's symptoms. Conversely, excess striatal DA can result in chorea and excessive movement as well as psychosis.
THE SMA & PARKINSON'S DISEASE
It has been reported that many patients with Parkinson's disease display reductions in norepinephrine (NE) as well as DA throughout the upper layers of the motor, premotor, and medial frontal lobes (Gasper et al. 2011; Hornykieciz, 1982). NE (and 5HT) depletion also occurs following heightened emotional states, including extreme fear, prolonged stress and heightened arousal, and with amygdala hyperactivation. As noted, individuals with Parkinson's disease are also sometimes described as excessively aroused and/or unable to relax, and to suffer from heightened autonomic nervous system activity and gastrointestinal disturbances (Stacy & Jankovic, 1992) --disturbances which also implicate the amygdala and excessive limbic system arousal. Conversely, the autonomic nervous sytem of those who suffer from Alzheimer's disease have been characterized has hypoactive (Borson et al. 1992); the limbic striatum being implicated in Alzheimer's disease.
The SMA typically becomes functionally activated prior to movement (chapter 19) and well before the caudate or putamen with which it is interconnected. When Parkinson's patients attempt to make repetitive movements, SMA functional activity is abnormally reduced as is putamen activity (Playford et al. 1992). This suggests that in addition to the corpus striatum one of the major regions of the neuroaxis involved in the "will" to move and the capacity to prepare to move; i.e. the SMA, is dysfunctional in those suffering from Parkinson's disease. Thus the SMA and medial frontal lobes as well as the corpus striatum, DA system, and amygdala, are implicated in the genesis of this disorder (e.g. Playford et al. 1992).
Consider, for example, that Parkinson's patient's have difficulty using both hands to perform two different movements (e.g. squeeze a ball with one hand, draw a triangle with the other). Rather, they tend to perform one action, then switch to the other (Caligiuri et al. 1992). SMA dysfunction can result in similar disturbances (see chapter 19). Similarly, the ability of Parkinson's patients to engage in sequential and repetitive movements (Playford et al. 1992), or to simultaneously engage in separate movements such as walking, turning, and talking (Stacy & Jankovic 1992) is severely effected; a disorder which is also associated with frontal lobe abnormalities.
THE HIPPOCAMPUS & SMA
The striatum is also innervated by the hippocampus which is involved not only in memory, but spatial and cognitive mapping of the environment (chapters 13, 14). Thus the corpus striatum presumably relies on hippocampal (as well as parietal lobe) input in order to coordinate movement in visual-space.
It is noteworthy that Parkinson's patients find it easier to perform movements that are cued or guided by external visual stimuli, whereas those which are internally generated are more severely effected (Dietz, et al. 2010). Hence, perhaps those striatal neurons which normally receive and integrate hippocampal input may also be dysfunctional.
However, the role of the hippocampus appears to be minimal in the development of Parkinson's disease, at least in those cases without memory loss or dementia. Instead, the beneficial effect of external cues again raises the specter of significant medial frontal lobe and SMA involvement.
For example, lesions to the medial frontal lobes can result in an inability to internally generate movements whereas external environmental and visual cues can exert an almost "magnetic" effect on limb activation and patients may involuntarily respond and react to external stimuli by reaching, grasping, or utilizing whatever objects may be near (see chapter 19). In contrast, the lateral frontal lobes are involved in externally driven movements (chapter 19).
Hence, Parkinson's patients are possibly better able to "move" in response to visual stimuli presumably because of preserved lateral frontal lobe functioning and the loss of inhibitory SMA control -which (in part) accounts for their difficulty internally "willing" purposeful and fluid movements.
DA, ACh, & STRIATAL IMBALANCE.
The DA and cholinergic, ACh system appear to exert counterbalancing influences. For example, loss of nigrostriatal DA results in increased ACh, neuron hyperactivity (see Aghanjanian & Bunney, 2017; Bloom et al. 1965) and tonic EMG activity and thus limb rigidity which can be reversed by anti-cholinergic drugs (Klockgether, et al., 1987). That is, drugs which decrease ACh and those which increase DA, can ameliorate Parkinsons symptoms (Fahn, 2009), which suggests these two transmitters play oppositional and balancing roles (see Stoof et al. 1992). Moreover, ACh can be inceased by drugs that decrease DA, and can be decreased by drugs which increase DA (reviewed by Stoof et al. 1992).
