Rhawn Gabriel Joseph, Ph.D.
BrainMind.com
Key Words: Consciousness, split brain functioning, mental duality, dreams, unconscious mind
1. THE MULTIPLICITY OF MIND
The mind is a multiplicity linked to the functional integrity of specific brain areas (Joseph 2010), including the brainstem, limbic system, and right hemisphere. Strictly speaking, the brainstem mediates and provides the neuroanatomical foundations for the most rudimentary, reflexive, and unconscious aspects of mind (Joseph 1996). The limbic system, which includes the amygdala, hypothalamus, hippocampus, septal nuclei and cingulate gyrus, is more clearly linked to what Freud and Jung likened to the unconscious, subserving basic emotional and motivational impulses, the desire for sex and love, and the most sublime of emotions, including spirituality and religious rapture (Joseph 1990c, 1992b, 1994, 1998, 2001).
The left cerebral hemisphere is classically associated with expressive speech and receptive language, and the temporal-sequential, mathematical, analytical, and linguistic aspects of consciousness. However, it has now been well established that the right cerebral hemisphere is dominant over the left in regard to the perception, expression and mediation of almost all aspects of social and emotional functioning, including the recall of emotional memories (Joseph 1982, 1986a,b, 1988a,b, 1990, 1992, 1996, 2003). This includes the comprehension and expression of emotional sounds, such as the melody of the voice, prosody, intonation, as well as singing, cursing and praying, and the perception of environmental sounds. In fact, as language consists of not just words but melody and prosody, the two hemispheres interact, via the corpus callosum, to produce and comprehend all aspects of human speech.




Further, the right hemisphere is dominant for most aspects of visual-spatial perceptual functioning, the recognition of faces including friend, loved ones, and one's own face in the mirror. Faces, of course, convey emotion whereas visual-space is related to the environment and the movement and localization of the body in space. Thus, recognition of one's own body and the maintenance of the personal body image is also the dominant realm of the right half of the brain (Joseph 1982, 1986a, 1988a,b, 1990a, 1992, 1996). The body image, for many, is tied to personal identity; and the same is true of faces.
Visual-spatial, facial recognition, body image dominance, the comprehension and expression of emotional sounds, all appear to be tied to a greater representation of these functions in the right vs the left hemisphere. Further, the right hemisphere and limbic structures within the right temporal lobe, provide the neurological foundation for the generation of dreams (Joseph 1988a, 1992b, 2001, 2011a). The right hemisphere, in fact, appears to maintain a realm of conscious-awareness which is completely different from that of the left (Bogen 1969; Sperry 1966, 1968, 1974, 1982). Right hemisphere mental functioning is more visual, spatial, emotional, personal, and non-verbal. By contrast, the mental realm of the left is more tied to language, including thinking in words (Joseph 1982, 1990b, 1996, 2011b)
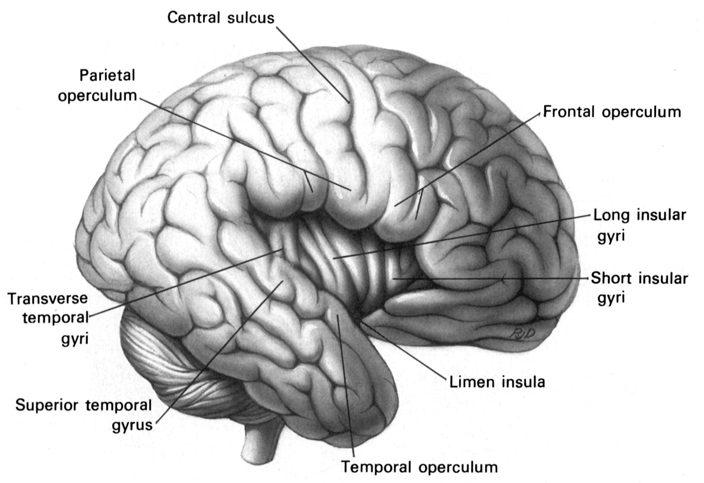
In part, it is believed that the right hemisphere dominance over social and emotional functioning is due to more extensive interconnections with the limbic system (Joseph, 1982, 1988a), including the fact that limbic system appears to be functionally and structurally lateralized (Joseph 1990c, 1992b, 2001). This right hemisphere limbic dominance came to include the expression and representation of limbic language, thus providing the right cerebrum with a functional dominance in regard to the expression and comprehension of emotional speech as well as all aspects of emotional perceptions, the image of the body, and the recognition of friends and loved ones.


By contrast, with the evolution of language, tool use, and right hand motor control, functions formally associated with the left half of the brain were crowded out, due to competition for neural space, and became the exclusive and enhanced domain of the right (Joseph 1990a,b, 1992a, 1993, 1996, 2000a).
Over the course of evolution, the limbic system and each half of the brain have developed their own unique strategies for perceiving, processing, and expressing information, as well as specialized neuroanatomical interconnections that assist in mediating these functions. Thus, whereas the limbic system mediates the more unconscious aspects of social-emotional and personal awareness, the neocortex and the cerebral hemispheres are organized such that two potentially independent mental systems coexist, literally side by side.
2. THE NEUROANATOMY OF MIND
The right and left half of the brain are functionally specialized. Each half of the brain is divided into a frontal, temporal, occipital, and parietal lobe and is covered with a 6-layered neocortex. Each neocortical layer is also functionally specialized and change in thickness or cellular composition depending on location (e.g. the frontal, temporal lobe). Beneath the outer layered neocortex is the white matter which consists of myelinated and unmyelinated axons traversing and interconnecting various regions of the brain. In the depths of each hemisphere subcortical and limbic structures predominate, with each half of the brain possessing an amygdala, hippocampus, and so on.
The two halves of the brain are also interconnected with thick ropes of myelinated and unmyelinated axons which form the corpus callosum and anterior commissure. Via these axonal pathways, the right and left halves of the brain, and the various lobes and subcortical structures, can interact and exchange information. This enables tasks to be performed more efficiently, as, literally "two heads are better than one."
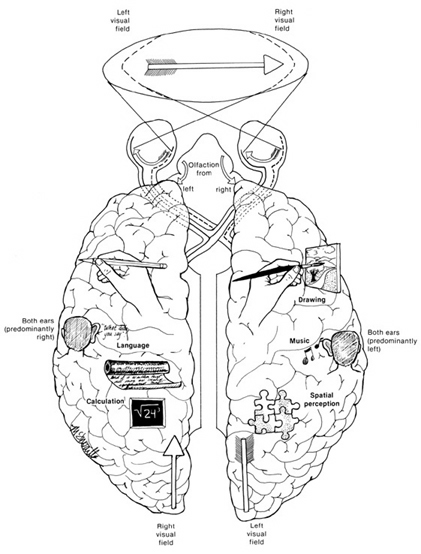
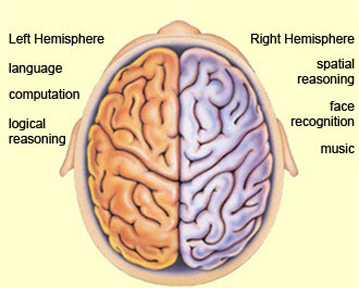

3. TWO BRAINS - TWO MINDS
That the right half of the brain is capable of conscious experience has now been well demonstrated in studies of patients who have undergone complete surgical destruction of the corpus callosum (i.e. corpus callosotomies, AKA split-brain operations (Bogen, 1969; Sperry 1966, 1968, 1974, 1982). These surgeries have been performed for the purposes of controlling intractable epilepsy. However, following surgery, the right and left half of the brain can function semi-independently, and when they do, they demonstrate the presence of two minds in the same head (Joseph 1986b, 1988a,b).
As described by Nobel Lauriate Roger Sperry (1966, p. 299), "Everything we have seen indicates that the surgery has left these people with two separate minds, that is, two separate spheres of consciousness. What is experienced in the right hemisphere seems to lie entirely outside the realm of awareness of the left hemisphere. This mental division has been demonstrated in regard to perception, cognition, volition, learning and memory."
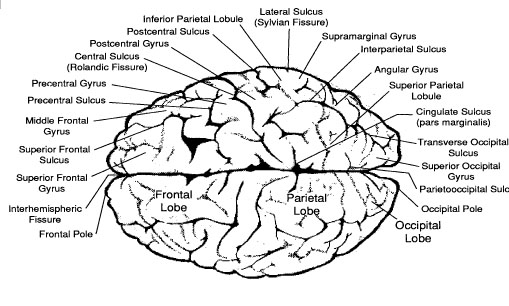
Normally, visual and tactile information from the left half of visual-tactile environment is transmitted to the right half of the brain which controls the left extremities, and input from the right visual-tactile environment the left half of the brain which controls the right extremities. Likewise, auditory information received by the right and left ear are predominantly transmitted to the opposite half of the brain. Therefore, when a split-brain patient is tactually stimulated on the left side of the body, their left hemisphere will not know it. Since the left half of the brain controls the comprehension and expression of (non-emotional) language, split-brain patients are unable to name objects placed in the left hand, and they fail to report the presence of a moving or stationary stimulus in the left half of their visual fields (Bogen, 1979; Gazzaniga & LeDoux, 1978; Joseph, 1988b; Levy, 1974, 1983; Seymour et al. 1994; Sperry, 1982). In addition, the disconnected left hemisphere cannot verbally describe odors, pictures or auditory stimuli tachistoscopically or dichotically presented to the right cerebrum. Moreover, the left hemisphere has difficulty recognizing, identifying, or naming emotional and environmental sounds (Joseph, 1986b, 1988b)--capacities associated with the functional integrity of the right hemisphere.
By contrast, the disconnected right hemisphere is able to indicate when they are tactually or visually stimulated on the left side. When tachistoscopically presented with words to the left of visual midline, or when objects are placed in the left hand (but out of view), the right hemisphere can correct point with the left hand to the word or object--even when the left hemisphere claims it has seen or felt absolutely nothing.
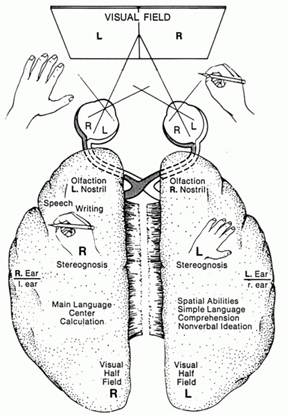

For example, when presented with words like "toothbrush", such that the word "tooth" falls in the left visual field (and thus, is transmitted to the right cerebrum) and the word "brush" falls in the right field (and goes to the left hemisphere), the left hand will point to the word viewed by the right cerebrum (i.e., tooth) and the right hand will point to the word viewed by the left hemisphere (i.e., brush). When offered a verbal choice, the speaking (usually the left) hemisphere will respond "brush" and will deny seeing the word "tooth."
