Rhawn Gabriel Joseph, Ph.D.
Brain Research Laboratory
BrainMind.com
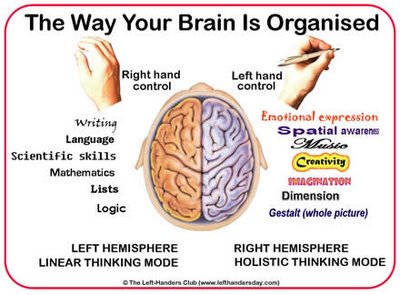

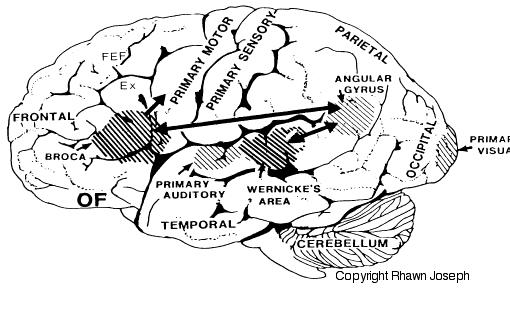
Among 80-90% of right handers, and over 50-80% of those who are left handed, the left cerebral hemisphere provides the neural foundation and mediates most aspects of expressive and receptive linguistic functioning (Frost et al., 2009; Pujol, et al., 2009). The left half of the brain is dominant for the perception and production of almost all aspects of non-emotional language (Albert, 1993; Barr et al. 1990; Benson, 1993; DeRenzi et al. 2007; Evers et al., 2009; Frost, et al., 2009; Goodglass & Kaplan,2009; Heiss, et al., 2009; Kapura et al. 1992; Kertesz, 2005ab; Kimura, 1993; Levin, 1993; Pujol, et al., 2009), including reading, writing, speaking, spelling, naming, and the comprehension of the grammatical, syntactical, and descriptive components of language, including time sense, rhythm, musical scales, verbal concept formation, analytical reasoning, and verbal memory (Albert et al.1972; Albert, 1993; Barr et al. 1990; Benson, 1993; Frisk & Milner 1990; Goodglass & Kaplan, 2000; Hecaen & Albert, 1983; Kapura et al. 1992; Kertesz, 2005ab; Kimura, 1993; Levin, 1993; Levine & Sweet, 2005; Luria, 1980; Milner, 1970; Parsons & Fox, 2007; Price, 2007; Vignolo, 2005).
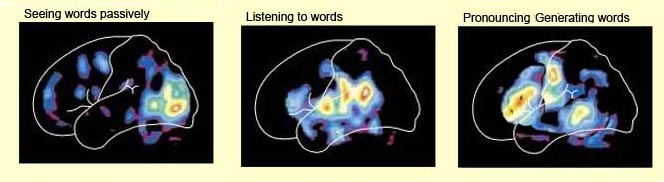
As demonstrated with functional imaging and blood flow studies, when reading, speaking, and naming, the left hemisphere becomes highly active (Buchel et al., 1998; Evers et al., 2009; Frost et al., 2009; Peterson et al., 1988, 1990; Price, 2007; Pujol, et al., 2009). Likewise, when playing musical scales, or listening to the rhythmical aspects of music, the left temporal lobe increases in activity (Evers et al., 2009; Parsons & Fox, 2007).
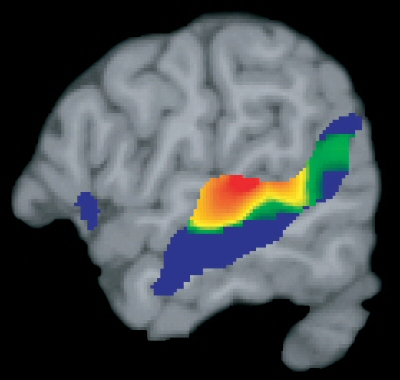
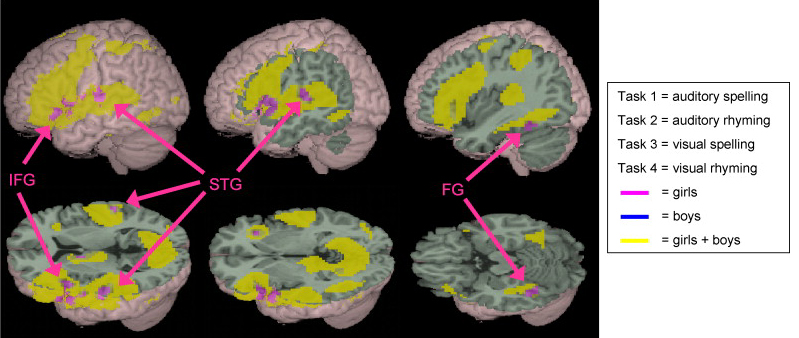
As demonstrated through lesion studies, functional imaging, and dichotic listening, the human left hemisphere adominates in the perception and processing of real words, word lists, rhymes, numbers, backwards speech, morse code, consonants, consonant vowell syllables, nonsense syllables, the transitional elements of speech, and single phonemes (Blumstein & Cooper, 1974; Cutting, 1974; Frost et al., 2009; Haglund et al. 1993; Kimura, 1961, 1993; Kimura & Folb, 1968; Levy, 1974; Mills & Rollman, 2012; Papcunn et al. 1974; Price, 2007; Pujol et al., 2009; Shankweiler & Studert-Kennedy, 1966, 1967; Studdert-Kennedy & Shankweiler, 1970). It is also dominant for recognizing phonetic, conceptual, and verbal (but not physical) similarities; for example, determining if two letters (g & p vs g & q) have the same vowel ending (Cohen, 1972; Levy, 1974; Moscovitch, 1973).
In this regard, it could be argued that because the lexical, syntactic, grammatical, and denotative aspects of language are localized to the left hemisphere, those aspects of consciousness which are dependent on language are also associated with the left hemisphere; a position advocated by a number of independent neuroscientists (e.g. Albert et al. 1976; Bogen 1969; Dixon 1981; Galin 1974; Hoppe 1977; Ornstein 1972; Miller 2001; Popper & Eccles 1977; see chapter 2).
Language and consciousness are tightly related, such that the verbal-dependent, linguistic-aspects of consciousness are clearly associated with the left hemisphere in the majority of the population.



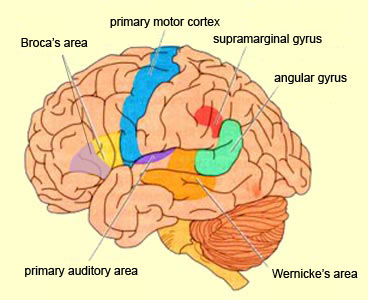
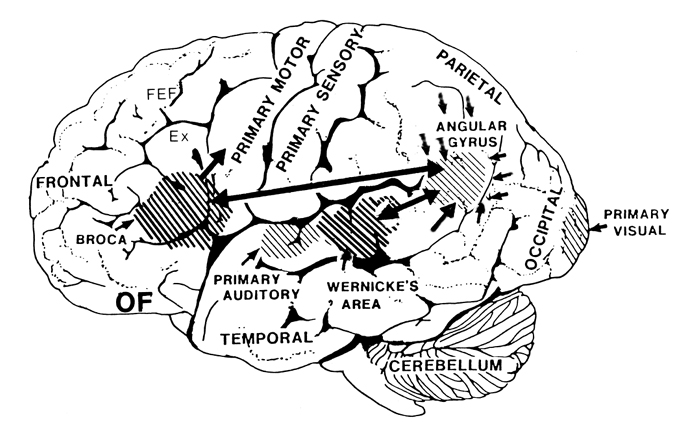
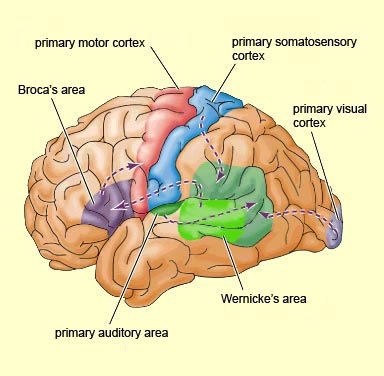
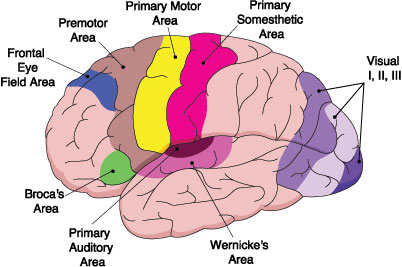
In 1861 and again in 1964, Paul Broca localized the loss of the "speech faculty" to the left frontal operculum; an area that is now referred to as Broca's expressive speech area. Ten years later, Wernicke (1874) demonstrated that a lesion to the posterior portion of the superior temporal lobe resulted in memory loss for the "auditory images" of words; a region now referred to as Wernicke's area.

Over the last hundred years, there has been a general consensus that these two regions, Wernicke's and Broca's areas serve as the primary centers for the reception and production of language. However, this position has also had its detractors who have ridiculed the "diagram makers" whereas others have claimed there is only one aphasia: Wernicke's aphasia.
Freud (1891), for example, argued against the localizationist position, but in so doing, introduced the concept of disconnection syndromes. Freud (1891) detailed that lesions to regions surrounding these language zones also gave rise to aphasic symptoms, and that damage which disconnected the parietal-occipital lobe from Wernicke's area, gave rise to "agnosia."
"Gnosis" means to "know" Agnosia means, not to know. Hence, with a lesion in the parietal-occipital area, visual information can not be matched with auditory information. Therefore, a person can't name what they see!
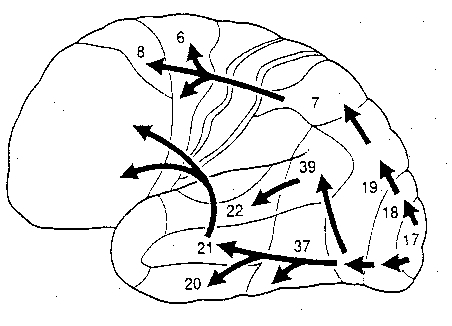

In a monograph which is now considered a classic, Geschwind (1965) reviewed and synthesized these theories and reintroduced Freud's concept of disconnection. Specifically, Geschwind (1965) showed how lesions to various regions of the brain can prevent the language areas from gaining access to necessary information, thus resulting in naming, reading, and related language disorders due to disconnection; i.e. disconnection syndromes.
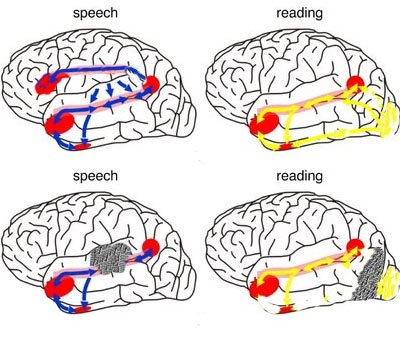
In a monograph that was subsequently translated and republished by numerous foreign journals (e.g. Joseph, 1984) and numerous American medical schools, and which resulted in invitations to speak world-wide, Joseph (1982) synthesized the works of Broca, Wernicke, Freud, Geschwind, and others, and introduced the concept of the "language axis." He also detailed the role of the limbic system and right frontal and temporal lobe in the melodic emotional aspects of speech, and the development of language. He also detailed the importance of convergence zones and the pivotal role of the inferior parietal lobe in assimilating and naming these associations, and was the first to explain how these areas of the brain fills the gaps in those associations received from other areas of the brain, thus producing vocabulary-rich grammatical human speech. These theories are now supported by an incredible body of evidence, and have been widely accepted, and numerous authors now claim these ideas as their own.
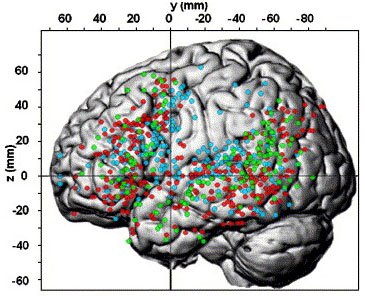
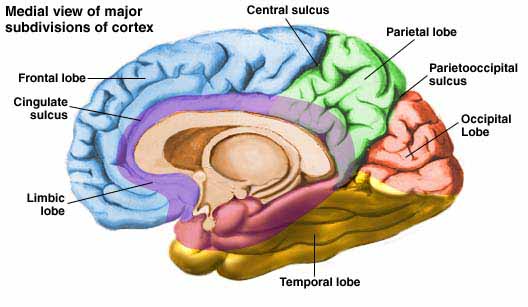
Language areas of the left hemisphere becoming activated during language tasks
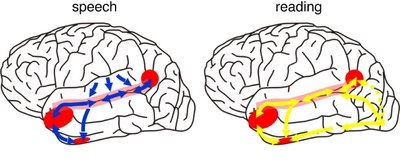
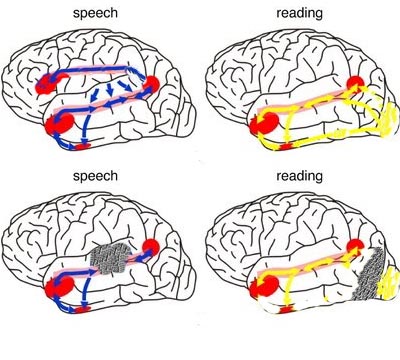
As detailed (Joseph, 1982, 1986a, 1988a; Joseph et al., 1984), language processing is sequential, serial, and parallel, involving the activation of widespread areas of both the right and left hemisphere and specific cortices in the left half of the cerebrum.
Moreover, because language is also melodic and emotional, language also requires activation of the right frontal and right temporal lobe (Joseph, 1982)--findings which have also been confirmed by functional imaging (e.g., Bottini et al., 1994; Price et al., 1996).
That is, when engaged in language and other cognitive tasks, the brain functions in both a parallel and localistic mode and thus engages in parallel/distributed as well as localized representation and processing. This involves the parallel and localized activation of Wernicke's and Broca's areas, and the inferior parietal lobule (IPL), with the IPL serving as the ultimate convergence zone.
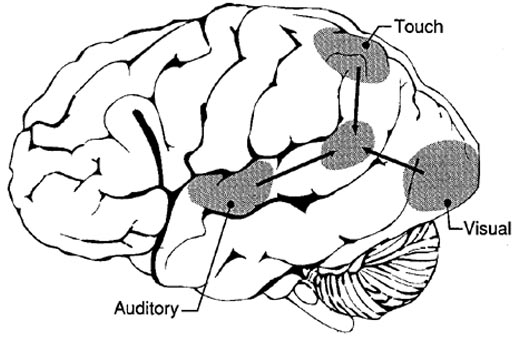
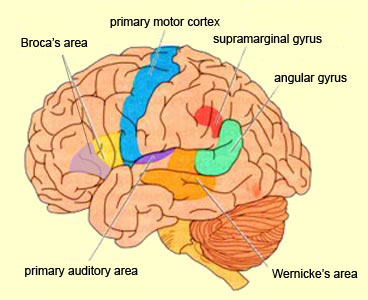
The IPL (which includes the angular and supramarginal gyrus) assimilates associations received from yet other areas of the left and right hemisphere (including the amygdala and cingulate gyrus), fills any gaps with relevant associations, and then injects the resulting verbal associations into the stream of language and thought via the arcuate and longitudinal fasciculus which interlinks the language areas. Hence, the concept of a "language axis" (Joseph, 1982, 2009e,f).
This parallel and localizationist convergence model of language is based on lesion studies and human brain dissection, has been confirmed by functional imaging studies, and has recently been adapted by other theorists (reviewed in Joseph, 2009f). For example, as based on functional imaging studies, it has been demonstrated that speech processing, reading, subvocal vocalization, and innerspeech activates the left frontal lobe (Buchel et al., 1998; Demonet, et al., 1994; Paulesu, et al., 1993; Peterson et al., 1988). And, left frontal activation increases as word length increases, and in response to unfamiliar words (Price, 2007).
In addition, reading and speaking activates the Wernicke's area and the left posterior temporal lobe (Bookheimer, et al., 1995; Howard et al., 1996), including the supramarginal gyrus (Bookheimer, et al., 1995), angular gyrus of (Price, 2007) and (when reading) the left medial extrastriate visual cortex (Peterson et al., 1990).
Moreover, when making semantic decisions (involving reading words with similar meanings), there is increased activity in the left posterior temporal lobe at the junction of the IPL (Price, 2007).
Moreover, like the left frontal lobes, the left temporal areas are activated during word generation (Shaywitz, et al., 1995; Warburton, et al., 1996), and sentence comprehension tasks (Bottini, et al., 1994; Fletcher et al., 1995), whereas the IPL in general appears to serve as a phonological storehouse that becomes activated while reading, listening, and when engaged in a variety of languages tasks including word retrieval (Bookheimer, et al., 1995; Demonet, et al., 1994; Menard, et al., 1996; Paulesu, et al., 1993; Price, 2007; Vandenberghe, et al., 1996). Moreover, activation in the IPL as well as the left frontal and left temporal lobe increases as word length increases and for long and umfamiliar words (Price, 2007).