DA normally inhibits ACh release and increases GABA activity. The DA system appears to exert an controlling influences not only on GABA but ACh neurons in the striatum (Le Moal & Simon 2011). Hence, reductions in DA result in decreased GABA activity and increased limbic striatal arousal and ACh activity, and reduced corpus striatal arousal and reduced GABA activity (see Stoof et al. 1992). Under these latter conditions the subject becomes rigid.
However, under conditions where striatal DA is depleted, the excessive release of ACh creates excessive neural activity (Aghanjanian & Bunney, 2017; Bloom, et al. 1965) which in turn may be manifested in the form of excessive motor actions; i.e. ballismus, chorea, if the medial and ventral globus pallidus-subthalamic circuit is effected, or hypomovement and Parkinson's symptoms if the corpus striatum and dorsal globus pallidus is effected.
For example, fluctuations in DA levels can either act to excite or inhibit motor and related cognitive-memory activity within the limbic striatum (Le Moal & Simon 2011; Mogenson & Yang 2011). However, with high levels of limbic striatal DA (and/or amygdala) activity, coupled with reduced GABA, animals may appear hyperactive and engage in running, "galloping," and biting, and related movements.
Presumably these latter actions are due to inhibitory release (a consequence of striatal dysfunction or hyperactivation), and thus the triggering of brainstem motor programs that subserve these specific behavioral acts. These same stimulus (predator) released actions, across evolution, and across most animal species, are directly related to the functional integrity of the limbic system and striatum, and the capacity to survive when living in close proximity to predators, the elements, and cospecies. Actions without thought are in fact the province not of the basal ganglia, but the brainstem, which is why these and related motor programs are stored within brainstem nuclei (see chapter 17).
The Amygdala, Temporal Lobe & DA.
Psychotic disturbances, including "schizophrenia" need not involve the basal ganglia or DA dysfunction, as disorders of thought and mood may also appear following frontal or temporal lobe damage. However, with frontal lobe damage the basal ganglia and SMA are often effected, whereas the amygdala is implicated in temporal lobe related psychotic states.
Indeed, abnormal DA as well as norepinephrine (NE) and serotonin (5HT) have been reported in the amygdala of those diagnosed as psychotic (Spoont 1993; Stevens 1992). In addition, a gross asymmetry in the post-mortem levels of mesolimbic DA has been noted in the brains of those diagnosed as schizophrenic (reviewed in Le Moal & Simon 2011). Specifically, an increase in DA has been noted in the central amygdala and in the striatum of the left hemisphere, whereas DA levels within the right cerebral are similar to normals.
In fact, the left amygdala and left temporal lobe (Flor-Henry, 1969; Perez, et al. 1985; Stevens, 1992) have long been thought to be a major component in the pathophysiology of psychosis and schizophrenia (Heath, 1954; Stevens, 2015; Torey & Peterson, 1974). For example, abnormal activity as well as size decrements have been noted in the left amygdala (Flor-Henry, 1969; Perez, et al. 1985) as well as the left inferior temporal lobe in schizophrenic patients (see Stevens, 1992). Spiking has also been observed in the amygdala of psychotic individuals who are experiencing emotional and psychological stress (see Halgren, 1992).
In this regard it has been suggested that stress, particularly when experienced prenatally may be responsible, in some cases, for the development of DA abnormalities (see Le Moal & Simon 2011), whereas adverse early environmental experiences and trauma may contribute to the development of kindling, spiking and abnormal amygdala functioning (see chapters 28, 30). As noted, Parkinson's symptoms in some respects also resemble the effects of chronic stress and an inability to relax.
THE LIMBIC STRIATUM, MEMORY & ALZHEIMER'S DISEASE
THE LIMBIC STRIATUM, DA, 5HT, ACH, NE, & MEMORY
The limbic striatum consists of the nucleus accumbens, olfactory tubericle, the extended (centro-medial) amygdala, as well as the ventral aspects of the caudate, putamen and globus pallidus (-substantia innomminata).
The nucleus accumbens is located immediately beneath the anterior portion of the caudate and from a microscopic level appears to be part of the ventral caudate (Olton et al. 2011; Zaborszky et al. 2011). However, it maintains extensive interconnections with the amygdala as well as the hippocampus via the fimbria-fornix fiber bundle (DeFrance et al. 1985), which implicates this nuclei in memory functioning and probably the learning of visual-spatial and perhaps social-affective relationships.