4. SPLIT-BRAIN CASE DESCRIPTION
Patient 2-M (AKA "Two Minds") had his first seizure at age eight and suffered from complex partial seizures as well as status epilepticus (with up to 100 seizures per day). Medication was unable to control these seizures.
EEG and depth electodes demonstrated right frontal abnormalities which led to bilateral synchronous seizure activity. Independent discharges were noted throughout the left cerebrum. CT-scan failed to indicate the presence of any gross lesions. The patient was of low average intelligence (WAIS), and sodium amytal anesthetization demonstrated left hemisphere dominance for speech.
Following callosotomy at age 20, neurological examination demonstrated that his speaking, left hemisphere, was unable to perceive visual or tactile information from the left visual field and left half of the body. It was noted that his left arm and leg would not consistently follow commands and/or would respond in a maner opposite to what was requested. 2-M also complained that the left half of his body and left hand would sometimes behave independently and engage in actions that he, his left hemisphere, could not control. This condition remained basically unchanged even 3-years post surgery.
STEREOGNOSIS: When common objects were placed in the right hand, 2-M named them without difficulty. When placed in the left hand he was not able to name them correctly; nor was he able to indicate, verbally or with the right hand (via pointing) the correct object when offered multiple (visual) choices: 0% accuracy. When asked to point with the left hand to objects felt with the left hand, 2-M responded with 90% accuracy.
DIFFERENTIAL SIMULTANEOUS TACTILE STIMULATION. This task involved simultaneously stimulating both hands (while out of view) with either the same or two different textured materials (e.g. sandpaper to the right, velvet to the left) and required the patient to point (with the left and right hand simultaneously) to an array of fabrics hanging in view on the left and right of the testing apparatus. Hence, following stimulation to hands while out of view, he would pull his hands out from inside the apparatus and point with the left to the fabric felt by the left, and with the right to the fabric felt by the right.
2-M almost consistently pointed with the left hand to the fabric experienced by that extremity and with the right hand t the material experienced by the right hand.
Suprisingly, when 2-M was stimulated with two different fabrics, e.g sandpaper to the right, wire mesh to the left, although his left hand (right hemisphere) responded correctly and pointed to the correct object, his left hemisphere vocalized: "Thats wrong!" Repeatedly he reached over with his right hand and tried to force his left extremity to point to the fabric experienced by the right (although the left hand responded correctly!). His left hand refused to be moved and physically resisted being forced to point at anything different. The left hand/right hemisphere insisted on pointing to the correct fabric applied to the left hand, whereas t he right hand/left hemisphere tried to force the left hand to point to the material experienced by the right. In one instance a physical struggle ensued, the right grappling with and hitting the left arm and hand.
Moreover, while performing this (and other tasks), 2-M made statements such as: "I hate this hand", or, "this is so frustrating", and would strike his left hand with his right or punch his left arm. Hence, it was readily apparent that 2-Ms left hemisphere had no idea that his left hand was responding correctly. Not once, however, did his left hand attempt to interfere with the choices made by the right.
UNILATERAL VS WHOLE BODY MOVEMENTS TO COMMAND. 2-M was presented with a series of 10 unilateral commands requiring movement of half the body (e.g. "salute like a soldier") or 10 whole body comands (e.g. "stand like a boxer"). He was able to respond correctly to all commands involving the right half of the body but to only 3 of the 10 whole body commands. This indicates his language dominant left hemisphere was unable to program the movements of the left half of the body which is under the control of the right hemisphere. In these instances, 2-M remarked that he hated the left half of his body.
VISUAL SPATIAL SKILLS. 2-M was required to draw and copy several geometric forms and objects (e.g. a flower) as well as a clock with all the numbers. The drawings and copies made by 2-M were distorted, macrographic, and he was unable to reproduce figures involving perspective with his right hand. Left sided neglect and inattention were also noted in his drawings. For example, when asked to draw the face of a clock, he drew only half the clock, and all the numbers appeared on the right half of his drawing.
RESPONSES TO T-SCOPE VISUAL-PICTORIAL STIMULI. The right and left cerebral hemisphere were separately and individually presented, via tachistoscope, with 40 visual-pictorial scenes (e.g. a boy with a ball, a man with an umbrella), 20 written words (e.g. "Hat"), and 10 digits. All visual stimuli were presented 3 degrees to the left or right of visual midline for 150 msec. 2-M was required to describe the pictures and to verbalize the words or digits presented.
He responded with 100% accuracy when the right visual field (left hemisphere) was stimulated; that is, he was able to accurately verbally described what he had seen. However, when the right hemisphere was presented with visual-pictorial material, 30% of his verbal responses were wildly confabulatory and characterized by gap filling. For example, when the right hemisphere shown a picture of a man walking in the rain and carrying an umbrella, he replied: "A pizza man making a pizza at a pizza joint". This indicates that some of this visual material was transferred to the left hemisphere, but it was incomplete. In consequence, the left hemisphere filled the gap with confabulatory ideation.
Following complete callosotomy 2-M displayed the disconnection syndromes unique to commissurotomy patients (Bogen, 1979; Gazzaniga & LeDoux, 1978; Sperry, 1966). Hence, when tactually stimulated on the left side of the body, (the left hemisphere's of) he demonstrated marked neglect when verbal responses were required, was unable to name objects placed in the left hand, and failed to report the presence of a moving stimulus in the left half of his visual fields. In addition, he demonstrated marked difficulties naming incomplete figures (and thus forming visual closure) as well as a reduced ability to name and identify non-linguistic and environmental sounds --capacities associated with the functional integrity of the right hemisphere (which was not afforded an opportunity to respond in this series of tests).
However, by raising his left hand, 2-M's right hemisphere was able to indicate when he was tactually or visually stimulated on his left side. When tachistoscopically presented with words to the left of visual midline, although unable to name them, when offered mutiple visual choices in full field his right hemisphere was able to point correctly with the left hand to the word viewed. With his left hand he was also able to respond correctly on 90% of the texture identification trials and resisted attempts by his right hand to make it point elsewhere.
In addition, he frequently experienced and demonstrated difficulty making the left half of his body respond to verbal and left hemisphere mediated commands. The left half of his body often responded independently of the left hemisphere, and contrary to its wishes. Indeed, the functional and expressive independence of 2-Ms right cerebrum caused this individual (or at least his left hemisphere) a considerable degree of frustration, anger and embarrassment. In this regard he complained of situations where his left hand would turn off or change chanells on the T.V. (even though he, i.e., his left hemisphere was enjoying the program), or his left leg refuse to walk in certain directions, act in a manner completely opposite to what he expressively intended, or his left hand would perform socially inappropriate actions (such as attempting to strike a relative).
In the laboratory, on several occassions, he became angry with his left hand, swore and expressed "hate" for it, struck his left hand/arm with his right hand, and engaged in physical struggles with his left extremity, the right attempting to force the left to comply with some particular activity or to cease to act in a maner which the left hemisphere found objectionable or annoying. In many (if not most) instances, the left resisted and continued to behave in the manner directed by the right hemisphere.
In general, these findings indicate the isolated right cerebral hemisphere of 2-M was able to understand and follow certain simple verbal commands, obtain and maintain knowledge of various visual, auditory, lingustic, and somesthetic events, as well as respond, make decisions and act purposefully --even when his left hemisphere had no idea as to what information the right possessed and why it was behaving in a particular manner.
Given the differential behavioral observations regarding the activity and responsiveness of the left half of his body, it appears that 2-M is possessed of two minds, one of which resides within the right, the other within the left cerebral hemisphere. Nevertheless, it appears that 2-Ms left hemisphere remains predominant in regard to most expressive activities.
6. RIGHT BRAIN PERVERSITY
The disconnected right and left cerebral hemispheres, although unable to communicate and directly share information, are nevertheless fully capable of independently generating and supporting mental activity (Bogen, 1969, 1979; Gazzaniga & LeDoux, 1978; Joseph, 1986b, 1988a,b; Levy, 1974, 1983; Sperry, 1982). In the right hemisphere we deal with a second form of awareness which differ dramatically from the mental system of te left, but which accompanies in parallel what appears to be the "dominant" temporal-sequential, language dependent stream of consciousness in the left cerebrum.
Moreover, as has been demonstrated by Sperry, Bogen, Levy, and colleagues, the isolated right cerebral hemisphere, like the left, is capable of self-awareness, can plan for the future, has goals and aspirations, likes and dislikes, social and political awareness, can purposefully initiate behavior, guide responses choices and emotional reactions, as well as recall and act upon certain desires, impulses situations or environmental events --without the aid, knowledge or active (reflective) participation of the left half of the brain.
In that the brain of the normal as well as "split-brain" patient maintains the neuroanatomy to support the presence of two psychic realms, it is surprising that a considerable degree of conflict does not arise during the course of everyday activity. Frequently (such as in the case of the "split-brain" patient, LB, described below), although isolated the right half of the brain is fully willing to assist the left in a myriad of activities. Presumably such difficulties do not occur because both minds, having once been joined, share the same goals and interests. However, common experience seems to argue otherwise, for even in the intact individual, psychic functioning often is plagued by conflict.
In its most subtle manifestations the disconnected right hemisphere may attempt to provide the left with clues when the left (speaking) hemisphere is called upon to describe or guess what type of stimulus has been secretly shown to the right (such as in a T-scope experiment). Because the corpus callosum has been severed transfer and information exchange is not otherwise possible.
Hene, when a picture has been shown to the right and the left has been asked to guess, the right hemisphere may listen and then nod the head or clear the throat so as to give clues or indicate to the left cerebrum that it has guessed incorrectly. In one case the right hemisphere attempted to trace or write an answer on the back of the right hand (e.g. Sperry et al. 1979). For example, after the right hemisphere was selectively shown a picture of Hitler, and then asked to indicate their attitude toward it before verbally describing it, the patient signalled "thumbs down".
EX: "That's another 'thumbs-down'?"
LB: "Guess I'm antisocial."
EX: "Who is it?"
LB: "GI came to mind, I mean..." Subject at this point was seen to be tracing letters with the first finger of the left hand on the back of his right hand.
EX: "You're writing with your left hand; let's keep the cues out."
LB: "Sorry about that."
Nevertheless, the behavior of the right hemisphere is not always cooperative, and sometimes it engages in behavior which the left cerebrum finds objectionable, embarrassing, puzzling, mysterious, and/or quite frustrating. This is probably true for the normal as well as the "split-brain" individual.
For example, Akelaitis (1945, p. 597) describes two patients with complete corpus callosotomies who experienced extreme difficulties making the two halves of their bodies cooperate. "In tasks requiring bimanual activity the left hand would frequently perform oppositely to what she desired to do with the right hand. For example, she would be putting on clothes with her right and pulling them off with her left, opening a door or drawer with her right hand and simultaneously pushing it shut with the left. These uncontrollable acts made her increasingly irritated and depressed."