HANDEDNESS, MOTOR CONTROL, AND LANGUAGE
Among almost 90% of the population the left hemisphere is also dominant for fine motor functioning including handedness (reviewed in Corballis, 2001).
It appears that language and motor functioning are both lateralized to the left hemisphere because modern human language is in large part an evolutionary outgrowth of and is directly related to neuroplastic changes induced in the left hemisphere in response to tool making and gathering activities by men and women over the course of human evolution.
When creating a stone tool, males generally use the right hand to manufacture and shape the tool, and the left hand to hold it.
Likewise, female tool-makers, and tool users, and female gatherers, also predominantly utilized the right hand for rapid temporal sequential movements, including very fine motor activity. Therefore, over the course of evolution, the right hand / left hemisphere also became adapted for shaping language, giving it a temporal sequential stamp.




That is, the left hemisphere became reorganized in reaction to the acquisition of handedness and those activities involving fine motor functioning, such that what had been emotional "limbic language" became subject to fine motor control and temporal sequencing thus producing grammatical speech.
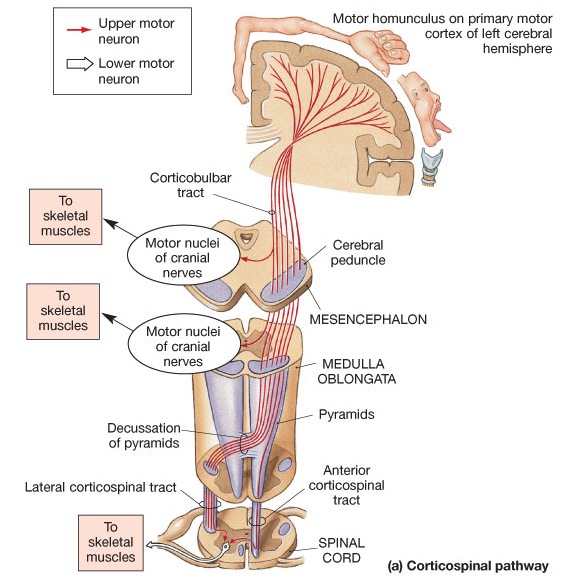
The left hemisphere became selectively reorganized due to its dominance for hand control. This dominance is due to the earlier maturation of the left cerebral corticospinal pyramidal tract (Joseph 1982) which descends from the neocortical motor areas into the brainstem, and then crosses at the pyramidal decussation (Kertesz & Geschwind 1971; Yakovlev & Rakic 1966), and then descends the spinal cord in advance of those fibers from the right. That is, as the neonate and the infant brain develops, the pyramidal neurons in the motor areas of the left hemisphere mature more rapidly than those neurons in the right hemisphere. Those from the left motor areas grow and establish their connections with the motor centers, giving them dominance as they take up more synaptic space.
The left hemisphere, therefore is provided a competitive advantage over the right in motor control, such that in response to tool making and gathering activities by men and women over the course of human evolution, this half of the brain became specialized for imposing temporal sequences on both external and internal stimuli (Chapters 6,7).
By contrast, among non-humans, there is little or no evidence that either hemisphere dominates in regard to "language," fine motor control, or handedness (see edited volume by Ward & Hopkins 1993). Indeed, 50% of primates and mammals tend to favor the right hand (paw, leg) and 50% tend to utilize the left extremity (however see MacNeilage 1993). Presumably the right and left corticospinal (pyramidal) tracts grow and mature at the same rate in these species.


Moreover, in contrast to humans, the tool making capabilities of primates is limited to using twigs, sticks, branches, leaves and rocks, such that they are basically devoid of temporal sequential fine motor manipulative capabilities which are important in human language. Thus, in contrast to the grammatically complex temporal sequences employed in human speech, non-human animals do not employ sound units in order to communicate, and the role of the neocortex is relative minimal in this regard (Jurgens et al. 1982; Myers 1976). Rather, vocalization is largely a function of the limbic system, and is thus referred to as "limbic language" (Joseph 1982, 1992a, 2009f; Jurgens 1990).
However, there are trends among non-human primates toward some degree of reliance on neocortical tissue and the superior temporal lobe in regard to the comprehension of species specific sounds. Ninety percent of primate auditory cortex neurons are activated by species-specific calls (Newman & Wollberg 1973), whereas destruction of the left superior temporal lobe disrupts that ability to make sound discriminations (Heffner & Heffner 1984; Hupfer et al. 1977; Schwarz & Tomlinson 1990). There is also some evidence to suggest that asymmetries in the planum temporal are apparent even in chimpanzees (Gannon 1998), and that in other hominoids and monkeys, the left hemisphere is dominant for the perception of primate vocalizations (Hauser & Anderson 1994; Peterson & Jusczyk 1984; Peterson et al. 2003).
Presumably these trends became exaggerated over the course of evolution, such that with the acquisition of handedness and tool making capabilities, left hemisphere dominance for vocalization, perception, and expression gradually increased in the transition from Australopithecus, to H. habilis, to H. erectus, to Neanderthals to Cro-Magon. In consequences, humans began speaking in sequentially ordered sound units (detailed in chapter 6).
Linguistic and motoric temporal sequencing is an exceedingly important capability which appears to be unique to humans. For example, disturbances of linguistic comprehension (i.e. receptive aphasia) are frequently a consequence of an impaired capacity to discern the individual units of speech and their temporal order. The sounds of speech must be separated into discrete interrelated linear units or they will be perceived as a blur, or even as a foreign language. Hence, a patient with Wernicke's aphasia may perceive a spoken sentence such as the "big black dog" as the "blickbakgod."
In fact, the superiority of the left hemisphere regarding temporal sequencing includes even the visual domain for the left is superior to the right in visual sequential memory and in detecting nonverbal sequences (Halperin, et al,. 1973; Zaidel, 1975). These capabilities in turn appear to be dependent on the IPL and the evolution of left hemispheric control over fine motor functioning. The parietal lobe is considered a "lobe of the hand."
The association between handedness, language and motor functioning has in fact, been noted by numerous investigators, such that there is now a convergence of opinion that the neural substrate and the evolution of language and linguistic thought are related to and are outgrowth of right hand and left hemisphere temporal-sequential motor activity (Bradshaw and Nettleton, 1982; Corballis, 2001; Corballis & Morgan, 2003; Faaborg-Anderson, 1957; Hewes, 1973; Jacobson, 1932; Joseph, 1982, 1993, 2009e; Kimura, 1973, 1976. 2012, 1980, 1993; MacNeilage, 1993; MacNeilage et al. 2007; McGuigan, 2003; Morgan & Corballis, 2003). This linkage accounts for why individuals often gesture with the right hand when they speak and why Broca's aphasia is almost invariably accompanied by paralysis of the right upper extremity.




Moreover, simultaneous activation of the right hand or Broca's area can result in spreading excitation such that both neocortical areas become excited simultaneously (see below); which in turn can result in mutual interference (Kimura & Archibald, 1974; Kinsbourne & Cook, 1971). That is, speaking and engaging in a right handed motor activity simultaneously disrupts both. In fact, immediately adjacent to Broca's expressive speech is a huge expanse of neocortical tissue representing the hand (see chapter 19), and the hand and mouth areas are also richly interconnected.
Indeed, among the majority of the population it is the right hand which is dominant for grasping, manipulating, exploring, writing, creating, destroying, and communicating. That is, although the left hand assists, it is usually the right which is more frequently utilized for orienting, pointing, gesturing, expressing and gathering information concerning the environment. We predominantly use the right for waving goodbye, throwing a kiss, delivering a vulgar gesture, greeting, etc. The right hand appears to serve as a kind of motoric extension of language and thought in that it acts at the behest of lingustic impulses via parietal lobe programming. As detailed in chapter 20, the parietal lobe is considered a "lobe of the hand."


However, the inferior parietal lobule (the angular and supramarginal gyrus) is not just a "hand" area but a multi-modal language area which acts to sequence language as well as inject words and categories into the stream of language and thought (Joseph, 1982). The role of the IPL in language is not only evident as based on lesion studies (as will be discussed) but functional imaging. For example, as based on functional imaging, it appears that the supramarginal gyrus may act as a phonological storehouse that becomes activated during short-term memory and word retrieval (Demonet, et al., 1994; Paulesu, et al., 1993; Price, 2007); whereas conversely, deficits in phonological processing are the most common correlate of reading disability (Brady & Shankweiler, 2001). Simply looking at words will activate the left supramarginal gyrus (Bookheimer, et al., 1995; Vandenberghe, et al., 1996; Menard, et al., 1996; Price, 2007) which also becomes active when performing syllable judgements (Price, 2007), and when reading (Bookheimer, et al., 1995; Menard, et al., 1996; Price, et al., 1996). Likewise, the IPL become highly active when retrieving the meaning of words during semantic processing and semantic decision tasks and activation increases as word length increases (Price, 2007).
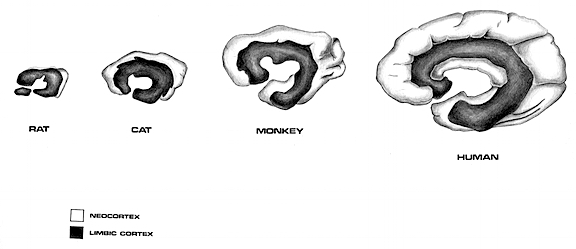
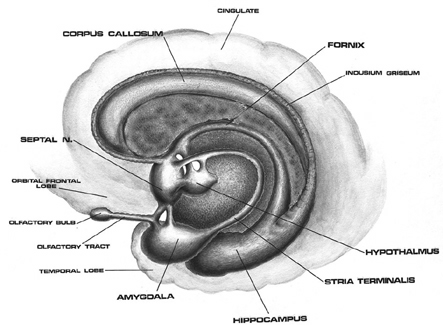
As detailed below and in chapters 6, with the evolution of the IPL, what had been limbic language became yoked to the neocortex. Briefly, the lateral frontal convexity, including Broca's area, may have evolved from the supplementary motor areas and medial frontal lobe which in turn evolved from and is richly interconnected with the anterior cingulate (e.g. Sanides 1964). The amygdala (and hippocampus) gave rise to the medial and inferior temporal lobes, the insula (see Sanides 1964), followed by the superior temporal lobe, Wernickes area, and by extention, portions of the inferior parietal lobule. The IPL, however, is also an evolutionary derivative of superior parietal tissue which expanded in accordance with and as represented by temporal sequential hand use and fine motor function involving the fingers and the thumb.
The evolution of the IPL (that is, the angular gyrus), therefore, may have served as a nexus, interlocking, at a neocortical level, the cingulate-Broca pathways, and the amygdala-Wernicke's pathway, thereby enabling limbic language impulses to become hierarchically represented as well as subject to temporal sequencing by neocortical neurons (Joseph 1993, 2009e,f). Prior to the evolution of the IPL/angular gyrus, Broca's area presumably was unable to receive sufficient input from primary auditory receiving and Wernicke's areas (and the amygdala), and language thus remained by and large, limbic and controlled by the anterior cingulate gyrus.
The impetus for inferior parietal and frontal lobe and Broca's area evolutionary development, however, appears to be two-fold, being in part limbic derivatives (amygdala-hippocampus, amygdala-cingulate) and a function of the evolution of fine motor control involving the facial-oral musculature, vocalization, and especially the establishment of handedness. Given that the human left corticospinal tract matures earlier and crosses the medullary pyramids at an earlier age than fibers from the right (Kertesz & Geschwind 1971; Yakovlev & Rakic 1966), thereby presumably establishing synaptic control over the spinal and cranial motor nuclei in advance of the right as well, dominance for hand control and temporal sequential processing became the province of the left hemisphere (Joseph 1982). With motor dominance, the left amygdala, cingulate gyrus, superior temporal lobe, inferior parietal and frontal were reorganized accordingly.