Similarly, the dorsal portion of the substantia innominata ("great unknown") merges (dorsally) with the ventral globus pallidus (and ventrally with the centro-medial amygdala), and maintains interconnections with the accumbens, hippocampus, lateral amygdala, and dorsal medial (DM) nucleus of the thalamus (Young et al. 2014; Zaborszky et al. 2011) -the DM being involved in regulating neocortical arousal and information reception as well as memory (see chapter 19).
Like the accumbens, the substantia innominata (SI) is also important in memory functioning and has been implicated as one of the principle sites (along with the nucleus basallis) for the initial development of Alzheimers disease (see Olton et al. 2011; Zaborszky et al. 2011, for review of related details).
The nuclei of the limbic striatum are also interconnected with the lateral and medial hypothalamus, brainstem reticular formation, and the frontal and inferior temporal lobes (Everitt & Robbins, 1992; Groenewegen, et al. 2011; Heimer & Alheid, 2011; Kelly, et al. 1982; Mogenson & Yang 2011; Olton et al. 2011; Parent & Hazrati 2013; Van Hoesen, et al. 1981; Zaborszky et al. 2011). The limbic striatum is able, therefore, to exert widespread effects on neocortical and subcortical structures.
As noted, the limbic striatum receives DA from the mesolimbic system as does the amygdala. The amygdala also sends axons that terminate in striatal neurons immediately adjacent to those innervated by mesolimbic DA neurons (Kelly et al. 1982; Yim & Mogenson, 1983, 1989). Thus, a complex interactional loop is formed between these nuclei, the integrity of which, in part is dependent on the mesolimbic DA system which can act on the amygdala, hippocampus and limbic striatum simultaneously so as to modulate striatal reception of amygdala (Maslowski-Cobuzzi & Napier, 1994) and hippocampal excitatory signals, and regulate the transmission of accumbens input into the SI. Alterations in mesolimbic DA activity, therefore, can significantly influence limbic striatal, amygdala and hippocampal activity as well as the motoric expression of limbic impulses.
For example, depletions in mesolimbic DA can disrupt motor and social-emotional memory functioning; a condition compounded by DA influences on acetylcholine (ACh) neurons, and the effects of striatal DA on the reception of hippocampal, amygala, and neocortical input.
Indeed, the limbic striatum (and limbic system) contain high densities of ACh neurons which are involved in memory as well as motor functioning (Olton et al. 2011; McGaugh et al. 1992; Zaborszky et al. 2011). In fact, of all striatal nuclei, densities of DA (and ACh) neurons are highest within the (SI) and nucleus accumbens (Meredith et al. 1989) which in turn greatly influences the SI (which is a major source of neocortical ACh), as does the amygdala (Yim & Mogenson 1983) and hippocampus; i.e. these signals converge on the SI.
Like the striatum, the hippocampus (and the amygdala) also receives meso-limbic DA input and contains D1 and D2 receptors (Camps et al. 2010) as well as ACh. ACh influences on neuronal activity can be excitory or inhibitory depending on the resting membrain potential of the receiving neuron (Ajima et al. 2010) as well as concurrent DA activity. Specifically, mesolimbic DA mediated signals are relayed from the accumbens to ACh neurons in the SI which in turn project to DM and the neocortex, the frontal lobes in particular (Mogenson & Yang 2011; Olton et al. 2011; Zaborszky et al. 2011). However, the DA and cholinergic, ACh system appear to exert counterbalancing influences.
For example, ACh is usually inhibited by DA. By contrast, DA depletion results increased ACh and neuron hyperactivity (see Aghanjanian & Bunney, 2017; Bloom et al. 1965; Klockgether, et al., 1987) which can greatly disrupt cognitive and memory functioning as well as motor activities, unless reversed by anti-cholinergic drugs. In fact, damage to this DA/cholinergic system can result in Alzheimers disease (Olton et al. 2011; Zaborszky et al. 2011).
As per memory functioning, it appears that mesolimbic DA modulates the reception of hippocampal (motor-spatial) input into the accumbens which projects to the SI (which in turn distributes these influences, perhaps via ACh) to the neocortex). DA accomplishes this via inhibitory influences on ACh and facilitation of the GABA system which appears to exert inhibitory influences within the striatum and the transmission of impulses from the nucleus accumbens (and amygdala/hippocampus) to the SI. Therefore, if the inhibitory influences of GABA and ACh on the SI are dampened, the SI becomes activated and memory functioning may be enhanced or disrupted at the neocortical level as this nucleus exerts widespread ACh influences on the cerebrum. Thus fluctuations in DA levels can either act to excite or inhibit cognitive-memory activity within the limbic striatum (see Mogenson & Yang 2011) as well as the corpus striatum (Packard & White 2011) and the neocortex.