Another patient experienced difficulty while shopping, the right hand would place something in the cart and the left hand would put it right back again. Both patients frequently experienced other difficulties as well . "I want to walk forward but something makes me go backward. ' A recently divorced male patient noted that on several occasions while walking about town he found himself forced to go some distance in another direction. Later (although his left hemisphere was not conscious of it at the time) it was discovered (by Dr. Akelaitis) that this diverted course, if continued, would have led him to his former wife's new home.
Geschwind (1981) reports a callosal patient who complained that his left hand on several occasions suddenly struck his wife--much to the embarrassment of his left (speaking) hemisphere. In another case, a patient's left hand attempted to choke the patient himself and had to be wrestled away.

In addition, Bogen (1979, p. 333) indicates that almost all of his "complete commissurotomy patients manifested some degree of intermanual conflict in the early postoperative period." One patient, Rocky, experienced situations in which his hands were uncooperative; the right would button up a shirt and the left would follow right behind and undo the buttons. For years, he complained of difficulty getting his left leg to go in the direction he (or rather his left hemisphere) desired. Another patient often referred to the left half of her body as "my little sister" when she was complaining of its peculiar and independent actions.
A split-brain patient described by Dimond (1980, p. 434) reported that once when she had overslept her "left hand slapped me awake." This same patient, in fact, complained of several instances where her left hand had acted violently. Similarly, Sweet (1945) describes a female patient whose left hand sometimes behaved oppositionally and in a fashion which on occasion was quite embarrassing.


The split-brain patient, 2-M, frequently was confronted with situations in which his left extremities not only acted independently, but engaged in purposeful and complex behaviors --some of which he (or rather, his left hemisphere) found objectionable and annoying. He complained of instances in which his left hand would perform socially inappropriate actions (e.g. attempting to strike a relative) and would act in a manner completely opposite to what he expressively intended, such as turn off the TV or change channels, even though he (or rather his left hemisphere) was enjoying the program. Once, after he had retrieved something from the refrigerator with his right hand, his left took the food, put it back on the shelf and retrieved a completely different item "Even though that's not what I wanted to eat!" On at least one occasion, his left leg refused to continue "going for a walk" and would only allow him to return home.
In the laboratory, he often became quite angry with his left hand, he struck it and expressed hate for it. Several times, his left and right hands were observed to engage in actual physical struggles with the right hand striking the left. His left hemisphere also made statements such as: "I hate this hand" or "This is so frustrating" and would strike his left hand with his right or punch his left arm. In these instances there could be little doubt that his right hemisphere was behaving with purposeful intent and understanding, whereas his left brain had absolutely no comprehension of why his left hand (right hemisphere) was behaving in this manner.
7. LATERALIZED GOALS AND ATTITUDES
Why the right and left cerebral hemispheres in some situations behave cooperatively and yet in others in an oppositional fashion is in part a function of functional lateralization and specialization and the differential representation of social-emotional analytical abilities predominantly within the right hemisphere. Hence, because each hemisphere is concerned with different types of information, even when analyzing ostensibly the same stimulus may react, interpret and process it differently and even reach different conclusions (Joseph, 1988a,b). Moreover, even when the goals are the same, the two halves of the brain may produce and attempt to act on different strategies.
Functional lateralization may thus lead to the development of oppositional attitudes, goals and interests. For example, one split brain individual's left hand would not allow him to smoke, and would pluck lit cigarettes from his mouth or right hand and put them out. Apparently, although his left ceebrum wanted to smoke, his right hemisphere didn't approve -apparently he had been trying to quite for years.
Nevertheless, these difficulties are not limited to split-brain patients, for conflicts of a similar nature often plague "normal" individuals as well. It doesn't take split-brain surgery for an individual to express self-hate or to engage in self-destructive behaviors.
8. LATERALIZED MEMORY FUNCTIONING & CONFABULATION
Although a variety of neurochemical and neuroanatomical regions are involved in the formulation of memory, functional specialization greatly determines what type of material can be memorized or even recognized by each half of the cerebrum. This is because the code or form in which a stimulus is represented in the brain and memory is largely determined by the manner in which it is processed and the transformations that take place. Because the right and left cerebral hemispheres differentially process information, the manner in which this information is represented also will be lateralized (Bradshaw & Mattingly, 1997). Hence, some types of information only can be processed or stored by the right vs. the left cerebrum.
For example, it is well known that the left hemisphere is responsible for the encoding and recall of verbal memories, whereas the right cerebrum is dominant in regard to visual-spatial, non-verbal, and emotional memory functioning (Joseph 1982, 1988a, 1990a,b, 1992, 1993, 1996, 2003). If the left temporal lobe were destroyed, verbal memory functioning would become impaired since the right cerebrum does not readily store this type of information. Conversely, the left has great difficulty storing or remembering nonlinguistic, visual, spatial, and emotional information including the ability to recall or recognize emotional faces or personal emotional memories.
Hence, it is the left hemisphere which is responsible for the encoding and recall of verbal, temporal-sequential, and language related memories, whereas the right cerebrum is dominant in regard to visual-spatial, non-verbal, and social emotional memory functioning. Each hemisphere stores the type of material that it is best at recognizing, processing, and expressing.
9. UNILATERAL MEMORY STORAGE: HIDDEN MEMORIES
In the intact, normal brain, even non-emotional memory traces appear to be stored unilaterally rather than laid down in both hemispheres (Bures & Buresova 1960; Doty & Overman 1977; Hasegawa et al., 1998; Kurcharski et al. 1990; Levy, 1974; Risse & Gazzaniga, 1979). Moreover when one hemisphere learns, has certain experiences, and/or stores information in memory, this information is not always available to the opposing hemisphere; one hemisphere cannot always gain access to memories stored in the other half of the brain (Bures & Buresova 1960; Doty & Overman 1977; Hasegawa et al., 1998; Joseph, 1986b, 1988ab, 1992b; Kucharski et al. 1990; Levy, 1974; Risse & Gazzaniga 1979).
To gain access to these lateralized memories, one hemisphere has to activate the memory banks of the other brain half via the corpus callosum (Doty & Overman, 1977; Hasegawa et al., 1998) or anterior commissure (Kucharski et al. 1990). This has been demonstrated experimentally in primates. For example, after one hemisphere had been trained to perform a certain task, although either hemisphere could respond correctly once it was learned, when the commissures were subsequently cut, only the hemisphere that originally was trained was able to perform--i.e., could recall it. The untrained hemisphere acted as though it never had been exposed to the task; its ability to retrieve the original memories was now abolished (Doty & Overman, 1977; Hasegawa et al., 1998).
In a conceptually similar study, Risse and Gazzaniga (1979) injected sodium amytal into the left carotid arteries of intact patients so as to anesthetize the left cerebral hemisphere. After the left cerebrum was inactivated, the awake right hemisphere, although unable to speak, was still able to follow and behaviorally respond to commands, e.g., palpating an object with the left hand.
Once the left hemisphere had recovered from the drug, as determined by the return of speech and motor functioning, none of the eight patients studied was able to verbally recall what objects had been palpated with the left hand, "even after considerable probing." Although encouraged to guess most patients refused to try and insisted that they did not remember anything. However, when offered multiple choices in full field, most patients immediately raised the left hand and pointed to the correct object!
According to Risse and Gazzaniga (1979), although the memory of touching and palpating the object was not accessible to the verbal (left hemisphere) memory system, it was encoded in a nonverbal form within the right hemisphere and was unavailable to the left hemisphere when normal function returned. The left (speaking) hemisphere was unable to gain access to information and memories stored within the right half of the brain. Nevertheless, the right brain not only remembered, but was able to act on its memories.
This indicates that when exchange and transfer is not possible, is in some manner inhibited, or if for any reason the two halves of the brain become functionally disconnected and are unable to share information, the possibility of information transfer at a later time is precluded ) -even when the ability to transfer is acquired or restored. The information is lost to the opposite half of the cerebrum.
Moreover, because some types of information are processed by the right and left hemisphere in a wholly different fashion, they are unable to completely share or gain access to the data or even the conclusions reached by the other -as they are unable to process or recognize it -which in turn precludes complete interhemispheric transfer; information is lost during the transfer process.
Nevertheless, although inaccessible or lost, these memories, details, and attached feelings can continue to influence whole brain functioning in subtle as well as in profound ways. That is, one hemisphere may experience and store certain information in memory and at a later time in response to certain situations act on those memories, much to the surprise, perplexity, or chagrin of the other half of the brain; one hemisphere cannot always gain access to memories stored in the other half of the brain.
Dreaming and Forgotten Dreams. Of course, complete functional deactivation is probably quite rare in the normal brain. However, there is some evidence to suggest that interhemispheric communication is reduced, for example, during sleep and possibly during dreaming (Banquet, 1983; Joseph, 1988a).
Most dreaming occurs during REM, which possibly is associated with right hemisphere activation and low-level left hemisphere arousal (Goldstein et al. 1972; Hodoba, 1986; Meyer et al. 1987). It also becomes progressively more difficult to recall one's dreams as one spends time in or awakens during NonREM (Wolpert & Trosman, 1958), which is associated with high left hemisphere and low right brain activation (Goldstein et al. 1972). Thus are dreams really forgotten, or are they locked away in a code which is not accessible to the speaking left hemisphere?
10. LONG LOST CHILDHOOD MEMORIES
For most individuals it is extremely difficult if not impossible to verbally recall events which occurred before the age of three and a half (Joseph, 1998, 1999a, 2003). There are several reasons for this: Information processed and experienced during infancy vs. adulthood is stored via certain transformations and retrieval strategies which are quite different. As the brain matures and new information processing strategies are learned and developed, the manner in which information is processed and stored is altered. Although these early memories are stored within the brain, the organism no longer has the means of retrieving them, i.e., the key no longer fits the lock.
That is, early experiences may be unrecallable because infants use a different system of codes to store memories whereas adults use symbols and associations (such as language) not yet fully available to the child (Joseph, 1982, 2003). Much of what was experienced and committed to memory during early childhood took place prior to the development of lingusitic labeling ability and was based on a pre- or nonlinguistic code (Joseph 2003). Hence, the adult, who is relying on more sophisticated and language-related coding systems, cannot find the right set of neural programs to open the door to childhood memories. The key does not fit the lock because the key and the lock have changed.
The inability to recall early memories is also a function of programmed cell death--the loss of memory-laden neurons which are shed by the millions over the course of early development and the immaturity of the corpus callosum in children (Joseph, 1982, 1999a,b). That is, non-verbal information perceived and processed by the right vs left hemisphere is generally stored in the right vs left hemisphere. Later, when the commissures mature, this information cannot be transferred except under special conditions.