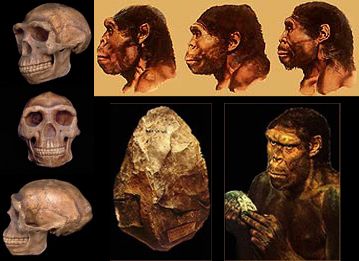
H. Erectus
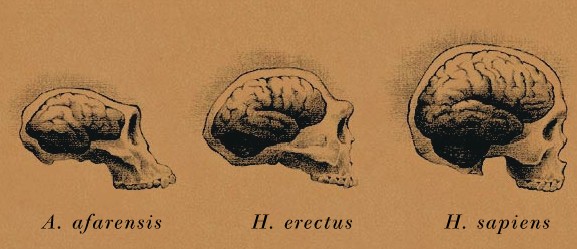
There is no evidence, however, to suggest that by 150,000 years ago modern human language had appeared, for the neural substrate that would provide the motoric foundations for grammatical speech; i.e. the angular gyrus of the inferior parietal lobule, had probably not yet evolved (chapter 6). Moreover, until as recently as 100,000 years ago, with the exception of motor control, the left and right hemisphere were probably more alike than disimilar in functional specialization -at least in regard to language. Although the Neanderthal genome possessed the FOXP2 "language gene" (as does reptiles and other animals), the life style of this archaic humans was not conducive to the development of complex human language.

A Neanderthal (top) vs a CroMagnon
The neocortex of the left hemisphere probably had not yet become as specialized or organized for expressing or comprehending modern human speech until around the late Middle Paleolithic.
This process of acquiring modern human speech and the accompanying alterations in the neural architecture of the left hemisphere may have began in earnest around 100,000 years ago, and then possibly underwent a rapid acceleration 50,000 years ago (with the evolution of the angular gyrus), followed by yet another possible rapid progression during the last 10,000 years with the invention of reading, writing, and math (see chapter 6). Human beings now had the ability to not only speak and think in words but to write them down.
Presumably, because of the increase in left hemisphere dominance over handedness and fine motor control, followed by language representation, former functions were displaced or minimized in importance and/or became the sole domain of the right hemisphere (chapter 6). That is, older functions were crowded out first by the development of fine motor control, and then again with the acquisition of language. The left and right hemisphere therefore became increasingly dissimilar in functional organization.
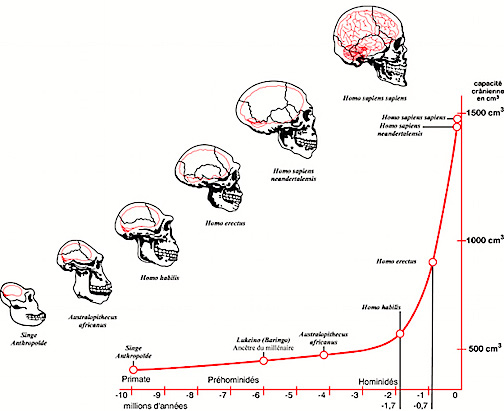
As per evolutionary changes in human neural motor organization, it appears that approximately 5 million years ago our pre-human ancestors had become increasingly adapted to walking, and running on two legs rather than on all four as do apes and monkeys. Thus by 3.5 million years ago Australopithecus and Homo hablis had become bipedal creatures and had learned to stand on their hind legs and to walk in an upright manner (Johanson & White 2012; Leakey & Hay, 2012). In consequence, the arms and hands ceased to be used as feet and were freed of the necessity of holding or hanging on to a tree branch for postural support and/or in order to move about. They ceased to be weight bearers. Subsequently the hands were also increasingly utilized to explore and manipulate objects, and to make simple stone tools.
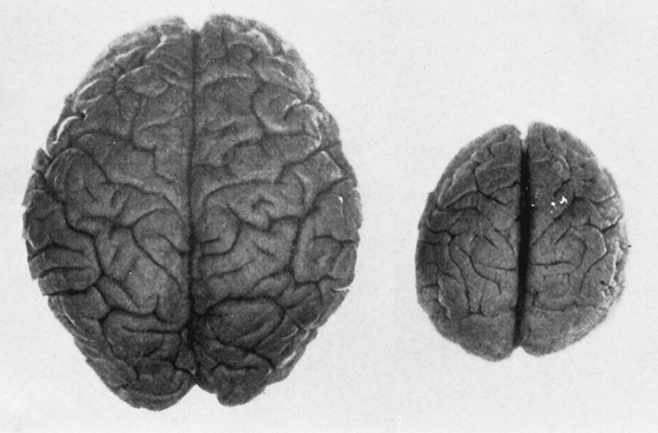
Brain of a Human vs Ape
However, once the feet had become modified for walking, they ceased to be employed for more complex acts such as grasping, and those areas of the neocortex devoted to foot control and sensory reception diminished in size (Richards, 1986; Falk, 1990). Correspondingly, neocortical areas subserving the fingers and hand, increased in size and sensory-motor importance. In fact more somatomotor neocortical space is devoted to representing the fingers and the human hand (in conjunction with the face and mouth) than the elbow, wrist, or forearm (see chapters 19, 20).
As detailed in chapter 18 the brain is exceedingly plastic and is capable of undergoing tremendous functional reorganization not just over the course of evolution, but within a few months (or years) of a single individuals lifetime (Feldman et al. 1992; Juliono et al. 1994; Strauss et al. 1992; Weiller et al. 1993). However, if the forearm is repeatedly stimulated, correspondingly increases in forearm representation are noted in the parietal lobe. Similarly, if a single digit is amputated, the remaining fingers will increase their neocortical representation and will take over the temporarily vacated space (Juliano et al. 1994).
Gradually, over the course of human evolution, the importance of the hands increased as did their neocortical representation. Subsequently, tool technologies became more complex as did those areas of the brain devoted to hand control; i.e. the inferior parietal lobule and frontal motor areas (chapters 6,7). However, because the motor neocortex of the human left hemisphere matures before the right (Corballis & Morgan, 2003, Joseph, 1982; Morgan & Corballis, 2003), and as the left frontal-parietal pyramidal tract develops and establishes its spinal-motor interconnections in advance of the right hemisphere (Kertesz & Geschwind, 1971; Yakovlev & Rakic, 1966), the left hemisphere is (and was) given a competitive advantage in motor control, and thus right hand motor dominance which in turn led to the establishment of the grammatical and temporal sequential foundations of modern human language.
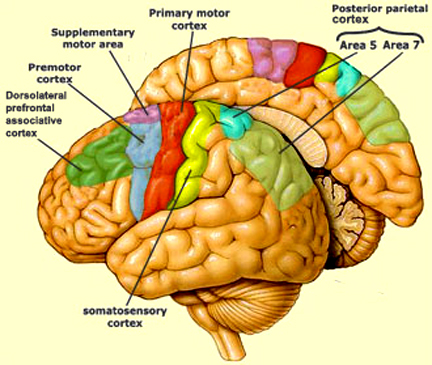
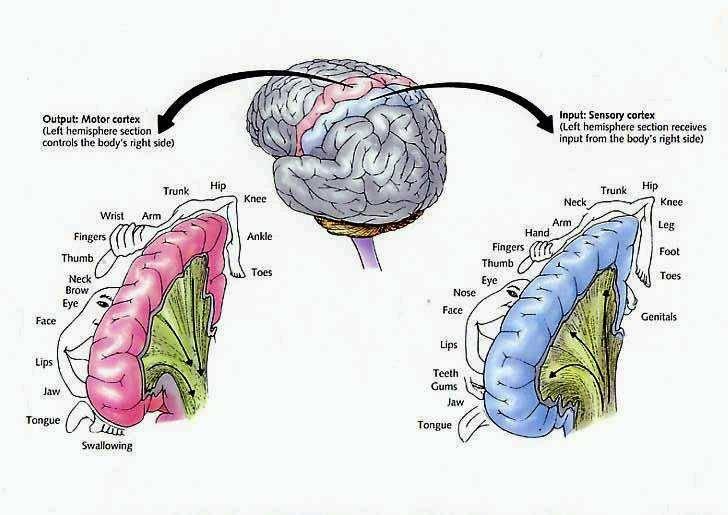
It is in part due to the interrelationship between sensory feedback, motor control and gesture, that hand control and gesture are greatly dependent on the parietal area, the left IPL in particular.
In addition, the motor engrams that make possible temporal and sequential motor acts (e.g. making a cup of coffee, fashioning a tool) appear to be localized within the inferior parietal lobe (Heilman et al. 1982; Kimura, 1993; Strub & Geschwind, 2005). It is the IPL which enables humans to engage in complex activities involving a series of related steps, create and utilize tools, produce and comprehend complex gestures, such as American Sign Language (ASL), and express and perceive grammatical relationships (Joseph, 1993, Kimura, 1993; see also Corina et al. 1992). Hence, when the left IPL has been injured patients may be afflicted with apraxia (Heilman et al. 1982) and have difficulty with tasks requiring complex motor sequencing.
Broca and Wernicke's areas and thus left cerebral linguistic functioning are exceedingly dependent on the IPL and it's capacity to impose rhythmic temporal sequences on auditory associations and motoric actions (Geschwind, 1966; Goodglass & Kaplan, 1982; Joseph, 1993, 2009e; Heilman et al. 1982; Kimura, 1993; Strub & Geschwind, 2005), including vocalizations which arise from the limbic system.
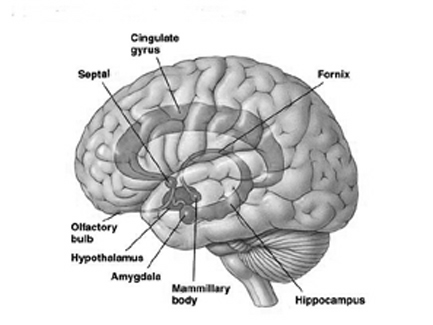
Presumably when the inferior parietal lobule and the angular gyrus fully evolved, humans acquired the capacity to segment incoming sounds and to hierarchically represent and punctuate social-emotional, limbic vocalizations so as to vocally express themselves in temporal and grammatical sequences. Thus social-emotional vocalizations came to be governed by grammatical rules of organization, thus producing "modern" human language.
MULTI-MODAL PROPERTIES
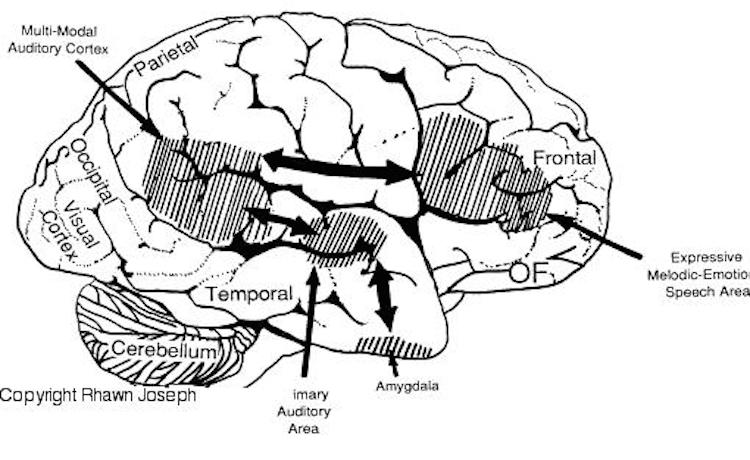
In humans, the left IPL being an indirect product of temporal lobe and superior parietal evolution (see chapter 6) is capable of multimodal processing of auditory, visual, as well as tactile impressions, and then naming this material by forming verbal associations. The IPL then injects this material into the stream of language and thought. For example, as based on functional imaging, the left IPL becomes highly active when looking at words and reading, and when engaged in word retrieval (Bookheimer, et al., 1995; Vandenberghe, et al., 1996; Menard, et al., 1996; Price, 2007). Indeed, because of its unique position at the juncture of the auditory, visual, somesthetic, and motor neocortex, it has gained the capability of analyzing, associating, and assimilating this divergent data in order to create multiple categories of visual, auditory, and tactile imagery and meaning.

Hence, because the IPL receives multi-modal input, one can feel an object while blindfolded and know what it would look like and be able to name it as well.
One can also integrate and assimilate these diverse sensory signals so as to abstract, classify and produce multiple overlapping categories of experience and cross modal associations (Geschwind, 1966; Joseph, 1982, 1986a 1993; Joseph et al., 1984).
The primary sensory receiving areas for vision, audition, and somesthesis are located in the occipital, temporal and parietal lobe respectively. Adjacent to each primary zone is a secondary-association neocortical region where higher level information processing occurs and where complex associations are formed. Wernicke's region is one such zone, as is the middle-inferior (basal) temporal and the superior parietal lobe. Moreover, there are complex third order association areas such as the middle-inferior temporal lobe (Brodmann's area 37).
Area 37 is located between the visual cortex and the anterior temporal cortex and becomes activated during a variety of language tasks, including reading and object and letter naming (Price, 2007)-- as demonstrated by functional imaging (Buchel et al., 1998; Price, 2007), direct cortical recoding (Nobre et al., 1994), and electrical stimulation (Luders et al., 1986). In fact, both normal, cognitally blind, and late-blind subject display activity in the medial temporal area (Buchel et al., 1998). Moreover, similar to injuries in the IPL, if the middle-inferior temporal lobe is injured, patients may suffer from reading and naming deficits (Rapcsak, et al., 2007); a condition referred to as phonological alexia. As noted, deficits in phonological processing are the most common correlate of reading disability (Brady & Shankweiler, 2001).
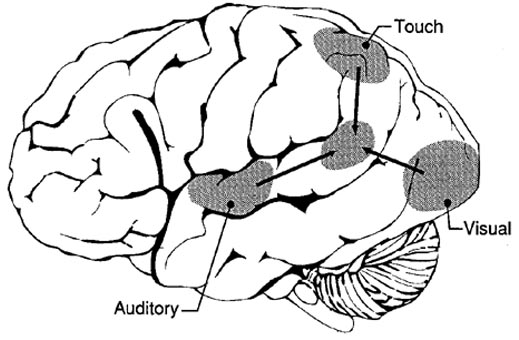
The IPL (which includes the angular and supramarginal gyri) is located at the junction where the all secondary and multi-modal association areas meet and overlap, and receives converging visual-linguistic input from the basal-lateral (middle-inferior) temporal lobe. In this regard, the inferior parietal region receives converging higher order information from each sensory modality and all association areas and in fact makes possible the formation of multiple associations based on the assimilation of this divergent sensory input (Geschwind, 1965, Joseph, 1982, 1993). One can thus feel an object while blindfolded and know what it would look like and be able to name it as well.
Through its involvement in the construction of cross-modal associations, this region acts so as to increase the capacity for abstraction, categorization, differentiation, and the verbal as well as visual labeling of sensory-motor experience. One is thus able to classify a single stimulus or event in multiple ways. In part this is made possible because the inferior parietal lobule is the recipient of the simple and complex associations already performed in the primary and association cortices via the ten billion axonal interconnections that occur in this region.
STIMULUS ANCHORS AND THE TRAIN OF THOUGHT
The left IPL of which it is part, makes possible the assimilation of complex associations which have been constructed elsewhere so that multiple classifications, categorizations, and descriptions are possible. The IPL also acts to integrate and arrange them according to preestablished (gestural) temporal sequences and the requirements of what needs to be communicated.