However, also of importance in this memory-striatal neural network is (5HT) serotonin (McLoughlin et al. 1994; Mogenson & Yang 2011) and (NE) norepinephrine (Roozendaal & Cools, 1994). For example, NE levels have been shown to fluctuate within the amygdala and nucleus accumbens when presented with novel stimuli (Cools et al. 2011) and during information acquisition. Presumably NE levels within the accumbens can regulate or influence the reception of amygdala and hippocampal impulses within the accumbens and SI which in turn projects to the neocortex and, in this regard, may act to shunt amygdala-hippocampal impulses to discrete or wide areas of the cerebrum.
Specifically, high alpha-NE activity is associated with reduced amygdala input, whereas low beta NE may reduce hippocampal input into the striatum. Conditions such as these, however, are most likely to result when stressed or traumatized in which case it is possible for amygdala (and thus emotional) input to be received in, learned, and expressed by the striatum in the absence of hippocampal participation (see Rozzendaal & Cools 1994). It is conditions such as these that can give rise to amnesia with preserved learning (see chapters 14, 30).
However, in some cases, it is also possible for both the alpha and beta NE system to be effected simultaneously such that amygdala and hippocampal input to the limbic striatum are inhibited in which case profound memory loss may result (see Rozzendaal & Cools 1994). Similar disturbances are associated with the serotonin (5HT) system (McLoughlin et al. 1994).
Depletion of 5HT can significantly effect the capacity to inhibit irrelevant sensory input (at the level of the brainstem, amygdala, basal ganglia, and neocortex). Hence, 5HT abnormalities or depletion typically results in confusion and sensory overload and in some instances the production of hallucinations (see chapter 30).
Thus disruption of the mesolimbic DA system and/or severe disruptions in the 5HT system in turn interferes with the hippocampal-limbic striatal memory system (Packard & White 2011) and can create neuronal hyperactivity -a condition which interferes with perceptual and hippocampal- memory-SI-amygdala functioning. In this regard it is noteworthy that the central 5HT system is severely disrupted among those with severe memory loss and Alzheimer's disease (McLoughlin et al. 1994) and that calcification of the globus pallidus/SI can induce visual and auditory hallucinations as well as cognitive deterioration (Lauterbach et al. 1994).
OVERVIEW: ALZHEIMER'S DISEASE
Alzhiemer's disease is associated with a profound loss of memory and cognitive and intellectual functioning, including, at its later stages, an inability to recognize friends, loved one's or their own personal identity. Alzhiemer's disease is estimated to afflict approximately 10% of those over age 65, and 50% of those over 85.
In its later stages, Alzhiemer's disease is associated with profound loss of cerebral functional capacity, coupled with neural degeneration and a loss of neurons and the development of amyloid (senile) plaque and neurofibrillary tangles. Because structures such as the substantia innominata, amygdala, and entorhinal cortex have been injured (Morrison et al. 2010; Gomez-Isla, et al., 2000; Rapoport 2010) it is thought that the destruction of this tissue and adjacent tissue accounts for the loss of memory, and social-emotional, facial recognition, and related visual abnormalities, including visual agnosia (Giannakoulos, et al., 2009).
However, because there is no single factor that has been implicated, and due to the number of associated disturbances, it has been argued that Alzheimer's disease and associated cognitive and memory disturbances are due to a "global cortico-cortical disconnection" syndrome (see Morrison et al. 2010; Rapoport 2010). Nevertheless, for the purposes of this chapter, the striatal contribution to this disorder will be emphasized.
MEMORY & ALZHEIMER'S DISEASE
As detailed in chapter 15, the amygdala and hippocampus provide massive input to the limbic striatum as well as the dorsal medial nucleus of the thalamus (DM) , the frontal lobes and reticular activating system, as does the accumbens and SI (see Heimer & Alheid, 2011; Koob et al. 2011; Mogenson & Yang 2011; Zaborszky et al. 2011). As noted, these nuclei, including the overlying entorhinal cortex, play a significant role in memory and the gating of information destined for the neocortex and appear to be part of a massive neural network designed to control information processing and to establish memory related neural networks (see also chapter 14). Presumably the nucleus accumbens apparently acts to integrate hippocampal and amygdala input, which is then transmitted to the SI which is also the recipient of limbic impulses concerned with cognitive, memory, and motoric activities (see Mogenson & Yang 2011).