However, under conditions of traumatic memory loss; e.g. repression, not just the the right and left hemisphere, but the differential activation of the amygdala and hippocampus are contributory; and later the recall of such memories may be opposed by the frontal lobes; the right frontal in particular (Joseph 1986a, 1988a, 1999c)
11. EMOTION AND RIGHT BRAIN FUNCTIONING IN CHIDLREN
As is now well known, the developing organism is extremely vulnerable to early experience during infancy such that the nervous system, perceptual functioning and behavior may be altered dramatically (Casagrande & Joseph 1978; 1980; Joseph 1979, 1998, 1999a,b, 2003; Joseph & Casagrande 1980; Joseph & Gallagher 1980; Joseph et al. 1978). There is some evidence that the right cerebral hemisphere and the right amygdala may be more greatly affected (Joseph 1998, 2003).
Moreover during these same early years our traumas, fears, and other emotional experiences, like those of an adult, are mediated not only by the limbic system, but also via the nonlinguistic, social-emotional right hemisphere. And, just as they are in adulthood, these experiences are stored in the memory banks of the right cerebrum.
However, much of what was experienced and learned by the right half of the brain during these early years was not always shared or available for left hemisphere scrutiny (and vice-versa). That is, a childs two hemispheres are not only functionally lateralized, but limited in their ability to share and transfer information. In many ways, infants and young children have split-brains (Gallagher & Joseph 1982; Joseph & Gallagher, 1985; Joseph et al. 1984).
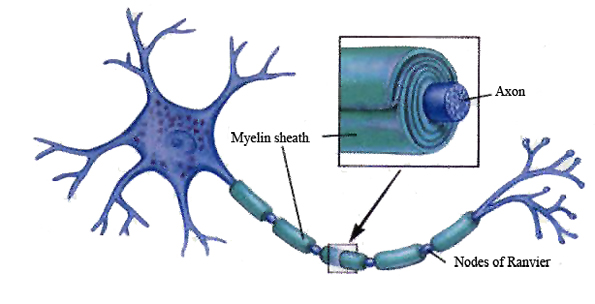

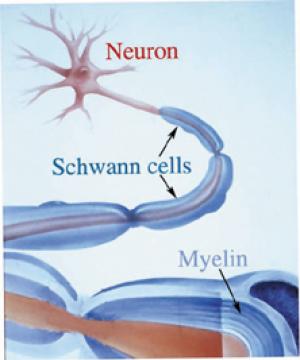
The corpus callosum, the major axonal fiber pathway that connects the right and left half of the brain, takes over 10 years to fully myelinate (Yakovlev & Lecours, 1967).
Indeed, although pain can be transmitted and received via axons devoid of myelin, this type of data is completely lacking in complexity and is devoid of initial cognitive attributes. Pain transmitting axons have very simple requirements. By contrast as data complexity increases, so too does the complexity of those neurons which transmit these signals (Konner, 1991; Ritchie, 1984).
For example, as axon diameter increases, so too does the extent of myelination. Similarly, quantities of axoplasm and cytoplasm, nuclear diameter, and neuronal packing density are also correlated with myelination (Konner, 1991). Conversely, lack of myelin, or those neurons which have not yet myelinated, are associated with an increased susceptibility to conduction failure and interference due extraneous influences, including signal modification by neighboring axons (Konner, 1991; Ritchie, 1984).
12. SPLIT-BRAIN FUNCTIONING IN CHILDREN
A considerable body of evidence has been presented showing that traumatic or surgical section of the corpus callosum results in interhemispheric deficits in information transfer. A number of investigators have provided evidence which purports to show that young children have similar difficulties in interhemispheric information transfer (Finlayson, 1976; Galin, Diamond and Herron, 1977; Galin, Johnstone, Nakell and Herron, 1979; Gallagher and Joseph, 1982; Joseph et al. 1976; Joseph et al., 1984; Kraft, Mitchell, Languis and Wheatley, 1980; O'Leary, 1980; Salamy, 1978). These deficits, presumably, are secondary to the prolonged development (e.g. myelination) of the callosum, the maturation of which is not complete until after the first decade (Yakovlev and Lecours, 1967). Hence, the capacity to transfer information between the cerebral hemispheres appears to progressively increase with age (Finlayson, 1976; Galin et al., 1977; Gallagher & Joseph, 1982; Kraft et al., 1980; Joseph & Gallagher 1985, Joseph et al., 1984; O'Leary, 1980).
Consonant with the behavioral evidence cited above are some electrophysiological studies. Briefly, Salamy (1978) failed to find ipsilateral evoked potentials (which are perhaps dependent on transfer via the corpus callosum) prior to age 3.5, while Joseph et al. (1976) found deficiencies in the spatio-temporal organization of the EEG and thus a general lack of interhemispheric linkage in newborn and premature infants.
The evidence is indicative of limited rather than absent interhemispheric information exchange in young children. Indeed, given the differing local rates of cortical and callosal fiber development there is likely to be considerable transfer depending on age and modalities employed. Furthermore, since the anterior commissure (a tract of fibers which interconnects the inferior temporal lobes and limbic areas) appears developmentally complete by the end of the first year of life, even the hemispheres of the very young child may be only partially disconnected.
Confabulation and Limited Interhemispheric Information Exchange In cases of complete section of the commissures each hemisphere appears ignorant of the cognitive activities occurring in the other. In cases of partial sectioning (e.g. sparing of the anterior commissure) some transfer occurs (Joseph 1986b), albeit limited and incomplete. For example, in one patient with an intact anterior commissure, following right hemisphere tachistoscopic pictorial stimulation all verbal reports, while containing some accurate descriptive elements, were largely contaminated with errors, gross embellishments, and other falsehoods (Joseph 1988b). Information transfer (from the right hemisphere) was thus characterized by confabulatory "gap-filling" (Joseph, 1982, 1986a,b, 1988a,b). The left hemisphere apparently attempted to make sense of the limited information received by filling the gaps with related ideation.
For example, when a complex picture, e.g. a man with a gun, was presented tachistoscopically to the patient's left hemisphere, he would typically respond with concrete and accurate two or three word replies, such as "guy with a gun". When the same stimulus was presented to the right hemisphere (requiring transfer to the language centers of the left) he would respond with elaborate, detailed, and vivid descriptions which bore little relation to the actual elements of the original stimulus around which other associations were erroneously anchored: "Gun, hold up...he has a gun and is holding up a bank teller, a counter separates them." Furthermore, the left hemisphere's of these patients believed the largely erroneous descriptions they provided. As noted by Gazzaniga and LeDoux (1978), the left hemisphere did not "offer its suggestion in a guessing vein but rather as a statement of fact" (pp. 148-149).
Likewise, because of the limited information exchange between the hemispheres of children, children also confabulate if questioned concerning pictorial stimuli presented to the right hemisphere (Joseph et al., 1984).
Confabulation. Anchors and Gaps. It has been argued and demonstrated (Joseph, 1982, 1986a,b, 1988a,b) that confabulation occurs when the language centers of the left hemisphere are isolated from sources of information about which the patient is questioned, such that complete and accurate information is not fully available. In these instances confabulation is a result of attempts to fill the gaps in the information received with associations which are in some manner related to the fragments available. Moreover, disconnection also results in failure to correct erroneous statements, as contradictory evidence is also not always accessible. Hence, confabulatory statements, although erroneous, often contain accurate elements around which erroneous (albeit related) ideations are anchored. However, confabulation may also result due to other factors such as frontal lobe dysfunction such that (in these instances) an uninhibited flow of irrelevant information is transmitted to and organized by the language regions.
Confabulation. For the purposes of this study confabulation was defined as an erroneous embellished description, such that perceptual-pictorial information not originally presented was inaccurately inserted and included in the subject's report. Hence, a detail may be reported correctly but the meaning of the detail may become overly significant and erroneously generalized such that a concept results which has little or, in fact, no bearing on the original stimulus.
Information Gaps and Inclusion Score An important feature of the gap filling hypothesis is the notion that information degradation and confabulation should be directly related, such that, the larger the gap, the more likely is the individual to confabulate. Hence, for purposes of this study the major pictorial-perceptual elements (features) of each tachistoscopic stimulus were determined. For example, if in response to a picture of a girl blowing out the candles on a birthday cake, a subject responded, 'girl" or "cake", the inclusion score would be "I" (as opposed to "2" for stating "girl" and "cake"). For each presentation the inclusion of these items in the child's verbal report was therefore tabulated.
Misses, Semantic, Syntactic, and Perceptual Errors Tabulation of the frequency of misses, semantic, syntactic and perceptual errors was largely for exploratory purposes. Briefly, misses were scored when no response was offered or the subjects claimed not to have seen the stimulus. Syntactic errors were noted when the subject's speech was agrammatical. A response was scored as a semantic error when the words used in the description, although incorrect, were from the correctly aroused semantic group pertaining to the stimulus. The erroneous response therefore bore a meaningful resemblance to the original stimulus. In contrast, a perceptual error was tabulated when an erroneous response/description bore an obvious similarity to some aspect of the stimulus whole; for example, calling, calling a "cake" a "drum". However, unlike a confabulation, no erroneous information could be added (for either a semantic or perceptual error). In this respect it was hypothesized that a perceptual error probably would result from a breakdown of serial feature-by-feature visual scene analysis as well as a failure to adequately engage in visual exploration. Thus, only certain details are recognized in isolation and stripped of related features. Hence, the left hemisphere should perform more poorly.
Subjects All subjects were right handed, aged 4, 7, 10. A total of 132 subjects were tested, with a minimum of 20 girls and 20 boys in each age group (4, N=48; 7, N=44; 10, N=40). Subjects were volunteers recruited from private schools and were obtained via parents' consent.
Materials All children were tested in an isolated and quiet room provided by the school. For assessing tendencies to confabulate or make other errors in description, a single-field tachistoscope was employed and all responses were tape-recorded. Visual stimuli consisted of pictures adopted from the Peabody Picture Vocabulary Test (PPVT). Eleven stimuli were employed for presentation to either the right, left, or visual-field midline, for a total of 33 presentations. In addition, two control stimuli and a pre-test "warm-up" stimulus from the PPVT were used. All stimuli were projected on a small white screen with a single fixation point (1.54 cm hole) at its center.
For the tactile-form recognition test, the standard Halstead-Reitan Form Board was employed. Briefly, this is a wooden rectangular box which contains a hole for hand insertion at is bottom front center. Across the face of the apparatus are embedded four small plastic chips, each of which is identical to one of the four stimulus objects (i.e. cross, triangle, square, circle) used for non-visual tactile exploration.