Moreover, via rich interconnections with Wernicke's area and the middle temporal lobe, the IPL it is able to associate auditory/verbal labels with other sensory experiences such that we can describe things as "sticky, sweet, moist, red, lumpy," as well as use single word descriptions, e.g. "jelly." This capability is particularly important in regard to reading and naming as described in further detail in chapters 20, 21. For instance, when a word is read, the pattern of visual input is transmitted from the visual areas in the occipital and temporal lobes to the left IPL (which is coextensive with Wernicke's area) and which then performs an auditory visual matching function. That is, it calls for and integrates the auditory equivalent of what is viewed or read so that we can name animals, objects, words and letters and know what the name sounds like. If this area were damaged, reading ability would be lost, a function in part, of disconnection between the IPL and the middle-inferior temporal lobe.
The Train of Associations.
As noted, the left IPL (including the left posterior-superior temporal lobe become more active when reading (Bookheimer, et al., 1995; Menard, et al., 1996; Price, 2007 Price, et al., 1996; Vandenberghe, et al., 1996) and becomes active during semantic processing (Price, 2007), and when making semantic decisions, such as when reading words with similar meanings (Price, 2007). These same areas are activated during word generation (Shaywitz, et al., 1995; Warburton, et al., 1996), and sentence comprehension tasks (Bottini, et al., 1994; Fletcher et al., 1995).
In most instances in which the IPL is activated via internal or external sources of stimulation, multiple trains of inquiry are initiated via the numerous interconnections this areas maintains. Impressions, memories, ideas, and feelings which are in any manner associated with the initial stimulus probe, are aroused in response.
If a student is asked: "What did you do in school today?" a number of verbal and memory associations and association areas are aroused in parallel and integrated within the Language Axis, all of which are related in some manner to each element of the eliciting stimulus. Finally, in the process of associational linkage, those associations with the strongest stimulus value and which most closely match each element of the question in terms of internal and external appropriateness and thus with the highest probability of being the most relevant, rapidly take a place in a hierarchical and sequential, grammatical arrangement that is being organized in a form suitable for expressing a reply.

To return to the question regarding "school," each speech segment and sound unit become triggers which first activate and then, like a magnet, draws associations accordingly. All aroused forms of mental imagery, verbal associations and so on which are received in the IPL are then arranged, individually matched and group matched such that the associations which correspond to all sources of relevant input with sufficient value of probability then act as templates of excitation that stimulate and attract other relevant ideas and associations. These in turn are assimilated and associated or are subsequently deactivated due to their low probability in contrast to the association already organized.
Moreover, because the strength and value of closely linked associations change in correspondence to the developing sequential hierarchy (or the initial parallel hierarchies), previously aroused and assimilated material may subsequently come to have a now lower value of probability or appropriateness within the the matrix of overall activity and may be deactivated (Joseph, 1982, 1986a, 1993).
Consider the question: "What is furry, small, loves milk and makes the sound Meoww?" At the level of the neocortex, each word, "furry," "small," "milk" and "meoww," acts to trigger associations (e.g. "furry = coat-animal-...," "milk = liquid-white-cow-..."). The grammatical linkage of these words also acts to trigger certain associations (e.g. "furry-milk-meoww = animal-cat-...") while deactivating others (e.g. "cow"). Following the analysis and comprehension of these sounds and words in Wernicke's area, the angular gyrus, and the middle temporal lobe, the IPL continues to call forth associations so that a reply to the question can be generated.
So that the animal can be named, the IPL via its interactions with the temporal lobe, activates the necessary phonemic elements (e.g. "k-a-t"), and then transfers this information to Broca's area and the question is answered: "Cat." If instead the individuals replies "tak" this would indicate a problem in organizing the correct phonemic elements once they were activated (see chapter 21 for an extended and detailed discussion).
The final product of this hierarchical, highly grammatical arrangement of mutually determining and parallel associational linkages is the train of thought or a temporal-sequential stream of auditory associations in the form of words and sentences. However, before this occurs, these verbal associations must receive a final temporal-sequential grammatical stamp which is a consequence of the organization imposed on this material as it passes from Broca's area to the oral-speech musculature.
Following massive lesions of a brain area with which it normally communicates, the language axis sometimes begins to invent an answer or reply to questions based on the information available despite the gaps in that data or the incongruent nature of what is being reported. Consider, for example, denial of blindness (following massive injuries to the visual neocortex) or denial or neglect of the left extremity which may also be paralyzed (due to massive right cerebral injuries involving the motor and parietal neocortex). Patients will claim to have sight although they bump into objects or fall, or they may claim that their paralyzed left arm belongs to the doctor or a person in the next room (chapter 10).
To be informed about the left leg or left arm, the Language Axis must be able to communicate with the neocortical area which is responsible for perceiving and analyzing information about the extremities. For example, since the right parietal area maintains the somesthetic body-image, as well the storage site for body-image memories, when that areas is destroyed, the left half of the "body-image" all associated memories and essentially "erased" -as if they never existed.
When no message is received by the Language Axis, due to destruction of the neocortical area responsible for that message or memory, and when the Language Axis is not informed that no messages are being received (because the brain area which would alert them is no longer functioning), the language zones instead rely on some other source even when that source provides erroneous input (Joseph 1982, 1986a). Substitute material is assimilated and expressed and corrections cannot be made (due to loss of input from the relevant knowledge source) and the patient begins to confabulate (see chapters 10, 19). That is, the Language Axis fills the "gap" with erroneous material.
DISORDERS OF LANGUAGE & THOUGHT
THE LANGUAGE AXIS
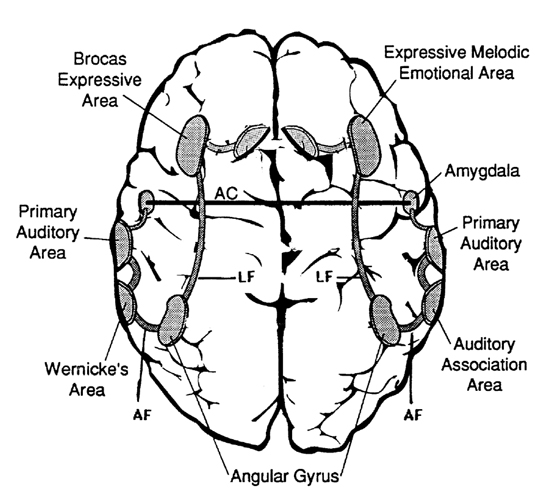
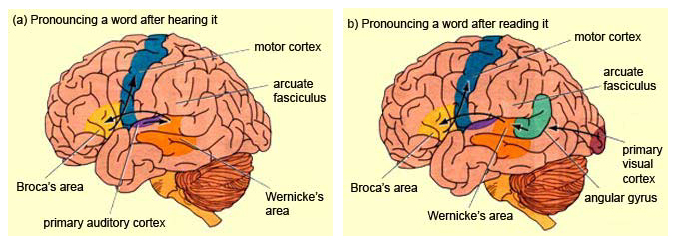
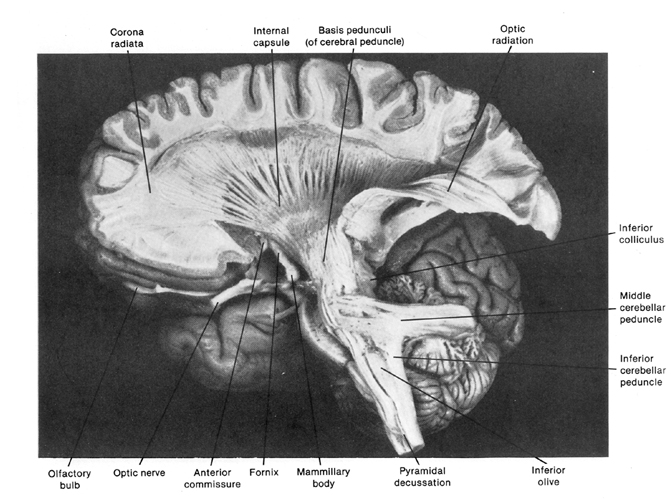
When listening to someone speak the information is transferred from the brainstem to the inferior colliculus and medial geniculate of the thalamus, and transferred to the amygdala and the primary auditory cortex where the data is extensively analyzed (see chapter 21). These auditory signals are then transferred to Wernicke's area (which merges with the angular gyrus) where the temporal-sequential, semantic and related linguistic features are stabilized, extracted, analyzed and labled.
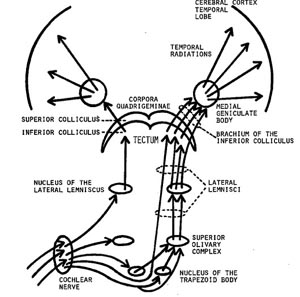
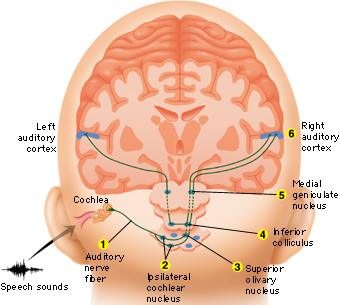
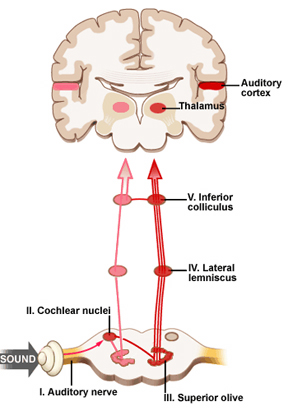
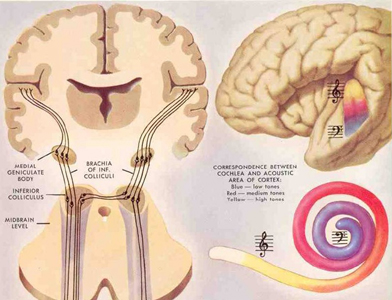
If it is a question which has been asked, the message is transferred from Wernicke's to the inferior parietal lobule where various ideational associations are aroused and organized via it's vast interconnections with other cortical regions. Presumably a series of interactions continue to occur between Wernicke's and the inferior parietal lobe (as these areas are coextensive) until a reply is formulated and properly organized for possible expression at which point it is transferred to Broca's area (Geschwind, 1965; Joseph, 1982)--as also demonstrated through functional imaging (Buchel et al., 1998; Demonet, et al., 1994; Paulesu, et al., 1993; Peterson et al., 1988; Price, 2007).
Specifically, the semantically correct and suitably chosen reply is transferred from Wernicke's region and the inferior parietal lobe, via the interlining axonal bundle, the arcuate fasciculus, to Broca's area which then programs the speech musculature and neocortical motor areas so that the reply can expressed.
CONDUCTION APHASIA
Damage involving the arcuate fasciculus and/or the supramarginal gyrus of the left hemisphere can result in a condition referred to as conduction aphasia (Benson et al. 1973; Geschwind, 1965). Broca's area is essentially disconnected from the inferior parietal lobule and Wernicke's area, and although comprehension is intact, the patient cannot repeat words or read out loud. In these cases the lesion may extend to the insula and auditory cortex and underlying white matter of the left temporal lob, thereby destroying the axonal fibers of passage.