In part, it appears that the accumbens and SI play an inhibitory (filtering) role on information processing, and may exert inhibitory (and counterbalancing) influences on the the medial frontal lobes, the DM, and corpus striatum (see Mogenson & Yang 2011). The amygdala exerts similar influences on these nuclei (chapter 15) and is also able to inhibit the SI and accumbens.
However, the role of the limbic striatum in cognitive and memory related activity also includes the learning of reward-related and aversion processes, the facilitation of approach and withdrawal responses and the memorization of where a reward or aversive stimulus was previously received (Everitt et al. 2011; Kelsey & Arnold 1994). In this regard the nucleus accumbens and SI are dependent on the medial and lateral amgydala, especially in the learning of negative experiences (Kelsey & Arnold 1994).
As noted, the SI is the primary source of neocortical cholinergic innervation (Carpenter 2011) and appears to be concerned with integrating social-emotional and cognitive input with motor memories and then storing them perhaps within the SI as well as within the neocortex. Hence, if the SI is lesioned the cholinergic projection system is disrupted, and cognitive as well as social-emotional memory functioning is negatively impacted.
Since the SI merges with and becomes coextensive with the centromedial amygdala (Heimer & Alheid 2011),and is an amygdala derivative, not suprisingly, a significant loss of neurons, neurofibrillary changes and senile plaques have been found in the amygdala (and hippocampus, Rapoport 2010), as well as in the SI, in patients with Alzheimer's disease and those suffering from degenerative disorders and memory loss (Herzog & Kemper, 1980; Mann, 1992; Sarter & Markowitsch, 1985). Degeneration in the amygdala would also account for the social-emotional agnosia and prosopagnosia that is common in the advanced states of Alzhiemer's disease; i.e. failure to recognize or remember loved ones (chapter 13).
WIDESPREAD NEURONAL DEATH & ALZHEIMER'S
Neuronal loss has also been reported in the entorhinal and parahippocampal areas of the inferior temporal lobe (see Morrison et al. 2010; Rapoport 2010) within which is buried the hippocampus and amygdala. Specifically, in addition to senile plaques, neurofibrillary tangles, a 40% to 60% loss of neurons during the early stages of this disease have been found in layers 4 and 2 of the entorhinal cortex (Gomez-Isla, et al., 2000)--the gateway to the hippocampus. Destruction of this tissue and adjacent tissue would also result in memory, social-emotional, facial recognition, and related visual abnormalities, including visual agnosia, as recently demonstrated among those with Alzheimer's disease (Giannakoulos, et al., 2009).
Indeed, among those with Alzheimer's disease, widespread atrophy, amyloid plaques, tangles, and metabolic disturbances have been noted in a variety of cortical areas with relative preservation of the motor and primary receiving areas (Rapoport 2010). Based on these findings it has been argued that Alzheimer's disease and associated cognitive and memory disturbances are due to a "global cortico-cortical disconnection" syndrome (see Morrison et al. 2010; Rapoport 2010); i.e., a loss of neurons in and interconnections with association neocortex, which in part would account for the cognitive deterioration.
The initiation of this deteriorative process may well be in the SI, and it may be due to an olfactory borne infection due to viral or bacterial invasion. That is, these bacterial or viral agents may enter the olfactory system and invade those structures directly innervated by the olfactory nerves, i.e. the amygdala, entorhinal cortex, and SI (Joseph, 1998d). On the other hand, or perhaps related to this scenario, are findings suggesting a genetic foundation for this disorder, such that defects in at least four specific genes (located on chromosomes 1, 14, and 21) may be responsible (Levy-Lehad et al., 2013; Saunders et al., 1993).
Given the likelihood that the limbic striatum and olfactory-limbic structures may be selectively involved in the early stages of Alzheimers, and given the fact that the SI provides cholinergic input to the neocortex, then the subsequent and progressive loss of SI (and amygdala) neurons might result in a progressive deterioration and cell death within the neocortex such that otherwise healthy neurons are killed. That is, since the SI contains high concentrations of cholinergic neurons which in turn project to widespread areas throughout the neocortex (reviewed in Carpenter 2011), perhaps the initial cell loss within the SI (also referred to as the nucleus basalis), may trigger further cell death in healthy neurons (which project to or receive fibers from the affected cells) which essentially become "pruned" and drop out form disuse.