Procedure for Tactile-Form Recognition Testing Prior to testing each child was questioned about and asked to demonstrate (on 3 tasks) hand usage. Inclusion criterion was right handedness on 2 of the 3 tasks. After being familiarized with the stimuli and procedures the subject was instructed to place his right hand through the hole of the apparatus. A single chip was placed in the fingers and the subject encouraged to feel it until he knew what it was. The subjects were then instructed to release the chip, remove his hand, and thus use the same hand (tactile-visual non-transfer, TVNT), to point to the object on the face of the form board. After 4 trials, once with each chip, the procedure for non-transfer recognition was repeated for the left hand.
Instructions for the transfer condition were similar except that the child was asked to point with the opposite hand (tactile-visual transfer, TVT) to the correct object. After this procedure was repeated once for each object, the child was instructed to insert the left hand and the transfer trials were repeated.
Testing always began with the right hand. However, for approximately half of the subjects at each age level, testing began with the non-transfer trials, and with the transfer trials for the remaining subjects.
Procedure for Tachistoscopic Testing Following the Tactile Form Recognition tests, each subject was presented with a picture from the PPVT and encouraged to give a detailed description of it. The child was then seated facing the viewing screen, asked to maintain visual fixation on the center point even when the picture fell to either side, and asked to describe each picture presented. Two practice trials in which images were presented at the screen's center were run and the child encouraged to describe what was seen. Testing was then initiated and 33 trials were run.
All stimuli were presented once to each visual field, the right or left hand edge at least 3 degrees from the central fixation point, for exactly 150 msec., and last to the screen's midline. Left and right presentations were counter-balanced (i.e. stimuli presented initially to the right then left and midline, 5 different stimuli initially to the left then right and midline, and one stimulus presented initially the midline followed by left then right sided presentations). All slides were presented in a pre-established random order which remained constant for all subjects. Eye movements were carefully monitored by an observer hidden behind the viewing screen, via the center-fixation hole.
After each presentation the subject was asked, "What did you see?" However, if the child's response was a one word reply (e.g. "girl"), the experimenter asked "what else did you see?" If the child again responded with a one word description the next stimulus was presented and the procedure repeated.
Scoring and Rater Reliability Three raters, blind to mode of presentation (i.e. right, left, midline) were given intensive instruction and training in scoring procedures and criteria for the tachistoscopic responses, and extensive practice in the scoring of protocols. Interrater reliability, based on the scoring of 8 research protocols, yielded reliability coefficients in excess of .90.
RESULTS A 4 x 2 x 3 (Experimenter by Sex by Age) analysis of variance (ANOVA) was performed to control for experimenter influences and for the six dependent T-scope variables: Inclusion, Confabulation, Semantic, Syntactic and Perceptual errors, and Misses, and the two tactile variables, tactile-visual transfer (TVT) vs tactile-visual non-transfer (TVNT) which were treated as repeated measures. For each T-scope dependent variable, mode of hemispheric tachistoscopic presentation (right, left, midline) were treated as repeated measures. No significant sex differences were discovered.
Inclusion Score Significant main effects were found for age (Fw96.20, p< .00001) and mode of visual-field presentation (Fw108.94, p<.00001). A Newman-Keuls (NK) analysis of means indicated that 4-year olds had significantly lower inclusion scores (summed across presentation modes) than 7 (Qw16.31, p<.01) or 10-years old children (Qw22.68, p<.01), and that subjects age 7 responded with significantly fewer inclusion items than the 10-year old group (Qw6.37, p<.01). In addition, across all age groups inclusion scores were found to be significantly larger for left hemispheres presentation than for right (Qw1.202, p<.01). Midline exposures also resulted in a significantly grater number of inclusion elements than left (Qw2.47, p<.01) or right presentation (Qw3.672, p<.01). However, this latter finding is probably due to having viewed all stimuli two times prior to midline presentations (once right and once left). The NK analysis of experimenter influences did not reveal any significant main effects.
Confabulation The ANOVA demonstrated a significant effect for age (Fw26.25, p<.00001) and visual field presentation (Fw64.37, p<.00001). The NK analysis on ordered pairs of group means indicated that 4-year olds made significantly more confabulatory responses that 7 (Qw4.24, p<.01) or 10-year olds (Qw5.383, p<.01). The age 7 group also confabulated significantly more than the older children (Qw1.04, p<.05). In addition, more confabulatory responses were found to occur following right vs. left hemisphere presentation (Qw.803, p<.01), or midline exposures (Qw.834, p<.01).
A significant interaction between presentation site and age was also indicated (Fw4.26, p<.002), such that regardless of right hemisphere (RH), left hemisphere (LH) or midline (MH) presentation, 4-year olds confabulated significantly more than 7 (RH: Qw2.133, p<.01; LH: Q<.932, p<.01; MH: Qw1.178; p<.01) or 10-year old children (RH: Qw2.492, p<.01; HM: Qw1.375, p<.01; MH, Qw1.517, p<.01). In addition, among the children age 4, there were significantly more confabulatory responses to the right vs. left (Qw1.542, p<.01) or midline presentation (Qw2.25, p<.01). Among 7 and 10-year olds no significant age or presentation mode differences were discovered. No significant main effects for experimenter influences were uncovered.
Semantic and Syntactic Errors and Misses No significant main effects for syntactic errors were found. A main effect for semantic errors was discovered (Fw9.10, p<.001), such that 10-year old subjects made fewer errors than 4 (Qw2.66, pw.01) or 7-year old children (Qw2.386, p<.01). Children age 4 and 7 did not significantly differ.
Significant differences in the tendency to omit responses (Misses) were found to be age dependent (Fw8.86, p<.0003). It was determined that 4-year olds had significantly more misses than 7 (Qw.602, p<.01) or 10-year olds (Qw.775, p<.01). The 7 and 10-year groups were not found to significantly differ.
Perceptual Errors Significant age-related differences in the tendency to commit perceptual errors were noted by the ANOVA (Fw5.67, p<.005). The NK analysis indicated that 4-year old children made significantly more perceptual errors than 7 (Qw.442, p<.01) or 10-year olds (Qw.994, p<.01). In addition, children age 7 committed more perceptual errors than older group (Qw.554, p<.01).
In regard to overall scores summed across age and mode of presentation, it was also noted that the experimenters were associated with significantly different amounts of perceptual errors (Fw7.33, p<.0002), such that Experimenters (E) 1 and 3 significantly differed from each other and from E2 and E4 (p<.01), who in turn did not differ from each other (Qw.12, pwNS). A check of the raw data for this and all other effects associated with the different subjects tested by each experimenter indicated, however, that these differences are largely a function random subject variations (probably associated with demographic characteristics- e.g. socio-environmental influences) and the tendency of a few subjects to respond with an unusually large number of errors.
Tactile-Form Recognition When considering total error scores for both the transfer (TVT) and non-transfer conditions (TVNT), significant differences were found across age groups (Fw8.86, p<.0003), such that the 4-year group made more errors overall than did 7 (Qw.6818, p<.01) or 10-year olds (Qw.7375, p<.01). Also, regardless of age, significantly more transfer vs non-transfer errors were discovered (Fw13.11, p<.0004). In addition, the ANOVA showed a significant interaction effect for task and age (Fw7.94, p<.0006). In this regard, 4's made significantly more transfer errors than either 7 (Qw.489; p<.05) or 10's (Qw.575, p<.01), and 4-year olds, unlike older subjects, were found to perform more poorly on the transfer vs non-transfer tactile conditions (Qw.4375, p<.05).
Correlations Among Tachiscopic and Tactile Conditions > A multiple correlational analysis (SCSS) demonstrated, as predicted, a highly significant inverse relationship between inclusion scores (i.e. pictorial elements accurately reported) and confabulation (rwy.484, p<.001) across modes of hemispheric presentation. Hence, the fewer pictorial elements reported, the more likely was there a tendency to confabulate. In addition, age was highly correlated with inclusion scores (rw.764, p<.001) and confabulatory responding (rwy.530, p<.005), confirming that with increasing age the number of pictorial features reported (inclusion score) increases, whereas the tendency to confabulate significantly diminishes.
Interestingly, both TVT (transfer) and TVNT (non-transfer) tactile-recognition errors were found to be positively associated with confabulatory responding (rw.215, p<.01; rw.192, p<.05, respectively), and negatively linked with inclusion scores (TVT: rwy.336, p<.01; TVNT: rwy.158, p<.05), with the overall magnitude of correlation for both confabulation and inclusion score being grater for transfer errors.
13. SPLIT-BRAIN FUNCTIONING IN CHILDREN: DISCUSSION
The results support the hypothesis that children have significant difficulty performing tasks which require inter-hemispheric transfer of tactile and visual material at age 4, a time period beyond which language has become lateralized to the left hemisphere. Verbal descriptions were thus often incomplete, impoverished, and erroneous when right and left hemisphere transfer was required.
For example, in response to the tachistoscopic presentation of a picture depicting a girl blowing out the candles on a birthday cake, many young children fail to report a number of major pictorial features (such as the girl or cake), and thereby earn a low inclusion score. Although after right hemisphere stimulation some elements are accurately reported (as determined by inclusion score), there is a significant failure to account for a number of major perceptual-pictorial elements as compared to the left hemisphere. It thus appears that some information is deleted or degraded during transfer.
In addition, a large number of children were found to erroneously extrapolate and to embellish their descriptions (i.e. confabulate), particularly when pictorial stimuli were tachistoscopically presented to the right hemisphere. This was especially evident among children age 4, as right hemisphere confabulatory errors greatly exceeded those of the left. In contrast, the transfer (i.e. from the right to left hemisphere) and non-transfer (left hemisphere presentation) confabulation scores of the older children were significantly fewer in number and did not significantly differ.
The lower the inclusion score (the fewer items reported), the more likely was a subject to confabulate. This latter significant inverse correlation, maintained regardless of age or presentation field, was particularly strong following right hemisphere stimulation and thus information transfer to the left cerebral cortex. Thus, in conjunction with information deletion (i.e. low inclusion score), erroneous information was somehow added to the subject's report, such that the fragments received became subsequently embellished. For example, in response to stimulus J (girl blowing out the candles on a cake), the child might respond: "a girl opening a package", or "a girl eating something", and thus be scored as confabulatory. In both instances, features such as cake, candles, girl blowing, are deleted and information such as opening a package, or eating something, are inserted.
Why didn't the child simply state, "I don't know" rather than confabulate when confronted by these gaps? We presume this response was rare because that information was not available to the language axis of the left hemisphere. According to the model discussed at length elsewhere (Joseph, 1982, 1986a) confabulation in part results in response to gaps in information, gaps which in turn appear to be due to the transmission of information which is degraded or incomplete when received by Wernicke's region. This type of confabulation is thus the result of the language center's attempts to organize and make sense of the limited information received, by insertion of additional information. The process of insertion is not random, but is partly determined by contextual variables (e.g. emotional status, past history), as well as the availability of associations/ideations which are linked to the perceived fragments of information. Essentially, the language axis of the left hemisphere does not know that it does not know as it attempts to make sense and report only what is transmitted to it.