Individuals with this disorder have great difficulty communicating because a lesion in this vicinity disconnects Broca's area from the posterior language zones. Although a patient would know what he wanted to say, he would be unable to say it. Nor would he be able to repeat simple statements, read out loud, or write to dictation, although the ability to comprehend speech and written language would remain intact.
Nevertheless, these individuals are still able to talk. Unfortunately, most of what they say is contaminated by fluent paraphasic errors, phonetic word substitutions and the telescoping of words due to impaired sequencing. Patients also tend to confuse words which are phonetically similar. Because they can comprehend they are aware of their disturbances and will try to come up with the correct words via the generation of successive approximations (Marcie & Hecaen, 2012). Hence, speech may be circumlocutious, seemingly tangential, as well as contaminated by paraphasic distortions. Sentences are usually short and are often unrelated to each other (Marcie & Hecaen, 2012).
When writing, grapheme formation is usually normal. However, because they produce the wrong words and/or misspell what they produce, patients may frequently cross out words and perform overwriting (i.e. writing over various letters with additional strokes). Patients are frequently very frustrated, irritable, and upset regarding their condition.
Various forms of linguistic information are transferred from the posterior portion of the Language Axis (via an axonal fiber bundle, the arcuate fasciculus) and converge upon Broca's area (Buchel et al., 1998; Demonet, et al., 1994; Paulesu, et al., 1993; Peterson et al., 1988) where they receive their final sequential (syntactical, grammatical) inprint so as to become organized and expressed as temporally ordered motoric linguistic articulations, i.e. speech. These impulses are then transferred to the adjacent frontal motor areas which in turn control the oral musculature. Hence, verbal communication and the expression of thought in linguistic form is made possible.
ANOMIA OR WORD FINDING DIFFICULTY
Because Broca's and Wernicke's area and the inferior parietal tightly interact in the productoon of language, and as these areas are also dependent on extensive interconnections with yet other neocortical areas and neurons, damage anywhere within the left hemisphere can therefore result in "word finding difficulty."
Some people refer to as as "tip of the tongue," when they have trouble coming up with or remembering a particular word. Every individual with aphasia has some degree of word finding difficulty, i.e. dysnomia (or if severe: anomia).
Dysnomia is very common and can occur with fluent output, good comprehension and ability to repeat, and in the absence of paraphasias or other aphasic abnormalities. Although many normal individuals who have trouble finding a word sometimes experience it as on the "tip of the tongue", anomia is a much more pervasive abnormality and may involve naming objects, describing pictures, etc. even when hints are provided.
In general, anomic difficulties suggest left hemisphere functional impairment, and is sometimes secondary to disconnection, or a deficit in activating the correct phonological sound-word patterns (Kay & Ellis, 2007); a disturbance that also contributes to dyslexia (Brady & Shankweiler, 2001). Frequently, if dysnomia is the only predominant problem, the patient may erroneously ascribe it to deficient memory. That is, the patient may complain of memory problems when in fact they have word finding difficulty. However, this is not due to nmenonic deficits. If provided with hints or even the initial letter there is little improvement (Goodglass et al. 1976). Moreover, once the word is supplied the patient may again experience the same problem almost immediately. In general, the ability to generate nouns is more effected than verbs.
In severe cases, while conversing the patient may be so plagued with word finding difficulty that speech becomes "empty", and characterized by many pauses as the patient searches for words. This condition is sometimes referred to as anomic aphasia (Hecaen & Albert, 2003). These same patients may erroneously substitute phrases or words for the ones that cannt be found. For example, calling a "Spoon" the "stirrer", a "pencil" a "writer", or various objects "the whatchmacallit". This can lead to circumlocution as they tend to talk around the word they are after: "Get me the uh, the uh thing over on the uh, on the top over there..." Children are often plagued by such difficulties which they grow out of, which is the result of immaturity within the inferior parietal lobe and the late establishment of the necessary axon fiber connections with other brain areas.
If accompanied by problems with reading and writing, the lesion is probably situated in the posterior portions of the left hemisphere near the angular gyrus. However, lesions anywhere within the left hemisphere can result in anomic difficulties.
However, in some cases, depending on where the damage and thus the disconnection occurs, a person may not be able to name an item when it is shown to them (due to visual cortex-inferior parietal disconnection), but can name it if they touch it, or it is described to them out loud due to preservation of the auditory and tactile pathways linking them with the inferior parietal lobe. For example, since the axonal fibers leading from Wernicke's area and the tactual association areas have not been injured, the person can name the object if he touches it or if it is described or makes a sound (e.g. running the fingers of the teeth of a comb).
If the lesion disconnected the inferior parietal lobe from the rest of the parietal lobe, they would be able to name an object if they saw or heard it but not by touch alone. If the lesion disconnected the inferior parietal lobe from Wernicke's area, or if the inferior parietal lobe was destroyed, they would be unable to name the object regardless of modality of presentation (Freud, 1891; Geschwind, 1965; Joseph, 1993). If provided with hints or even the initial letter there is little improvement. Moreover, once the word is supplied, the patient may again experience the same problem almost immediately. In general, the ability to generate nouns is more affected than verbs.
EXPRESSIVE APHASIA
Because Broca's area acts as the final common pathway via which language is vocally and temporally-sequentially expressed (Buchel et al., 1998; Demonet, et al., 1994; Paulesu, et al., 1993; Peterson et al., 1988), damage to the left frontal convexity result in a dramatic curtailment of the ability to speak. Often immediately following a massive stroke in this region individuals suffer a paralysis of the right upper extremity and initially are almost completely unable to speak; i.e., Broca's (or expressive) aphasia (Bastiannse, 1995; Benson, 1993; Goodglass & Kaplan, 2000; Haarman & Kolk, 1994; Hofstede & Kolk, 1994; Levine & Sweet, 2005). Comprehension, however, is generally intact.

Although symptoms differ depending on the severity of the lesion, in general, individuals with Broca's aphasia are very limited in their ability to articulate or repeat statements made by others. In severe cases speech may be restricted to a few stereotyped phrases and expressions, such as "Jesus Christ" or to single words such as "Fine", "yes", "No", which are produced with much effort. Even if capable of making longer statments, much of what they say is poorly articulated and/or mumbled such that only a word or two may be intelligible (Bastiannse, 1995; Haarman & Kolk, 1994; Hofstede & Kolk, 1994). However, this allows them to make one word answers in response to questions. Nevertheless, speech is almost always aggrammatical (i.e. the production of some correct words but in the wrong order), contaminated by verbal paraphasias, i.e. "orrible" for "auto", and/or the substitution of sematically related words, e.g. mother for father and characterized by the ommission of relational words such as those which tie language together, i.e. the propositions, modifiers, articles and conjunctions.
Similarly, they sometimes have difficulty comprehending these same grammatical features (Samuels & Benson, 1993; Tyler et al. 1995; Zurif et al. 1972), as well as related verbal material such as demonstated on the Token Test (DeRenzi & Vignolo, 1962). Their ability to repeat what is said to them, although grossly deficient, is usually not as severly reduced as is conversational speech.
The ability to write is always effected. Similarly, their capacity to write to dictation is severely limited. However, the ability to copy is much better preserved. In addition reading comprehension is usually intact although they cannot read aloud.
Patients with moderate and severe expressive abnormalties also have difficulty performing 3-step commands although 2-step requests may be performed without difficulty. However, they are better able to comprehend as well as verbalize semantically significant words.
Even in moderate and mild cases a consistent defect in the syntactical structure of speech is noted including reductions in vocabulary and word fluency in both speech and writing (Bastiannse, 1995; Goodglass & Berko, 1960; Haarman & Kolk, 1994; Hofstede & Kolk, 1994; Levine & Sweet, 2005).
Individuals with expressive aphasia, although greatly limited in their ability to speak, are nevertheless capable of making emotional statements or even singing (Gardner, 1975; Goldstein, 1942 ; Joseph, 1988a; Smith, 1966; Smith & Burklund, 1966; Yamadori, Osumi, Mashuara, & Okuto, 1977). In fact, they may be able to sing words they cannot say.
Except for emotional speech, however, their language production is largely monotonic (Goodglass & Kaplan, 1982), or characterized by prosodic distortions such that in some cases they sound as if they are speaking with a foreign accent. (Graff-Radford, Coper, & Colsher, 1986). This is often due to shifts in the enunciation of vowels; i.e. increased duration of the utterance and the pauses between words as they struggle to speak. However, in some instances this is secondary to deep lesions involving perhaps the anterior cingulate and other nuclei (see Chapter 15).
Sex Differences in Expressive Aphasia.
As discussed in Chapter 7, Broca's speech area appears to be more functionally developed in women than men. That is, there is some evidence which suggests that expressive (and emotional) speech tends to be more clearly concentrated in the anterior regions of their brain (Kimura, 1993) In consequence, females are far more likely to become severely aphasic with left frontal injuries, whereas males become more severely aphasic with left parietal damage (Hier et al. 1994; Kimura, 1993).
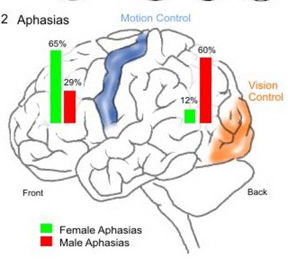

However, there is some evidence which suggests that the posterior portions of the corpus callosum, i.e. the rope of fibers that interconnect the right and left parietal lobes, is thicker and larger in females than males (Holloway et al. 1994). As gathering and related activities are more likely to involve both hands (vs hunting and throwing with a single hand), this findings might be expected, particularly in that the motor areas are dependent on the parietal somesthetic area for fine movement programming. Thus it might be expected that the posterior female corpus callosum would be larger because the female right and left parietal lobe were probably simultaneously activated and utilized by females for the last 100,000 or so years -a consequence of woman's role as gatherer, tool maker, food and hide preparer (Joseph, 2009e); activities that promote temporal-sequential motor activities and the production of speech. Thus females are more resistant to posterior injuries in regard to developing aphasic abnormalities, because they have an advantage over males in this regard.
It is thus noteworthy that boys also tend to stutter more than girls (Corballis & Beale, 2005), and stuttering is largely a male problem for adults as well as children. This suggests either dysfunction or a lack of functional (temporal sequential) development in the male inferior parietal lobule (although medial frontal, anterior cingulate, and/or abnormalities involving Broca's area and the anterior cingulage gyrus and medial frontal lobes may well be equally responsible. These sex differences in evolutionary history and thus language acquisition might also account for the many other "learning disabilities" which predominately effect boys and men.
DEPRESSION & BROCA'S APHASIA
Individuals with left frontal damage and Broca's aphasia often become frustrated, sad, tearful and depressed (Gainnoti, 1972; Joseph, 2009a; Robinson & Downhill 1996; Robinson & Szetela, 1981). Presumably this is because individuals with Broca's aphasia are fairly (but not completely) able to comprehend. Hence, they are aware of their deficit and become appropriately depressed (Gainnoti, 1972). Indeed those with the smallest lesions become the most depressed (Robinson & Benson, 1981) --the depression as well as the ability to sing being mediated, presumably by the intact right cerebral hemisphere.
It has also been reported that psychiatric patients classified as depressed (who presumably have no signs of neurological impairment) often demonstrate, electrophysiologically, insufficient activation of the left frontal lobe (d'Elia & Perris, 1973; Perris, 1974). With recovery from depression left hemisphere activation returns to normal levels.
Functional imaging of depressed states also indicates reduced activity in the left frontal lobe and anterior cingulate (Bench, et al., 1992). And when patients ceased to be depressed, activity levels increase (Bench et al., 1995). Sex Differences.
Females are far more likely to experience depression and depressive episodes than males (DSM IV). Although this sex difference is no doubt related to differential stresses and hormonal factors affecting women vs men, this may also be secondary to a greater likelihood that females are at greater risk for anterior cerebral artery dysfunction (e.g. embolism; Hier et al. 1994) and become depressed with even subtle injuries involving the left (and right) frontal cortices. That is, females (and males) with small left frontal strokes may be diagnosed as depressed when in fact they have suffered a cerebrovascular infarct.
APATHETIC STATES
Depression and depressive-like features also occur with left frontal and medial lesions which spare Broca's area (Robinson et al. 1984; Robinson and Szetela 1981; Sinyour, et al. 1986). However, rather than depression per se (particularly when the frontal pole is damaged), such patients frequently appear severely apathetic, blunted, hypoactive with reduced motor functioning, and are poorly motivated (Blumer and Benson 1975; Freeman and Watts 1942, 1943; Girgis 1971; Hecaen 1964; Luria 1980; Passingham 1993; Stuss and Benson 1986; Strom-Olsen 1946). Of course they may also be depressed. Even so, when questioned, rather than worried or truly concerned about their condition the overall picture may be one of bland confusion, disinterest and emotionally blunting and related disturbances suggestive of a blunted form of schizophrenia (Akbarian et al. 1993; Andreasen et al. 1990; Buchanan et al. 1998; Buchsbaum 1990; Carpenter et al. 1993; Casanova et al. 1992; Curtis et al. 1998; Wolkin et al. 1992; Wolkin et al. 1992).
Presumably, these depressed, blunted, and apathetic states are sometimes due to disconnection from the limbic system and/or right cerebral hemisphere. Unfortunately, with frontal pole injury, tumor or degeneration the underlying neurological precursors to their condition are not very apparent until late in the disease. Hence, misdiagnosis is likely.

For example, with deep medial frontal lesions involving the supplementary motor areas, the individual may develop catatonic symptomology (Joseph, 2009a). In contrast, apathetic, blunted, and "negative" forms of schizophrenia coupled with "psych-motor retardation" reduced verbal and intellectual output and slowness of thought, tend to associated with left frontal lobe dysfunction (Buchsbaum, 1990; Carpenter et al. 1993; Casanova et al. 1992; Leven 1994). In fact, left lateral as well as bilateral convexity abnormalities are often associated with apathetic, blunted, and "negative" forms of schizophrenia (Buchsbaum 1990; Carpenter et al. 1993; Casanova et al. 1992; Weinberger 2007; Wolkin et al. 1992). Patients classified as schizophrenic have also been reported to demonstrate abnormal left (or bilateral) frontal lobe EEG's indicative of hypoarousal (Akbarian et al. 1993; Ariel et al. 2005; Ingvar and Franzen 1974; Kolb and Whishaw 2005; Levin 1984). Lateral frontal gray matter and brain volume reductions and decreased activity have been repeatedly noted (Andreasen et al. 1990; Buchanan et al. 1998; Curtis et al. 1998), including decreased blood flow (Weinberger et al. 1986) hypoactivity (Wolkin et al. 1992), and reduced metabolism (Buchsbaum et al. 1992).