Defective Axonal Transport.
There is evidence which indicates that perhaps due to head injury, drug or toxic exposure, or perhaps the loss of synaptic junctions (due to the death of unhealthy cells and dendritic retraction), axonal transport becomes defective due to the death of its target neuron. However, if a healthy cell cannot discharge and exchange information it too may die, thus leading to a domino effect and thus widespread cell death (see Burke et al. 1992). That is, due to the loss of terminal synaptic junctions (due to cell death) or to other chemical abnormalties including defects in microtubule assembly (which participates in neuronal transmission), axonal transport becomes dysfunctional as the receiving cell and it's dendrite have died. Hence, there is a buildup of toxic oxidatative metabolites and naturally occurring neurotoxins in the healthy cell body that project to the dead cell, which causes the normal cell to die as well. This would result in a progressive loss of neurons such that widespread areas of the cerebrum soon become effected.
Presumably this what may occur if the limbic striatum becomes abnormal and cells within the SI and accumbens begin to die. As the disturbance and deterioration spreads, cognitive, emotional, memory and related abnormalities including Parkinsonian symptoms begin to appear and become progressively worse as neocortical, striatal, and limbic neurons die and drop out.
LIMBIC & CORPUS STRIATAL COUNTERBALANCING INFLUENCES
The limbic and corpus striatum are richly interconnected and send projections to many of the same brain areas. However, due to differential input from the amygdala and DA systems, these nuclei exert tremendous counterbalancing influences on each other and their associated neural networks. For example, the centro-medial as well as the basolateral amygdala projects to and exerts excitatory influences on the the limbic striatum (Maslowski-Cobuzzi & Napier, 1994; Yim & Mogenson 1982), whereas the medial and posterior-lateral amygdala projects to the corpus striatum and the ventral and dorsal globus pallidus. Via these dual interconnections, the amygdala can exert simultaneous and even oppositional influences on these nuclei.
In addition, the corpus striatum receives the bulk of its DA from the nigrostriatal systems, whereas the limbic striatum receives DA from the mesolimbic DA system (see Fibiger & Phillips, 1986 for review) -transmitter systems which interact at the level of the brainstem, limbic system, striatum, and neocortex (Maslowski-Cobuzzi & Napier, 1994). For example, it has been shown mesolimbic DA can act on the amygdala and limbic striatum simultaneously so as to modulate striatal reception of amygdala excitatory signals (Maslowski-Cobuzzi & Napier, 1994).
However, because the corpus and limbic striatum are largely (but not completely) innervated by different clusters of midbrain DA neurons, reductions or increases in one DA system can exert profound influences on those neurons innervated by the others -for example, by eliminating inhibitory or counterbalancing influences. Thus depletion of DA in the nigrostriatal (but not the mesolimbic) pathways can result in increased activity within the limbic striatum (see Yim & Mogenson, 1983, 1989) and medial amygdala, but deceased activity within the corpus striatum. If this occurs, movement programming may be disrupted resulting in rigidity or tremors -a consequence, in part, of an imbalance in amygdala-DA-striatal activation.
Conversely, mesolimbic DA depletion can result in enhanced corpus striatal activity and decreased limbic striatal and lateral amygdala and hippocampal activity, such that motor and social-emotional memory functioning may be disrupted; a condition compounded by DA influences on acetylcholine (ACh) neurons and the reception of hippocampal, amygala, and neocortical input within the striatum. Hence, the functioning of the limbic or corpus striatum can be severely disrupted even when the functional integrity of its own neurotransmitter systems are otherwise intact; i.e. due to a loss of counterbalancing influences.
CONCLUDING STATEMENT: THE AMYGDALA
The basal ganglia motor circuit, as conceptualized and described in this chapter, consists of multiple neural networks involving the caudate, putamen, globus pallidus, limbic striatum, motor thalamus, subthalamus, brainstem, SMA, pre-motor and primary motor areas, and the amygdala.
Unfortunately, the role of the amygdala in movement is often ignored by investigators (and almost all textbooks) although all aspects of the motor circuit are directly or indirectly innervated by this nuclei and respond to amygdala mediated impulses. Although the amygdala can be surgically destroyed bilaterally without significantly effecting normal motor functioning, in many instances the amygdala is implicated in the genesis of abnormal motor activities and may play a significant role in striatal dysfunction and the development of Parkinson's and related diseases.
REFERENCES