It is noteworthy that unilateral left cerebral presentation also resulted in confabulation. This suggests that intra-hemispheric substrate immaturity as well as that of the corpus callosum may also be responsible for part of the observed findings. One candidate for the site of intra-hemispheric deficiencies is the inferior parietal lobule. This area is one of the last to myelinate (Flechsig, 1901), the last in which dendrites appear, and at age 4 is only 64% developmentally complete (Blinkov and Glezer, 1968). Fiber interconnections linking this with other neocortical areas are also not completely matured until approximately 7 years of age (Lecours 1975). In that the inferior parietal lobule presumably has a major role in information assimilation and transfer to Wernicke's region, the lack of maturation in this area may contribute significantly to the observed deficiencies.
Significant differences in transfer vs. non-transfer were not noted for misses (i.e. trails in which no response was elicited), perceptual, semantic, or syntactic errors. In part, the failure to discover transfer effects on these processes may be due to greater efficiency and degree of maturation in the anatomical systems involved, or inadequacies within the methodology. However, the former explanation does not seem completely valid as significant age-related differences were apparent. It is noteworthy that misses, confabulation, and perceptual and semantic errors were significantly large in number regardless of hemisphere of presentation. That is, significant developmental increases in the ability to process and express select forms of information was apparent on almost every measure employed including TVNT. In part, these findings bolster the suggestion advanced above, that possible intra-hemispheric age-related deficits/immaturities may be responsible for these errors in addition to (and perhaps independent of) corpus callosum immaturity.
Indeed, intra-hemispheric deficits in processing may be a significant factor which differentially influenced the overall results. That is, the tendency to make errors in either the right or left hemisphere independent of transfer, may, when transfer is required, give rise to errors in the right and left hemisphere and thus a larger error score independent of and/or addition to the corpus callosum.
On the other hand, particularly as related to semantic and perceptual errors, correct and efficient visual, tactile, and linguistic identification may require the dual and shared (cooperative) activity of both cerebral hemispheres. Hence, processing deficits within each hemisphere may due to an inability to gain complete access to perceptual activity occurring in the other. Assuming the mature callosum does not merely relay information but acts to mediate interhemispheric information integration by allowing for the participation and unique contribution of each hemisphere to the analysis of information, the immature callosum may in effect prevent or decrease each hemispheres, ability to contribute to and integrate the analysis being performed by the other. As such, unilateral processing may suffer as much as operations requiring transfer from one hemisphere to the other.
14. SPLIT-BRAIN COGNITION IN CHILDREN
Human psychological development and the expression of certain cognitive abilities parallels cerebral development and the myelination of the corpus callosum (Gallagher & Joseph, 1982; Joseph, 1982; Joseph, Gallagher, Holloway, & Kahn, 1984). Joseph et al. (1984) have provided evidence that indicates that information processing in certain modalities may be deficient in young children due to immaturity of the callosum as well as the inferior parietal lobule, a brain area intimately involved in cross-modal (auditory, visual, somesthetic) assimilation (Joseph, 1982). In consequence, information processing and the verbal expression and description of operations performed in other brain regions are often incomplete and/or erroneous due to incomplete transfer (e.g., from the right to left hemisphere). There is also evidence that strongly suggests that the immature corpus callosum, a pathway that takes over 10 years to mature, may be partially responsible not only for deficits in the interhemispheric transfer of learning, but also the failure of pre-operational children to demonstrate knowledge of operations involved in the conservation of liquid volume. Neural fiber pathway development and gross increments in brain tissue volume also appear to directly related to learning ability and the stages of cognitive development as defined by Piaget.
For example, conservation of volume is the ability to realise that two different containers may have the same volume, even though the containers are of a different shape. Specifically, Piaget demonstrated that if you show a child two beakers of water, one of which is short and fat, the other tall and thin, and ask the child which beaker contains more water, children under the age of 7 will generaly choose or say 'the tall one', even though both contain the same amount of water.
According to Piaget (1952, 1962, 1974), prior to the development of the understanding of conservation, the thought of the child is guided by "appearances" rather than logical thought or deductive reasoning. That is, appearances (or perception) dominates, such that, for example, if an object such as a ball of malleable clay is stretched out and elongated, the child may believe that the elongated clay is greater in size and has more volume than the ball of clay.
Piaget argued that children fail to "conserve" and are fooled by the differences in the size of the container, because children younger than 7 have not developed the cognitive and symbolic ability to conserve volume or to engage in reversible operations (to think back and make mental comparisons).
In order to engage in conservation, the child, therefore, must become capable of engaging in though patterns that allow for "reverse" thinking; i.e. to remember that the volumes of water were the same when in the same size contains, and that the ball of clay contains the same amount of clay despite changed in its shape.
Once a child reaches the age of 7 or 8, they become increasingly capable of performing reversible operations. Thus, if you pour water from the short wide glass into the tall, thin glass, the child realize there is the same amount of water as before, despite the illusion of appearances.
However, the nature of Piaget's conservations tasks almost always requires that decision making take place within the domain of language; the child is "asked" to engage in "verbal reasoning" and to indicate his or her understanding through language; that is, what is examined is verbal thought, and only those reasoning processes that can be communicated through language. Perception and reasoning, however, can also take place through the non-verbal domain (Joseph, 1982, 1986b, 1988a,b, 2010). Depending on the task, correct analysis and the demonstration of cognition may require the exchange of information between the right and left hemisphere; the left being dominant for language. However, if the left hemisphere is unable to give an accurate account of operations performed by the right until the callosum is sufficiently matured, it is likely that higher-order cognitive abilities mediated by the right actually may appear earlier than is suspected. That is, children have a non-linguistic knowledge which cannot be communicated verbally, as this information is processed and stored in the right half of the brain, but cannot be communicated to the language dominant left hemisphere.
For example, Kraft et al. (1980), recorded EEGs from the right and left parietal area of children 6 to 8, and found during the performance of a Piagetian conservation task that those who were able successfully to perform conservation operations responded with greater right hemisphere involvement during the response phase than those who could not. Gallagher and Joseph (1982) also found that non-conserving children engage the left cerebral hemisphere more than the right and more so than adults.
When coupled with the data presented by Gallagher and Joseph (1982) and colleagues (Joseph et al., 1984), it appears that young children fail to demonstrate successfully these operations because the left hemisphere, which is required to provide a response in the absence of right hemisphere input, confabulates an explanation based on current and externally available information. This strengthens the supposition that young children are in fact able to adequately process and perform certain operations, but are unable to give accurate verbal descriptions of their (the right hemisphere's) conclusions.
The right hemisphere is dominant for all aspects of emotion (Joseph 1982, 1988a, 1996). Interhemispheric information transfer from the right to the left hemisphere is also facilitated when questions and/or stimuli are couched in an emotionally arousing or reinforcing context even when the hemisphere are disconnected (Gazzaniga & LeDoux, 1978; Geschwind, 1965).
Hence, although the right and left hemispheres of children are partially disconnected due to corpus callosum immaturity, it is nevertheless possible to demonstrate that these children in fact posses knowledge concerning equivalence relationships and have the ability to perform internal reversals, even though they are in the "pre-operational-nonconserving" stage of childhood (Gallagher & Joseph 1982; Joseph & Gallagher 1985).
Cognition Experiment: Subjects, Materials & Procedures. Sixty-six children 4 and 5 years of age were assigned to one of three groups, with all groups approximately balanced for number and sex. Materials used for testing conservation included a small flask that contained colored water, two 6.5 x 5 cm cylindrical glass beakers, and one 19 x 2.5 cm glass cylinder.
Immediately prior to the experiment all participants were given a preliminary test to determine whether they were conservers. Each child was brought individually to the testing room and was seated at the table facing the experimenter. The child was asked to take the flask and pour equal amounts of colored water into the two short glasses. In all cases the experimenter would, if necessary, ensure equivalence by adding an appropriate amount. The experimenter then asked the child to take one of the beakers and pour its contents into the tall beaker. The child then was asked, 'Do these glasses both have the same amount, or does one have more?' One child responded correctly and was excused from further testing. Those who indicate that the taller beaker had more water were classified as non-conservers and then were tested according to one of the three conditions described below.
Condition 1. The experimenter asked the child to pour the liquid from the tall glass back into the short beaker used during the pretest. The child was asked whether the two now held the same amount or whether one had more. When the child confirmed that the two quantities were the same, he or she was asked to pour a little liquid from one short beaker into the other, so that one obviously would contain more. The child then was instructed to pour one of the short glasses (indicating the one with less liquid in it) into the tall beaker and was asked a relational question, 'which glass has more?' The experimenter then asked a question that required an empirical reversal of the presently perceived context, 'Suppose we were to pour it back into the other glass again. Which of the two (short glasses) would have more?' After responding, the child was asked actually to perform this reversal and to compare the quantities. He then was told to return the smaller amount of liquid to the tall cylinder, and the experimenter asked the child to imagine the stimulus in a reinforcing context and to indicate a preference: 'Let's pretend that you are very thirsty and that this is your favorite drink. Which one would you rather have?' These final three questions, relational, reversal, and preferences, were recorded as correct or incorrect as determined by the child's selection of the vessel that did indeed contain the greater volume of liquid.
Condition 2. Procedures for Condition 2 were exactly the same as for Condition 1 except that the reversal situation was eliminated. Thus the relational question was followed immediately by the preference question.
Condition 3. Condition 3 was identical to 1 except that the order of the first and third questions was reversed, so that the preference question was asked first and the one that required a specific relational judgment was asked last.
RESULTS The results were quite clear and supported the hypothesis. Chi square analyses were applied to the data, with correction factors computed when necessary. The preference question was answered correctly with significantly greater frequency than was the relational question, x2 = 9.98, p < .01. The reversal question also was answered correctly significantly more often than the relational question, x2 = 32.71, p < .005, and the preference question, x2 = 7.87, p < .025. No significant sex differences were found.
15. DISCUSSION: RIGHT HEMISPHERE COGNITION IN NON-CONSERVING CHILDREN
The results demonstrate that non-conserving children maintain an awareness of equivalence relationships, but are more likely to conserve when the situation involves imagining a rewarding experience, as opposed to merely verbally describing a currently perceived static relation between substances. Coupled with the data provided by Gallagher and Joseph (1982) this evidence demonstrates that the left hemisphere of non-conserving children does not have complete access to the results of right hemisphere cognitive processing. Thus the speaking left hemisphere appears ignorant of this information and provides incorrect answers, and these performance deficits can only be overcome when emotionally aroused which results in information transfer. In this regard, we believe that presenting the problem in the context of a reinforcer allowed for right hemisphere activation and, thus, inter-hemisphere transfer of this knowledge through the immature callosal fibers that connect the two cerebral hemispheres. A similar mode of inter-hemispheric information transfer in a split brain patient has been presented by Joseph et al., (1984).