When the lesions or pattern of abnormality involve the lateral convexity, underlying white matter and long distance axons of passage leading to the head of the caudate, there can also result a disconnection involving impulse and thought control. For example, a lesion involving the frontal-caudate-putamen "loop" may derail the entire system thereby giving rise to retarded motor, cognitive and verbal functioning such that the patient appears to be suffering from a blunted "schizophrenic" psychosis. These and related frontal lobe and basal ganglia disorders will be detailed in chapters 16, 19.
SOUND PERECEPTION, LANGUAGE & APHASIA
PURE WORD DEAFNESS
When the primary auditory area is destroyed, the auditory association areas (Wernicke's area in the left hemisphere) are disconnected from all sources of auditory input and thus cannot extract meaning from the auditory environment. When individuals are unable to perceive and identify linguistic and non-lingusitic sounds the deficit is described as a global or generalized auditory agnosia (Schnider et al. 1994). Cerebrovascular disease is the most common cause of this abnormality.
These patients are not deaf, however, as this can be ruled out by testing pure-tone threasholds. In fact, even with complete bilateral destruction of the primary auditory cortex there is no permanent loss of acoustic sensitivity (Rubens, 1993). This is because sounds continue to be received in thalamic and subcortical centers including the amygdala. Hence, patients can still detect sounds. They just don't know what the sounds are (Schnider et al. 1994). Moreover deficits in loudness discrimination and sound localization are common.
In some instances, damage may be unilateral and involve predominantly the primary auditory receiving area of the left hemisphere as well as the underlying white matter such that Wernicke's region becomes disconnected from all sources of auditory input. Although the patient can hear and identify non-linguistic and environmental sounds, they are unable to comprehend spoken language. These patients do not have Wernickes aphasia, however, and are very aware of their deficits. Their own verbal output is normal (albeit sometimes loud and dysprosodic) and frequently they describe the sounds they hear as being "muffled", or they may complain that voices sound like "echos" or "noise" (Buchman et al. 1986; Tanaka et al, 2007).
Although they cannot repeat what is said to them, patients can read, and write in a normal manner and there is little or no evidence of aphasic symptoms (Schnider et al. 1994). Nor do they demonstrate difficulty understanding non-verbal or pantomimed actions.
Nevertheless, some patients, as they recover from Wernickes aphasia move to the stage of pure word deafness. However, in these instances there are remnants of aphasic abnormalities.
Many such patients, like those who suffer some forms of deafness, may experience auditory hallucinations and/or exhibit paranoid ideation (Rubens, 1993). The hallucinations are likely a consequence of the attribution or erroneous extraction of meaning from randomly produced neural "noise". The paranoia is largely a normal reaction arising secondarily from the confusion associated with reduced verbal and social-linguistic contact. That is, patient's become fearful and mistrustful as they are not sure what is "going on".
In part, pure word deafness is sometimes due to disturbances in processing rhythm and temporal sequences (Tanaka et al. 1974), or making phonemic or semantic descrimations (Denes & Semenza, 1975; Schnider et al. 1994). Thus, slowing the rate of sound presentation or rate of speech can improve comprehension (Buchman et al. 1986).
WERNICKE'S APHASIA
Within the left superior temporal lobe extending from the border zones of the primary auditory reception area toward the inferior parietal lobule is located Wernicke's area. Wernickes area (in conjunction and with the aid of the inferior parietal lobule; Kimura, 1993) acts to decode and encode auditory-linguistic information (be it externally or internally generated), so as to extract or impart temporal-sequential order and related linguistic features. In this manner denotative meaning may be discerned or applied (Efron, 1963; Lackner & Teuber, 1973; Joseph, 1982, 1988a; Lenneberg, 1967). That is, this area acts to verbally label information transmitted from external sources as well as from other brain regions. For example, it may act to provide the auditory equivalent of a visually perceived written word. In this manner, we know what the words we read sound like.
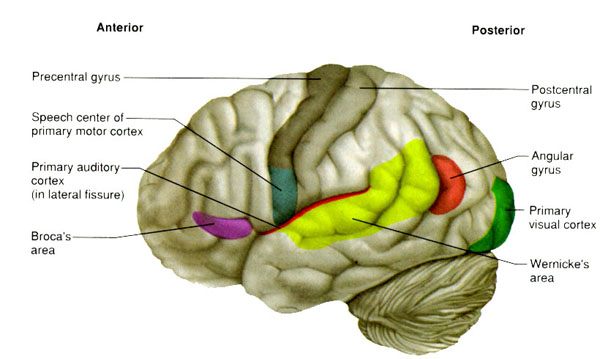

RECEPTIVE APHASIA
If the auditory association (Wernicke's) area is damaged patients will have great difficulty comprehending spoken or written language (Goodglass & Kaplan, 1982; Kertesz, 2005a; Hecaen & Albert, 2003). Naming, reading, writing and the ability to repeat or understand what is said are severely effected, i.e. Wernicke's (receptive) aphasia. When patients prove capable of reading, then they are probably suffering from "pure word deafness" rather than receptive aphasia (to be explained).
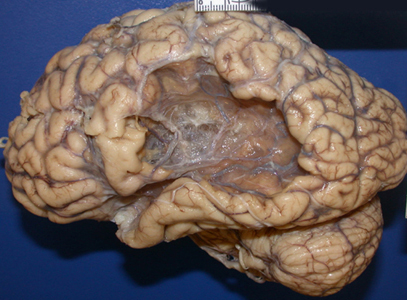
Frequently disturbances involving linguistic comprehension are due to an impaired capacity to discern the individual units of speech and their temporal order -due to destruction of the superior temporal lobe and adjacent tissue. Sounds must be separated into discrete interrelated linear units or they will be perceived as a blur, or even as a foreign language (Carmon & Nachshon, 1975; Efron, 1963; Joseph, 1982, 1988a; Lackner & Teuber, 1973). Hence, a patient with Wernicke's aphasia may perceive a spoken sentence such as the "big black dog" as "the klabgigdod". However, comprehension is improved if the spoken words are separated by long intervals.
Many receptive aphasics can comprehend frequently used words but have difficulty with those less frequently heard. Thus loss of comprehension is not an all-or-none phonemeonon. They will usually have the most difficulty understanding relational or syntactial structures, including the use of verb tense, possessives, and prepositions. However, by speaking slowly and by emphasizing the pauses between each individual word, comprehension can be modestly improved.
Fluent Aphasia.
Patients with damage to Wernicke's area are still capable of talking (due to preservation of Broca's area). However, because Wernicke's area also acts to code linguistic stimuli for expression (prior to its transmission to Broca's area), expressive speech becomes severely abnormal, lacking in content, containing neologistic distortions (e.g. "the razgabin"), and/or characterized by non-sequitars, literal (sound substitution) and verbal paraphasic (word substitution) errors, a paucity of nouns and verbs, and the ommission of pauses and sentence endings (Christman, 1994; Goodglass & Kaplan, 1982; Kertesz, 2005a; Hecaen & Albert, 2003). Patients may speak in a rush (e.g. press of speech) and what is said often convey very little actual information; a condition referred to as fluent aphasia. They also may have difficulty establishing the boundaries of phonetic (confusing "love" for "glove") and semantic (cigarrette for ashtray) auditory information.
The speech of these patients may also be characterized by long, seemingly complex (albeit unintelligible) grammatically correct sentences, such that speech is often hyperfluent and produced at an excessive rate. They thus have difficulty bringing sentences to a close and many words are unitelligibly strung together. They also suffer severe word finding difficulty which adds a circumlocutory aspect to their speech which can deteriorate into jargon aphasia such that no meaningful communication can be made. (Christman 1994; Kertesz, 2005a; Marcie & Hecaen, 2012).
For example, one patient with severe receptive aphasia responded in the following manner: "....Oh hear but that was a long time ago that was when that when before I even knew that much about this place although I am a little suspicious about what the hell is the part there is one part scares, uh estate spares, Ok that has a bunch of drives in it and a bunch of good googin, nothing real big but that was in the same time I coached them I said hey stay out of the spear struggle stay out of trouble so dont get and my kidds, uh except for the body the boys are pretty good although lately they have become winded or something...what the hell...kind of a platz goasted klack..."
Presumably because the coding mechanisms involved in organizing what they are planning tosay are the same mechanisms which decode what they hear, expressive as well as receptive speech becomes equally disrupted. In fact, one gauge of comprehension can be based on the amount of normalcy in their language use. That is, if they can repeat only a few words normally, it is likely that they can only comprehend a few words as well. Nevertheless, in testing for comprehension it is important to insure that the patient's major difficulty is not apraxia or agnosia, rather than aphasia (to be discussed).
In addition, like speech the ability to write may be preserved, although what is written is usually completely unitelligible consisting ofjargon and neologistic distortions. Copying written material is possible although it is also often contaminated by errors.
ANOSOGNOSIA

Although their speech is bizarre, in severe cases patients with receptive "fluent" aphasia do not realize that what they say is meaningless (Maher et al. 1994). Moreover, they may fail to comprehend that what they hear is meaningless as well (Lebrun, 2007). Nor can you tell them since they are unable to comprehend. This is because when Wernickes area is damaged, there is no other region left to analyze the linguistic components of speech and language. The brain cannot be alerted to the patient's disability. They don't know that they don't know; that they don't understand. However, they may be somewhat more capable of recogning that their writing is abnormal (Marcie & Hecaen, 2012).
APHASIA & EMOTION
Patients with Wernicke's aphasia in some instances may display euphoria due to the disorganizing effect of comprehension loss on emotional functioning, and/or due to involvement of the temporal lobe limbic nuclei (e.g. amygdala). That is, the patient's right hemisphere continues to respond to signals generated by the left even though they are abnormal.
In some cases patients become paranoid as there remains a non-linguistic emotional awareness that something is not right; that what they hear and what they observe does not mesh or make sense. That is, although aphasic, emotional functioning and affective comprehension may remain intact, though it is sometimes disrupted due to erroneously processed verbal input (Boller & Green, 1972; Boller et al. 2012). As such, they are exceedingly sensitive to social-emotional nuances which they may interpret and respond to correctly. Similarly, the ability to read and write emotional words (as compared to non-emotional or abstract words) is also somewhat preserved among aphasics (Landis et al. 1982). This is because the right hemisphere is intact.
Since these paralingusitic and emotional features of language are analyzed by the intact right cerebral hemisphere, the aphasic individual is able to grasp in general the meaning or intent of a speaker, although verbal comprehension is reduced. This in turn enables them to react in a somewhat appropriate fashion when spoken to. Unfortunately, this also makes them appear to comprehend much more than they are capable of.
Similarly, Wernicke's aphasics also retain the melodic, prosodic, and intonation contours of speech . In fact, at times their speech can become hypermelodic as well as characterized by inapropriate abnormal fluctuations in melodic contour. In addition, these patients often show normal gestural and facial expressions (Marci & Hecaen, 2012).


According to Benson (1993, p. 42), "those with Wernicke's aphasia often have no apparent physical or elementary neurological disability. Not infrequently, the individual who suddenly fails to comprehend spoken language and whose output is contaminated with jargon is diagnosed as psychotic. Patients with Wernicke's aphasia certainly inhabited some of the old lunatic asylums and probably are still being misplaced."
In fact, and as detailed in chapter 21, the left temporal lobe has been repeatedly implicated in schizophrenia as measured by positron-emission tomography (e.g. McGuire et al. 1998), P300 evoked potential amplitude (Bruder et al., 2009; Salisbury et al. 1998) and MRI (Jacobsen et al. 1998; Shidhabuddin et al. 1998; Kwon et al., 2009).
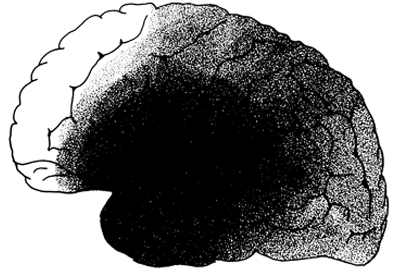
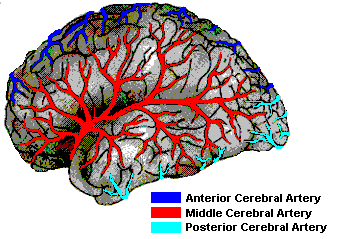
Frequently this disturbance is secondary to cerebrovascular disease involving the middle cerebral artery. However, tumors and head injuries can also create this condition.
Nevertheless, like global aphasia, the rest of the cerebrum is unable to communicate with the language zones. As such, an individual is unable to verbally describe what they see, feel, touch, or desire. Moreover, because the Language Axis cannot communicate with the rest of the brain, linguistic comprehension is largely abolished.
That is, although communication between Wernicke's, Broca's and the inferior parietal lobule is maintained, associations from other brain regions cannot reach the speech center. Although able to talk, the patient has nothing to say. Moreover, although able to see and hear, the patient is unable to linguistically understand what they perceive. However, they are capable of generating automatic-like responses to well known phrases, prayers, or songs.
In an interesting case described by Geschwind, Quadfasel and Segarra, (1968), a 22 year old woman with massive destruction of cortical tissue due to gas asphyxiation was found to have a preserved Language Axis. It was noted once the patient regained "consciousness" that "she sang songs and repeated statements made by the physicians. However, she would follow no commands, resisted passive movements of her extremities, and would become markedly agitated and sometimes injured hospital personnel. In all other regards, however, she was completely without comprehension or the ability to communicate.
The patients spontaneous speech was limited to a few stereotyped phrases, such as "Hi daddy", "So can daddy", "mother", or "Dirty bastard". She never uttered a sentence of propositional speech over the nine years of observation. She never asked for anything and she never replied to questions and showed no evidence of having comprehended anything said to her. Occasionally, however, when the examiner said, 'ask me no questions', she would reply 'I'll tell you no lies,' or when told, 'close your eyes' she might say 'go to sleep.' When asked, "Is this a rose?" she might say, "roses are red, violets are blue, sugar is sweet and so are you.": To the word "Coffee" she sometimes said, "I love coffee, I love tea, I love the girls and the girls love me. An even more striking phenomenon was observed early in the patients illness. She wold sing along with songs or musical commercials sung over the radio or would recite prayers along with the priest during religious broadcasts. If a record of a familiar song was played the patient would sing along with it. If the records was stopped she would continue singing correctly both words and music for another few lines and then stop. If the examiner kept humming the tune the patient would continue singing the words to the end. New songs were played to her and it was found she could learn these as evidenced by her ability to sing a few lines correctly after the record had been stopped. Furthermore, she could sing two different sets of words to the same melody. For example, she could sing "Let me call you sweetheart" with the conventional words, but also learned the parody beginning with "Let me call you rummy". Her articulation of the sounds and her production of melody were correct although she might sometimes substitute the words "dirty bastard" for some of the syllables" (pp 343-346).
Nevertheless, although such patients are able to sing, curse, and even pray, it is not clear if these expressions are the product of right hemisphere activity or reflexive activation of the intact language zones (or both). I favor the former rather than the later explanations.
In some cases the isolation is only partial, involving either than anterior or posterior regions. In these instances the disorder is referred to as transcortical motor or transcortical sensory aphasia respectively (see Hecaen & Albert, 2003 for greater detail). Individuals with a partial transcortical aphasia have been reported able to read out loud, to write to dictation, and to repeat simple verbal statements. However, although repetition is somewhat preserved, spontaneous speech is severely limited. In sensory transcortical disturbances comprehension is largely lost, whereas with motor transcortical there is a greater preservation of comprehension.
Because the hand and oral-articulatory neural structures occupy adjoining neocortical space sometimes dual and simultaneous activation of these two modalities results in competitive interference. For example, while speaking the ability to simultaneously track, manually sequence, position, or maintain stabilization of the arms and hands is concurrently disrupted (Bathurst & Kee, 1994; Hicks, 1975; Lomas & Kimura, 1976; Kimura & Archibald, 1974; Kinsbourne & Cook, 1971). And as the phonetic difficulty of the verbalizations increase (Hicks, 1975), motor control decreases--a function presumably of simultaneous activation of (and thus competition for) the same neurons.
Conversely, when the left hemisphere is damaged, the right extremity may become paralyzed, the individual may be plagued by apraxic disorders (see below) and performance on problems involving not only language but temporal order are selectively impaired (Carmon & Nachshon, 1971; Efron, 1963; Haaland & Harrington, 1994; Kimura, 1993; Lackner & Teuber, 1973) as is non-verbal manual or oral performance (Mateer & Kimura, 1977; Heilman, et al. 1975; Kimura, 1993), the copying of meaningless movements (Kimura & Archibald, 1974) rapidly of limb and pursuit-rotor movements (Heilman et al. 1975; Wyke, 1967) and the analysis of temporal speech sequences (Lenneberg, 1967).
Temporal-sequencing is of course a fundamental property of language as demonstrated by the use of syntax and grammar. That is, syntax is a system of rules which govern the positioning of various lexical items and their interrelations to one another. This allows us to do more than merely name but to describe and to analyze how various parts and segements of speech interrelate. We can determine what comes first or last (e.g. "point to the door after you point to the window"), and what is the subject and object. When the left hemisphere is damaged, particularly the anterior portions, expressive and receptive aspects of synactical information processing suffer.
Hence, we find that the ability to extract denotative meaning from language is dependent on the ability to organize and coordinate speech into temporal and interrelated units --an ability at which the left hemisphere excells, and an ability which is at least in part an outgrowth or a function of motoric processing and the predominate use of the right hand for gathering, tool making, food preparation, and related temporal and sequential activities.



As the child and it's brain matures, instead of predominantly touching, grasping, and holding, the fingers of the hand are used for pointing and then naming the object indicated. It is these same fingers which are later used for counting and the development of temporal-sequential reasoning; i.e. the child learns to count on his or her fingers, then to count (or name) by pointing at objects in space.
In this regard, counting, naming, object identification, finger utilization, and hand control are ontogenetically linked. In fact, these capacities seem to relie on the same neural substrates for their expression; i.e. the left inferior parietal lobule. Hence, when the more posterior portions of the left hemisphere are damaged, naming (anomia), finger recognition (finger agnosia) object identification (agnosia), arithmetical abilities (acalculia), and temporal-sequential control over the hands and extremities (apraxia) are frequently compromised.

It is relationships such as these which lend considerable credence to the arguement that over the course of evolution the predominant useage of the right hand enabled the left brain to develop neurons specialized for counting, naming, and for subserving the development of the temporal-sequential properties necessary for the mediation of grammatical-syntactial speech and language.
Nevertheless, it sometimes occurs that patients not only fail to recognize their errors, but fail to even recognize whatever object they are attempting to examine or reproduce. This condition is referred to as Agnosia (a term presumably coined by Sigmund Freud during his studies in neurology).
FINGER AGNOSIA
Finger agnosia is not a form of finger blindness, as the name suggests. Nor is it an inability to recognize a finger as a finger. Rather, the difficulty involves naming and differentiating among the fingers of either hand as well as the hands of others (Gerstmann, 1940). This includes pointing to fingers named by the examiner, or moving or indicating a particular finger on one hand when the same finger is stimulated on the opposite hand.
In addition, if you touch their finger while eyes are closed, and ask them to touch the same finger they may have difficulty. Often patients who have difficulty identifying fingers by name or simply differentiating between them non-verbally also suffer from receptive language abnormalities (Sanquet, et al. 1971). However, disturbances differentiating between the different fingers can occur independent of language abnormalities or with right parietal injuries (in which case the problem is seen only with the patient's left hand).
Usually, however, finger agnosia is associated with left parietal (as well as temporal-occipital) lobe lesions in which case the angosia is demonstrable in both hands. It is also part of the constellation of symptoms often referred to as Gerstmanns Syndrome, i.e. agraphia, acalculia, left-right disorientation, finger agnosia (Gerstmann, 1942). Gerstmann's symptom complex is most often associated with lesions in the area of the supramarginal gyrus and superior parietal lobule (Hrbek, 1977; Strub & Geschwind, 2005).
VISUAL AGNOSIA
Visual agnosia is a condition where the patient loses the capacity to visually recognize objects, although visual sensory functioning is largely normal. That is, objects are detected but ability to evoke or assign meaning is lost and the object cannot be identified (Critchley, 1964; Shelton et al. 1994; Teuber, 1968). The percept becomes stripped of its meaning.
For example, if shown a comb, the patient might have no idea as to what it is or what it might be used for. However, they may be capable of giving a rough estimate of its size and proportions and other features.
In general, this disturbances is associated with damage involving the medial and deep mesial portion of the left occipial lobe as well as the basal (middle-inferior) temporal lobe. Indeed, the left posterior basal temporal lobe is a convergence zone for visual, auditory, and tactile information, and becomes activated during a variety of language tasks, including reading and object and letter naming (Price, 2007). This has been demonstrated not only by functional imaging (Buchel et al., 1998; Price, 2007), but direct cortical recording (Nobre et al., 1994), and electrical stimulation (Luders et al., 1986). In fact, both normal, cognitally blind, and late-blind subject display activity in this area (Buchel et al., 1998). Hence, if injured, patients may suffer from naming deficits (Rapcsak, et al., 2007), i.e. anomia, which is distinguished from agnosia.
Agnosia, however, is not a naming disorder. Individuals with anomia have difficulty naming an object if it is presented by touch, by sound, via visual inspection, or through auditory description. Individuals may have agnosia limited to a single or multiple modalities (Davidoff & De Bleser, 1994; Shelton et al. 1994). If shown a comb they may be unable to name or describe it's use. However, if placed in their hand and encouraged to palpate and tactually explore it, they may be able to identify it without difficulty (Davidoff & Bleser, 1994). Conversely, patients who can recognize an item by sight but not by touch suffer from stereognostic abnormalities due to parietal lesions. Hence, anomia and angosia can occur across modalities or it may be limited to a single modality.

Simultanagnosia is an inability to see more than one thing or one aspect of an object at a time, although individual details may be correctly perceived (Bousen & Humphrey, 2009; Laeng, et al., 2009). However, the patient is unable to relate the different details so as to discern what is being viewed. For example, if shown a picture of a man holding an umbrella and a suitcase, they may see the suitcase and the man, and the umbrella, but be unable to relate these items into a meaningful whole. In fact, by surrounding the object with other objects perceptual recognition may deteriorate even further (Luria, 1980; Shelton et al. 1994).
With severe damage the patient may be unable to even recognize individual objects. Indeed, one patient I examined with bilateral posterior parietal-occipital damage (following a bilateral posterior artery stroke) could only identify parts of objects, but not the object itself.

As described by Luria (1980), this is due to a breakdown in the ability to perform serial feature-by-feature visual analysis, and is sometimes accompanied by abnormal eye movements. Nevertheless, a variety of anatomical regions, when compromised can give rise to this abnormality. For example, simultanagnosia has been noted to occur with left, right and bilateral superior occipital lobe lesions, or injuries involving the frontal eye fields (Bousen & Humphrey, 2009; Kinsbourne & Warrington, 1962; Laeng, et al., 2009; Luria, 1980; Rizzo & Hurtig, 2007). Moreover, I have observed patients with diffuse degenerative disturbances who demonstrate simultangnosic deficiencies.
RIGHT-LEFT DISORIENTATION
Right-left disorientation (e.g. "show me your right hand") is usually associated with left hemisphere and left parietal-occipital damage. It occurs only extremely rarely among individuals with right cerebral injuries (Gerstamann, 1930; McFie & Zangwill, 1960; Sauguet et al. 1971).
In general, these patients have difficulty differentiating between the right and left halves of their body or the bodies of others. This may be demonstrated in a number of ways: By asking patient to touch or point to the side named by the examiner, e.g. "touch your left cheek", or "point to my right ear"; to point on their own body to the body part the examiner has pointed to on his/her body, or in performing crossed commands, "touch your left ear with your right hand". In mild cases patients have difficulty only with the crossed commands (Strub & Geschwind, 2005).
In part, it seems somewhat odd that right-left spatial disorientation is more associated with left than right cerebral injuries given the tremendous involvement the right half of the brain has in spatial synthesis and geometrical analysis. However, orientation to the left or right transcends geometric space as it relies on language. That is, "left" and "right" are designated by words and defined linguistically. In this regard, left and right become subordinated to language usage and organization (Luria, 1980). Hence, left-right confusion is strongly related to problems integrating spatial coordinates within a linguistic framework. It is perhaps for this reason that individuals with aphasic disorders generally perform most poorly of all brain damaged groups on left-right orientation tasks (Sauguet et. al. 1971).
ACALCULIA
Mathematical thinking can be language dependent or dependent on spatial thinking (Dehaene, et al., 2009; Joseph, 1988a). Einstein, for example, stated that "words and language, whether written or spoken, do not seem to lay any part in my thought processes. The psychological entities that serve as building blocks for my thought are certain signs or images, more or less clear, that I can reproduce at will." In fact, as recently demonstrated by Dehaene and colleagues (2009), some aspects of mathematical thinking, such as "approximate" arithmetic, is language independent and "relies on a sense of numerical magnitudes, and recruits bilateral areas of the parietal lobes involved in visuo-spatial processing."
By contrast, "exact arithmetic" is language dependent and is acquired in a language context, and as based on functional imaging, activates the language areas of the left hemisphere. In this regard it is noteworthy that whereas the IPL is directly implicated in mathematical and geometric thinking, that Einstein IPL was in fact larger than normal by 15% (Witelson et al., 2009), and that the Sylvian fissure stopped well short of his IPL which in turn may have allowed more neurons in this area to flourish and grow and to establish interconnections.
When an individual has difficulty working with numbers or performing arithmetical operations secondary to a brain injury, the manner and level at which the problem is expressed will differ depending on which part of the cerebrum has been compromised (e.g. Cohen & Dehaene 1994; Watson et al., 2009). For example, an individual with a parietal-occipital lesion may suffer an Alexia/Agnosia for Numbers, in which case they are unable to read or recognize different numerals. An individual with a posterior right cerebral injury may have difficulties with spatial-perceptual functioning and thus misalign numbers when adding or subtracting (referred to as Spatial Acalculia). Or they may suffer a very selective disconnection syndrome involving arithmetic long term memory (Cohen & Dehaene 1994).
Hence, in many instances a patient may appear to have difficulty performing math problems when in fact the basic ability to calculate per se is intact. That is, the difficulty is due to spatial, linguistic, agnosic or alexic abnormalities. On the otherhand, with left posterior lesions localized to the vicinity of the inferior parietal lobe (or temporal-parietal junction), patients may have severe difficulty performing even simple calculations, e.g. carrying, stepwise computation, borrowing (Boller & Grafman, 2005; Hecaen & Albert, 2003; Watson et al., 2009). When this occurs in the absence of alexia, aphasia, or visual-spatial abnormalities, and is accompanied by finger agnosia, agraphia, and right-left orientation, it is considered part of Gerstmann's syndrome (Gerstmann, 1930). This deficit is the purest form of acalculia and is due to an impairment in the ability to maintain order, to correctly plan in sequence, and thus a loss of the ability to appropriately manipulate numbers. Calculation disturbances will be discussed in greater detail in Chapter 20.
Apraxia is a disorder of skilled movement in the absence of impaired motor functioning or paralysis (Barrett et al., 1998; Buxbaum et al., 1998; Kimura, 1993; Heilman et al. 1982; Heilman, 1993). Usually apraxic patients show the correct intent but perform the movement in a clumsy fashion. Like many other types of deficiencies following brain damage, patients and their families may not notice or complain of apraxic abnormalities. This is particularly true if they're aphasic or paralyzed on the right side. That is, clumsiness with either extremity may not seem significant. Hence, direct examination is necessary to rule out the presence of this problem even in the absence of complaints.
Apraxia is usually mildest when objects are used, and performance deteriorates the most when required to imitate or pantomime the correct action. For example, the patient may be asked to show the examiner, "how you would use a key to open a door", or "hammer a nail into a piece of wood". In many cases the patient may erroneously use the body, i.e. a finger, as an object (e.g. a key). That is, they may pretend their finger is the key (inserting it into a "key hole" and turning it) rather than the finger and thumb holding an imaginary key. Although performance usually improves when they use actual objects (Kimura, 1993; Geschwind, 1965; Goodglass & Kaplan, 1972), a rare few may show the disturbance when using the real object as well (Heilman, 1993).
In addition, patient's with apraxia may demonstrate difficulty properly sequencing their actions. For example, if you were to pretend to place a cigarette and matches in front of the patient and ask them to demonstrate by pantomime how they would light it and take a drag, they may pretend to hold up the match, blow it out, strike it, and then pretend to suck on the cigarrete. That is, they incorrectly sequence a series of acts. However, the individual acts may be performed accurately.
Broadly speaking, there are several forms of apraxia, which like many of the disturbances already discussed may be due to a number of causes or anatomical lesions. These include, Ideational Apraxia, Ideomotor Apraxia, Bucal Facial Apraxia, and Dressing Apraxia. With the exception of dressing apraxia (which is due to a right cerebral injury), apraxic abnormalities are usually secondary to left hemisphere damage, in particular, injuries involving the the left frontal and inferior parietal lobe (see chapters 19, 20).
The inferior parietal lobule IPL appears to be the central region of concern in regard to the performance of skilled temporal-sequential motor acts. This is because the motor engrams for performing these actions appear to be stored in the left angular and supramarginal gyri (Geschwind, 1965; Heilman, 1993; Kimura, 1993). These engrams assist in the programing of the motor frontal cortex where the actions are actually executed.
If the inferior parietal region is destroyed the patient loses the ability to appreciate when they have performed an action incorrectly. If the motor region is destroyed, although the act is still performed inaccurately (due to disconnection from the IPL), the patient is able to recognize the difference (Heilman et al. 1982; Heilman, 1993). Thus, apraxic abnormalities secondary to left cerebral lesions tend to either involve destruction of the inferior parietal lobule or lesions resulting in disconnection of the frontal motor areas (or the right cerebral hemisphere) from this more posterior region of the brain. Apraxia will be discussed in more detail in Chapter 20.
PANTOMIME RECOGNITION
Individuals with left cerebral and inferior parietal lobe lesions not only have difficulty performing motor acts but comprehending, recognizing and discriminating between different types of motor acts performed and pantomimed by others (Bell, 1994; Corina et al. 1992; Heilman et al. 1982; Rothi et al. 1986; Wang & Goodglass, 1992). That is, in the extreme, if one were to pantomime the pouring of water into a glass vs. lighting and smoking a cigarette, these indviduals would have difficulty describing what was viewed or in choosing which was which.
Wang and Goodglass (1992) and Kimura (1993) argue that pantomime imitation and production are related to both apraxia and the ability to interpret purposeful movements; the engrams for which are located in the inferior parietal lobule (Heilman et al. 1982). However, with anterior lesions, although the ability to imitate may be impaired, the ability to comprehend pantomime is retained (Heilman et al. 1982). Nevertheless, some scientists view pantomime and apraxic disorders as strongly related to language processing and aphasia (Bell 1994; Corina et al. 1992; Kertesz et al. 1984). That is, if an individual cannot comprehend what is being asked, or what their own verbal intentions might be, they are going to fail to correctly perform associated movements and will fail to generate the correct verbal label so as to achieve linguistic comprehension as to what is being demonstrated via pantomime. Even so, most investigators are in agreement as to the common source of this deficit, the inferior parietal lobule (Heilman et al. 1982; Wang & Goodglass, 1992).
However, different regions of the brain also interact during various stages of reading, such that in consequence, abnormalities of or lesions to different areas can result in different symptoms, such as an inability to recognize sentences or long words, whereas the ability to recognize letters and short words remains intact. Or, an inability to derive semantic meaning from words once they are read. Or an inability to read words, although spelling ability is intact (Coltheart, 1998; Miceli et al., 2009; Miozzo & Caramazza, 1998; Goodglass & Kaplan, 2000).
For example, injuries the left IPL can disrupt the ability to read and spell. Damage to the adjacent but more anterior-lateral supramarginal gyrus can disrupt the ability to spell by sound and to engage in phonological processing--which is one of the most common correlate of reading disability (Brady & Shankweiler, 2001; Miceli et al., 2009). Injuries to Wernicke's area can disrupt reading, writing, and all aspects of linguistic comprehension (Goodglass & Kaplan, 2000; Sarno, 1998). Lesions to the basal (middle/inferior) temporal lobe can disrupt both reading and naming (Rapcsak, et al., 2007); a condition referred to as phonological alexia and which is also associated with injuries of the supramarginal gyrus. In fact, patients with developmental dyslexia have been found to have abnormalities in the middle/inferior temporal lobe (Rumsey et al., 2007) as well as in the IPL and left superior temporal lobe (Denays et al., 1989; Galaburda et al., 2005); Rumsey et al., 1992; Tzourio et al.,1994).
The IPL, middle temporal lobe, and Wernicke's area are richly interconnected, and in some respects coextensive with Wernicke's area. They are also linked to Broca's area. The IPL in particular evolved as an extension of the superior temporal lobe over the course of the last million years which in turn significantly contributed to the evolution of reading, writing, and most aspects of modern human language.