As noted, a significant number of non-conserving children were capable of performing the empirical reversal and preference inversion problems. However, by directly linking the preference and relational questions to the presently perceived (albeit transformed) stimuli and requiring the children to address their responses accordingly, the possibility that these same children were basing their responses on knowledge that involved empirical return rather than a non-linguistic knowledge of inversion was controlled for. Although both reversal and preference were demonstrated significantly more often than knowledge of relational non-equivalence, the failure of many subjects to respond correctly on both question types mitigates any suggestion that these two processes also may be related in some way. Although both involved communication of knowledge that concerns conservation, the preference response was tied directly to an immediate perception of the stimulus array, whereas the reversal involved responding to the experimenter's questions with regard to a previously perceived or expected stimulus context. Hence, we are presented with two fundamentally different types of knowledge, both of which appear separately yet exist simultaneously in a form that appears to resist integration in children at this age level.
This evidence is consistent with the view that young children are functioning as though they had split brains and suggests that non-conserving children fail to express knowledge associated with the right hemisphere because they appear to be relying on a left hemisphere 'cognitive' strategy due to the linguistic referents traditionally involved with the task. Hence, when understood in terms of functional asymmetry and interhemispheric disconnection, the statement that children do not comprehend equivalence relationship loses its meaning.
16. SPLIT-BRAIN FUNCTIONING IN CHILDREN: THE ONTOLOGY OF EMOTIONAL CONFLICT
Thus, due in part to the slow pace of corpus callosum myelination, coupled with differential right and left cerebral specialization, the left hemisphere of a young child has at best incomplete knowledge of the contents and activity that are occurring within the right. This sets the stage for differential memory storage and a later inability to transfer this information between the cerebral hemispheres once the child reaches adulthood.
Because of lateralization and limited exchange, the effects of early "socializing" experience can have potentially profund effects. "As a good deal of this early experience is likely to have unpleasant if not traumatic moments, it is fascinating to consider the later ramifications of early emotional learning occurring in the right hemisphere unbeknownst to the left; learning and associated emotional responding that later may be completely inaccessibile to the language centers of the left half of the brain. That is, although limited transfer in children confers advantages, it also provides for the eventual development of a number of very significant psychic conflicts --many of which do not become apparent until much later in life."
Moreover, due to the immaturity of the callosum, children frequently can encounter situations in which the right and left cerebrum not only differentially perceive what is going on, but are unable to link these experiences so as to understand fully what is occurring or to correct misperceptions (Joseph, 1982, 1992b). Consider, for example, a young divorced mother with ambivalent feelings toward her young son (Joseph, 1988a, 1992b).
Although she does not express these feelings verbally, she conveys them through her tone of voice, facial expression, and in the manner in which she touches her son. She knows that she should love him, and at some level she does. She wants to be a good mother and makes herself go through the motions. However, she also resents her son because she has lost her freedom, he is a financial burden, and he may hinder her in finding a desirable mate. She is confronted by two opposing attitudes, one of which is unacceptable to the image she has of a good mother. Like many of us, she must prevent these feelings from reaching linguistic consciousness. However, this does not prevent them from being expressed via the limbic system or right hemisphere.
Her son, of course, also has a right hemisphere which perceives her tension and amibivalance. The right half of his brain notes the stiffness when his mother holds or touches him and is aware of the manner in which she sometimes looks at him. Worse, when she says, "I love you," his right hemisphere senses the tension and tone of her voice and correctly perceives that what she means is, "I don't want you" or, "I hate you. " His left hemisphere hears, however, "I love you" and notes only that she is attentive. He is in a "double bind" conflict, with no way for his two cerebral hemispheres to match impressions.
The right half of this little boy's brain feels something painful when the words "I love you" are spoken. When his mother touches him, he becomes stiff and withdrawn because his right hemisphere, via the analysis of facial expression, emotional tone, tactile sensation, etc. is fully aware that she does not want him.
Later, as an adult, this same young man has one failed relationship after another. He feels that he can't trust women, often feels rejected, and when a girl or woman says "I love you," it makes him want to cringe, run away, or strike out. As an adult, his left hemisphere hears "Love," and his right cerebrum feels pain and rejection.
Because the two halves of his cerebrum were not in communication during early childhood, his ability to gain insight into the source of his problems is greatly restricted. The left half of his brain cannot access these memories. It has "no idea" as to the cause of his conflicts.
In this regard, this asymmetrical arrangement of hemispheric function and maturation may well predispose the developing child in later life to come upon situations in which it finds itself responding emotionally, nervously, anxiously, or neurotically, without linguistic knowledge, or without even the possiblity of linguistic comprehension as to the cause, purpose, eliciting stimulus, or origin of its behavior. As a child or an adult, it may find itself faced with behavior that is mysterious, embarrassing, etc. "I don't know what came over me."
17. FUNCTIONAL COMMISSUROTOMIES AND LIMITIED INTERHEMISPHERIC TRANSFER
The human brain is organized so that two potentially independent mental systems coexist, literally side by side. Each hemisphere has, in the course of evolution, developed its own unique strategy and capability for processing information, and a neuroanatomical substrate to subserve each independent function. Therefore each hemisphere or mental system may act independently on specific information, initially without interference from the other. A result of this duality is the dual processing of sensory and cognitive information. The illusion of mental unity is made possible, in part by the corpus callosum, which both aids and inhibits interhemispheric transfer.
Not only are these systems dual, but they are asymmetrical as each hemisphere utilizes predominantly verbal-analytical or visual-spatial, sensory-affective associational strategies in the analysis and expression of information. When one hemisphere learns, thinks, or 'experiences,' the information can be transferred via the corpus callosum for further analysis or so that the other hemisphere also may 'learn,' being a beneficiary of the other's activity. Although admittedly there is considerable overlap of function, in that both hemispheres may act to analyze all input, and given that some types of information are dealt with more efficiently by one than the other, in the long run this relationship is very adaptive as the range and speed of information analysis is enlarged and the efficiency of response is increased; i.e. every individual has two brains and two minds, in the same head.
However, in that the left codes, processes, and stores information using linguistic, categorical, functional, and motoric referents, whereas the right relies on spatial, emotional, and non-linguistic modes of understanding, complete and efficient interhemispheric communication may not always be possible. Thus there is the potential for much miscommunication, misinterpretation, and distortion regardless of the direction of transfer.
The corpus callosum is the gateway via which information may travel from one brain half to the other. However, it also acts to limit information exchange even in the adult, intact, brain, since almost 40% of the adult callosum lacks myelin (Selnes, 1974). Since mylein acts to insulate and, thus, preserve information transmission by minimizing leakage and increasingly conduction velocity and integrity, some information is lost and degraded even when transfer is possible (Berlucchi & Rizzolatti, 1968; Hicks 1974; Joseph et al. 1984; Marzi, 1986; Merriam & Gardner, 1987; Myers, 1959, 1962; Rizzolatti et al. 1971; Taylor & Heilman 1980).
Moreover, particularly when one is dealing with complex or emotional information situations often arise in which one brain half has little or no knowledge as to what is occurring in the other (Joseph, 1982, 1988a, 1992b, 2010; Joseph et al. 1984). In part, this is a consequence of lateralized specialization. Certain forms of information can only be processed and recognized by the right or left half of the brain, but not both and in not the same manner. Even information that is transferred may be subject to interpretation and miss-interpretation (Joseph, 1982, 1986ab, 1988ab; Joseph et al., 1984). When information or knowledge which is stored or processed in the language of the right hemisphere is then transferred to the left, that information may not be translated correctly, and gaps in the translation may be filled with confabulatory ideation.
In addition, one brain half can be prevented from knowing what is occurring in the opposite half due to inhibitory or suppressive actions initiated by the frontal lobes, such that certain forms of information are suppressed, censored, and inter-hemispheric (as well as intra-hemispheric) information transmission prevented (Joseph, 1982, 1988a, 1992b, 2011). Even the normal intact adult brain may experience functional commissurotomies.
Therefore, these three conditions --lateralized specialization, frontal lobe inhibitory activity, and incomplete myelination of callosum axons-- can reduce the ability of the two hemispheres to communicate among normal, intact individuals. Hence, in many ways the brain of even a normal adult is functionally split and disconnected, and for good reason. These conditions protect the brain and linguistic consciousness from becoming overwhelmed. As we have seen with frontal lobe damage, when communication is allowed to occur freely (due to disinhibition) the overall integrity of the brain to function normally is curtailed dramatically.
Nevertheless, a unique side effect of having two hemispheres that are not always able to communicate completely or accurately is intra-psychic conflicts. That is, we sometimes find ourselves feeling happy, sad, depressed, angry, etc. without a clue as to the cause. In other instances, we actually may commit certain thoughtless, impulsive, overly emotional, or embarrassing actions and "have no idea" as to "what came over" us.
Moreover, unbeknownst to the left hemisphere, sometimes the right perceives, remembers, or responds to some external or internal source of experience and/or to its own memories and, thus, and reacts in an emotional manner. The left (speaking ) hemisphere in turn only knows that it is feeling something but is unsure what or why , or, conversely, confabulates various denials, rationalizations and explanations which it accepts as fact.
18. CONCLUDING COMMENTS
The two halve of the brain begin life functionally disconnected. Functional disconnection remains a fact of life, even in adulthood. This can give rise to any number of intra-psychic conflicts.
Although there is evidence of considerable functional overlap as well as interhemispheric cooperation on a number of tasks, the mental systems maintained by both the right and left cerebral hemispheres are highly developed. Whereas the consciousness of the left hemisphere is linked to language, math, temporal-sequencing, and control over the right hand, the mind of the right hemisphere is more social-emotional, bilateral, and in many ways dominant over the temporal-sequential, language-dependent half of the cerebrum. Indeed, the right cerebrum can independently recall and act on certain memories with purposeful intent; is the dominant source of our dreams, psychic conflicts and desires, and is fully capable of motivating, initiating as well as controlling behavioral expression --often without the aid or even active (reflective) participation of the left half of the brain.
Banquet,J. P. (1983).Inter- and intrahemispheric relationships of the EEG activity during sleep in man.Electroencephalography and Clinical Neurophysiology, 55, 51-59.
Berlucchi, G., & Rizolatti, G. (1968). Binocularly driven neurons in visual cortex of split-chiasm cats. Science, 159, 308-310.
Blinkov, S. M., & Glezer, I. I. (1968). The human brain in figures and tables. New York: Plenum.
Bogen, J. E. (1969). The other side of the brain. Bulletin of the Los Angeles Neurological Societies, 34, 135-162.
Bogen, J. E. (1979). The calosal syndrome. In K. M. Heilman & E. Valenstein (Eds.), Clinical neuropsychology.(pp. 308-358). New York: Oxford University Press.
Bures, J., & Buresova, O. (1960). The use of Leao's spreading depression in the study of interhemispheric transfer of memory traces. Journal of Comparative and Physiological Psychology, 59, 211-214.
Dimond, S. J. (1980). Neuropsychology. London: Butterworths.
Doty R. W., & Overman, W. H. (1977). Mnemonic role of forebrain commissures in macaques, In S. Harnad, R. W. Doty, L. Goldstein, J. Jaynes, & G. Krauthamer, (Eds.), Lateralization in the nerovus system, (pp.75-88). New York: Academic Press.
Finlayson, M. A. J. (1975). A behavioral manifestation of the development of interhemispheric transfer of learning in children. Cortex, 12, 290-295.
Flechsig, de P. (1901). Developmental (mylogenetic) localisation of the cerebral cortex in the human subject. Lancet, 2, 1027-1031.
Galin, D,. Diamond, R., &. Herron, J.(1979). Development of crossed and uncrossed tactile localization on the fingers. Brain and Language, 4, 588-590.
Galin, D., Johnstone, J., Nakell, L., & Herron, J. (1979). Development of the capacity for tactile information transfer between hemipsheres in normal children. Science, 204, 1330-1332.
Gallagher, R. E., & Joseph, R. (1982). Non-linguistic knowledge, hemispheric laterality, and the conservaton of inequivalence. Journal of General Psychology, 107, 31-40.
Gazzaniga, M. S., & LeDoux, J. E. (1978). The integrated mind. New York: Plenum Press.
Geschwind, N. (1965). Disconnexion syndromes in animals and man. Brain, 88, 585-644.
Geschwind, N. (1981). The perverseness of the right hemisphere. Behavioral and Brain Sciences, 4, 106-107.
Goldstein, L., Stoltzfus, N. W., & Gardocki, J. F. (1972). Changes in interhemispheric amplitude relationships in the EEG during sleep. Physiology and Behavior, 8, 811-815.
Hasegawa, I., Fukushima, T., Ihara, T., & Miyahsita, Y. (1998). Callosal window between prefrontal cortices: Cognitive interaction to retrieve long-term memory. Science, 281, 814-818.
Hicks, R. E. (1974). Asymmetry of bilateral transfer. American Journal of Psychology, 87, 667-674.
Hodoba, D. (1986). Paradoxic sleep facilitation by interictal epileptic activity of right temporal origin. Biological Psychiatry, 21, 1267-1278.
Joseph, R. (1979). Effects of rearing and sex differences on learning and competitive exploration. Journal of Psychology, 101, 37-43.
Joseph, R. (1982). The Neuropsychology of Development: Hemispheric Laterality, Limbic Language, and the Origin of Thought. Journal of Clinical Psychology, 44, 3-34.
Joseph, R. (1986a). Confabulation and delusional denial: Frontal lobe and lateralized influences. Journal of Clinical Psychology, 42, 845-860.
Joseph, R. (1986b). Reversal of cerebral dominance for language and emotion in a corpus callosotomy patient. Journal of Neurology, Neurosurgery, and Psychiatry, 49, 628-634.
Joseph, R. (1988a) The Right Cerebral Hemisphere: Emotion, Music, Visual-Spatial Skills, Body Image, Dreams, and Awareness. Journal of Clinical Psychology, 44, 630-673.
Joseph, R. (1988b). Dual mental functioning in a split-brain patient. Journal of Clinical Psychology, 44, 770-779.
Joseph, R. (1990). Neuropsychology, Neuropsychiatry, Behavioral Neurology, Plenum, New York.
Joseph, R. (1992a) The Limbic System: Emotion, Laterality, and Unconscious Mind. The Psychoanalytic Review, 79, 405-456.
Joseph, R. (1992b). The Right Brain and the Unconscious. New York, Plenum.
Joseph, R. (1994) The limbic system and the foundations of emotional experience. In V. S. Ramachandran (Ed). Encyclopedia of Human Behavior. San Diego: Academic Press.
Joseph, R. (1996). Neuropsychiatry, Neuropsychology, Clinical Neuroscience, 2nd Edition. Williams & Wilkins, Baltimore.
Joseph, R. (1998). Traumatic amnesia, repression, and hippocampal injury due to corticosteroid and enkephalin secretion. Child Psychiatry and Human Development. 29, 169-186.
Joseph, R. (1999b). Environmental influences on neural plasticity, the limbic system, and emotional development and attachment, Child Psychiatry and Human Development. 29, 187-203.
Joseph, R. (1999a). The neurology of traumatic "dissociative" amnesia. Commentary and literature review. Child Abuse & Neglect. 23, 715-727.
Joseph, R. (1999c). Frontal lobe psychopathology: Mania, depression, aphasia, confabulation, catatonia, perseveration, obsessive compulsions, schizophrenia. journal of Psychiatry, 62, 138-172.
Joseph, R. (2000). Limbic language/language axis theory of speech. Behavioral and Brain Sciences. 23, 439-441.
Joseph, R. (2000). Fetal brain behavioral cognitive development. Developmental Review, 20, 81-98.
Joseph, R. (2000a). The evolution of sex differences in language, sexuality, and visual spatial skills. Archives of Sexual Behavior, 29, 35-66.
Joseph, R. (2003). Emotional Trauma and Childhood Amnesia. journal of Consciousness & Emotion, 4, 151-178.
Joseph, R. (2009). Quantum Physics and the Multiplicity of Mind: Split-Brains, Fragmented Minds, Dissociation, Quantum Consciousness. Journal of Cosmology, 2009, 3, 600-640.
Joseph, R. (2011). Origins of Thought: Consciousness, Language, Egocentric Speech and the Multiplicity of Mind. Journal of Cosmology, 14. 4577- 4600.
Konner, M. (1991). Universals of behavioral development in relation to brain myelination. In K. R. Gibson & A. C. Petersen (Eds). Brain maturation and cognitive development. New York, Aldine De Gruyter.
Kraft, R. H., Mitchell, O. R., Languis, M. L., & Wheatley, G. H. (1980). Hemispheric asymmetries during six-to-eight year-olds' performance on Piagetian conservation and reading tasks. Neuropsychologia, 18, 637-644.
Kurcharski, D., & Hall, W. G. (1987). New routes to early memories. Science, 238, 786-788.
LeCours, A. R. (1975). Myelogenetic correlates of the development of speech and language. In E. Lenneberg & E. Lenneberg (Eds.), Foundations of language development. New York: Academic Press.
Levy, J. (1974). Psychological implications of bilateral asymmetry. In S. Dimond & J. G. Beaumont (Eds.), Hemisphere function in the human brain, (pp.121-183). London: Paul Elek.
Levy, J. (1983). Language, cognition and the right hemisphere. American Psychologist, 38, 538-541.
Marzi, C. A. (1986). Transfer of visual information after unilateral input to the brain. Brain and Cognition, 5, 163-173.
Merriam, A. E., & Gardner, E. B. (1987). Corpus callosum function in schizophrenia: A neuropsychological assessment of interhemispheric information processing. Neuropsychologia, 25, 185-193.
Meyer, J. S., Ishikawa, Y., Hata, T., & Karacan, I. (1987). Cerebral blood flow in normal and abnormal sleep and dreaming. Brain and Cognition, 6, 266-294.
Myers, R. E. (1959). Interhemispheric communication through the corpus callosum: Limitations under conditions of conflict. Journal of Comparative and Physiological Psychology, 52, 6-9.
Myers, R. E., (1962). Transmission of visual information within and between the hemispheres: A behavioral study. In V. B, Mountcastle (Ed). Interhemispheric Relations and Cerebral Dominance. Baltimore, Johns Hopkins Press.
O'Leary, D. S. (1980). A developmental study of interhemispheric transfer in children aged 5 to 10. Child Development, 51, 743-750.
Piaget, J. (1952). The origins of intelligence in children. New York: Norton.
Piaget, J. (1962). Play, dreams and imitation in childhood. New York: Norton.
Piaget, J. (1974). The child and reality. New York: Viking Press.
Risse, G. L., & Gazzaniga, M. S. (1979). Well-kept secrets of the right hemisphere: A carotid amytal study of restricted memory transfer. Neurology, 28, 950-953.
Ritchie, J. M. (1984). Pathophysiology of conduction in demyelinated nerve fibers. In P., Morell (Ed.), Myelin, 2nd Ed. New York, Plenum.
Rizzolatti, G., Umilta, C., & Berlucchi, G. (1971). Opposite superiorities of the right and left cerebral hemispheres in discriminative reaction time to physiognomical and alphabetical material. Brain, 94, 431-442.
Salamy, A. (1978). Commissural transmission: Maturational changes in humans. Science, 200, 1409-1411.
Selnes, O. A. (1974). The corpus callosum: Some anatomical and functional considerations with special reference to language. Brain and Language, 1, 111-139.
Seymour, S. E., Reuter-Lorenz, P. A., & Gazzaniga, M. S. (1994). The disconnection syndrome. Brain, 117, 105-115.
Sperry, R. (1966). Brain bisection and the neurology of consciousness. In J. C. Eccles (Ed). Brain and conscious experience, ( pp 298-313). New York: Springer Verlag.
Sperry, R. W. (1968). Hemisphere dissconnection and unity in conscious awareness. American Psychologist. 23, 723-733.
Sperry. R. (1974). Lateral specialization in the surgically separated hemispheres. In F. O. Schmitt & F. G. Worlden (Eds.), The neurosciences: Third study program, (pp 1-12). Cambridge. MIT Press.
Sperry, R. (1982). Some effects of disconnecting the cerebral hemispheres. Science, 217, 1223-1226.
Sperry, R. W., Zaidel, E., & Zaidel, D. (1979). Self recognition and social awareness in the deconnected minor hemisphere. Neuropsychologia, 17, 153-166.
Sweet, W. H. (1945). Intracranial aneurysm simulating neoplasm. Syndrome of the corpus callosum . Archives of Neurology and Psychiatry, 45, 86-103.
Taylor, G. H. & Heilman, K. (1980). Left-hemispheric motor dominance in right handers. Cortex, 16, 587-603.
Wolpert E. A., & Trosman, H. (1958). Studies in psychophysiology of dreams I. Experimental evocation of sequential dream episodes. Archives of Neurology, 79, 603-606.
Yakovlev, P. I., & Lecours, A. (1967). The myelogenetic cycles of regional maturation of the brain,. In A. Minkowski (Ed.), Regional development of the brain in early life, (pp.404-491). London: Blackwell.