Hands of an Ape
Presumably the evolution of the capacity to read and write is directly associated with the evolution of the hand and the inferior parietal lobule and is related to tool making, gathering, gestulating and gossiping for over 100,000 years.
The evolution of the IPL/angular gyrus not only made language possible but provided the neurological foundations for tool design and construction, the ability to sew clothes, and the capacity to create art, and pictorial language in the form of drawing, painting, sculpting, and engraving. Finally it enabled human beings to not only create visual symbols but to create verbal one's in the form of written language (Joseph 1993).
It could be said the evolution of the hand, and the capacity to construct tools, has enabled "Man" to conquer the world.


In fact, with the exception of written language, all these abilities, including, perhaps, spoken language, may have appeared essentially within the same time frame, the first evidence of which is approximately 40,000 years old and was left in the deep recesses of caves and rock shelters throughout Europe, Africa, and perhaps Australia (see chapter 6).
Although we have no recordings of what people may have sounded like, or any evidence regarding the presence of grammatical sequences in their speech, complex and detailed paintings left in the deep and forgotten recesses of ancient caves indicates that Homo sapiens sapiens of the Upper Paleolithic were capable of felling stories and making signs which are still comprehensible and pregnant with meaning 40 millinea later.
However, the earliest preserved evidence detailing the evolution of written symbols comes from ancient Sumer, around 6000 or more years ago. In Sumer (a land where ancient Babylon would be established) as elsewhere, the first forms of "writing" were pictorial, a tradition that was already in use at least 20,000 years before their time; i.e. during the Upper Paleolithic.


Like those of the Upper Paleolithic, Sumerians initially utilized single pictorial symbols to convey specific messages, such as a "lion" to indicate "watch out, lions around," or that of a "grazing gazelle" so as to inform others that good hunting could be found in the vicinity (Chiera, 1966; Kramer 1981; Wooley 1965). Although the Cro-Magnons sometimes used single two-step pictorial displays to indicate a sequence of action, as based on available evidence, it was not until the time of the Sumerians and the Egyptians that people began to skillfully employ a series of pictures to represent not just actions, but abstract as well as concrete ideas.
For example, prior to the Sumerians and the rise of the Egyptian civilization, a depiction of a lion or a man might indicate the creature itself, or the nature-God that was incarnate in its form. The Sumerians and Egyptians then took symbolizing to its next evolutionary step. They strung these symbols together.
For example, by drawing a "foot," or a "mouth," or an "eye," they could indicate the idea "to walk," or to "eat," and to "look" or "watch out." By combining these pictures they were able to indicate complex messages, such that "so and so" had to "to walk" to a certain place where "gazelles" might be found in order "to eat," but they would have to keep an "eye" out for "lions" (Chiera, 1966; Kramer 1981; Wooley 1965).
This was a tremendous leap, for earlier in Sumerian and Egyptian civilization, to indicate a complex message such as the above would have required elaborate pictorial detail of entire bodies engaged in particular actions. Although beautiful to behold, and easily understood by all, this was a very cumbersome and time consuming process.
The next step in the evolution of writing was the depiction not just of actions and ideas, but sounds and names. As argued by Edward Chiera (1966), if they wanted to write a common Sumerian name such as "kuraka," they would draw objects which contained the sounds they wanted to depict, such as a "mountain" which was pronounced "kur," and "water" which was read as "a," and then a "mouth" which was pronounced "ka." By combining them together one was able to deduce the sound or name that the writer desired.
This was a tremendous leap in abstract thinking and in the creation of writing, for now visual symbols became associated with sound symbols and one could now not only look at pictures and know what was meant, but what the symbols sounded like as well. In this manner, writing, although still in pictorial form, became much more precise. Now ideas, actions, and words, and thus complex concepts could be conveyed.
Nevertheless, although this was a highly efficient means of communication, it still remained quite cumbersome which required that further steps in symbolic thought be invented. Hence, pictures came to be placed in temporal sequences, and then the pictures were transformed and their transformed images became represented by sequences as well: a series of wedge shaped lines which were read from left to right. That is, the Sumerians invented wedge form cuneiform characters which gradually began to replace the older pictographs and ideographs (Chiera, 1966; Kramer 1981; Wooley 1965) and which were read from left to right.

Initially these characters resembled the pictures they were destined to replace. However, as the use of cuneiform continued to evolve, pictorial details were gradually minimized, until finally these characters lost all pictorial relationship to that which they were meant to describe. Instead purely abstract visual images could now evoke sound images, without the necessity of conjuring forth pictorial images. Reading and writing had thus evolved--a capacity directly related to the evolution of the angular gyrus which enables visual signals to be matched with sounds so that auditory equivalents can be conjured up. In this manner people are able to not only look at a visual symbol, but recognize it as a "word " and know what it sounds like as well.
From a neurodynamic perspective the process of reading involves the reception of visual impulses in the primary and association visual receiving areas where various forms of perceptual analyses are initiated (Peterson, et al., 1988, 1990; Price, 2007). This information is next transferred to the visual association cortex where higher level information processing is carried out and visual associations formed. These visual associations are next transmitted to a variety of areas (Haxby, et al., 2001; Peterson, et al., 1988, 1990; Price, 2007; Zeki, 2007) including Broca's and Wernicke's area, the inferior and middle temporal lobe and the IPL. It is in these latter cortical regions where multimodal and linguistic assimilation take place so that the auditory equivalent of the visual stimulus may be retrieved. That is, via these interactions visual grapheme clusters become translated into phonological sound images (see Barron, 1980; Ellis, 1982; Friederici et al. 1981; Miceli et al., 2009). In this manner we know what a written word looks and sounds like. It is also possible, however, to bypass this phonological transcoding phase so that word meanings can be directly accessed (i.e. lexical reading).
It is generally believed, that in the acquisition of reading ability, graphemes, once recognized are decoded are then converted to phonemes (via the IPL which performs an auditory matching function)), such that the child learns to associate written symbols with spoken words. This requires that language also be broken down into syllables and phonemes so that auditory-visual associations can be made; i.e. "phonological awareness." The greater a child's phonological awareness, the greater is their reading ability (Brady & Shankweiler, 2001). Presumably, it is only after the child develops their phonological awareness, and after increased experience with reading, that they are able to bypass the phonological transcoding phase and engage in lexical reading whereas phonological processing becomes subsumed and employed only secondarily when confronted with unfamiliar words.
Although it is likely that most individuals utilize both lexical and phonological strategies when reading, in either case the temporal-parietal cortex (Rumsey et al. 1992), the lingual, fusiform, and angular and marginal gyrus (Gloning et al., 1968; Greenblatt, 1973; Price, 2007, as well as the middle (basal) temporal lobe are involved (Buchel et al., 1998; Price, 2007; Rapcsak, et al., 2007). These structures all participate, to varying degrees in phonological and lexical processing. Indeed, as demonstrated through functional imaging, reading activates Wernicke's area and the left posterior temporal lobe (Bookheimer, et al., 1995; Howard et al., 199), including the supramarginal gyrus (Bookheimer, et al., 1995), and angular gyrus (Price, 2007). Conversely lesions to these different areas produce alexic disturbances.
For example, with lesions involving the angular gyrus, or when damage occurs between the fiber pathways linking the left inferior parietal lobule with the visual cortex (i.e. disconnection), a condition referred to as Pure Word Blindness sometimes occurs--also referred to as Pure Alexia (Coltheart 1998; Miozzo & Caramazza, 1998). Patients can see without difficulty, but are unable to recognize written language. Written words evoke no meaning. However, because the left posterior basal temporal lobe is a convergence zone for visual, auditory, and tactile information, and becomes activated during a variety of language tasks, including reading and object and letter naming (Buchel et al., 1998; Price, 2007), if injured, patients may suffer from reading and naming deficits (Rapcsak, et al., 2007); a condition referred to as phonological alexia. Moreover, patients with developmental dyslexia have been found to have abnormalities in this latter area (Rumsey et al., 2007).
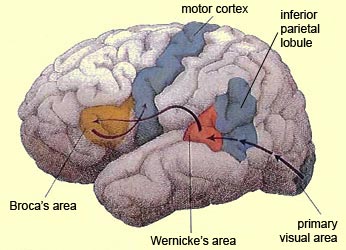
Thus there are several subtypes of reading disturbances which may occur with left cerebral damage or congenital disturbances involving these tissues (Coltheart 1998; Miceli et al., 2009; Miozzo & Caramazza, 1998). These include literal, verbal, and global alexia, and alexia for sentences, as well as developmental dyslexia. In addition, alexia can sometimes result from right hemisphere lesions, a condition referred to as Spatial Alexia. All these disorders, however, are acquired and should be distinguished from developmental dyslexia which is present since childhood (see Njiokiktjien, 1988).
DEVELOPMENTAL DYSLEXIA
Developmental dyslexia is one of the most common of all learning disabilities, effecting up to 8% of the school aged population. There is an obvious genetic foundation to this disorder, as is evident from the high familial risk rates that range from 35% to 45% (Smith et al., 1990) and many of these children suffered from even more profound language disorders during their preschool years, e.g. developmental dysphasia. Developmental dysphasias affect from 8% to 15% of all preschool children and most of these children, even as they acquire language, continue to suffer severe disturbances in reading and spelling (Aram et al. 1984); a function, in part, of not only brain abnormalities, but persisting deficits in phonological processing.
Congenital disturbances involving these tissues (dysplasia, ectopias), including the middle temporal lobes, the perisylvian regions and the planum temporal, are associated with developmental dyslexia (Galaburda et al., 2005). Using functional imaging, Rumsey et al. (1992) and Tzourio et al., (1994) found that dyslexic children failed to activate the left temporal-parietal cortex when engaged in word tasks, whereas Denays et al. (1989) found hypoperfusion in this same area.
PURE WORD BLINDESS. ALEXIA WITHOUT AGRAPHA
Pure alexia, or alexia without agraphia (due to the preservation of the ability to write) is a condition where patients are unable to read written words, or even recognize single letters (Coltheart 1998; Miozzo & Caramazza, 1998). However, if words are spelled out loud, they have little difficulty with comprehension (due to intact pathways from Wernicke's to the angular gyrus). Moreover, they are able to speak, spell as well as write without difficulty. Nevertheless, although able to write, they are unable to read what they have written.
In general, the lesion appears to be between the left angular gyrus and the occipital lobe (in the arterial distribution of the left posterior cerebral artery), and extends to within the splenium of the corpus callosum (Benson, 2012; Vignolo, 2005) which prevents right hemisphere visual input from being transferred to the inferior parietal lobe of the left half of the brain. Sometimes this condition is due to a tumor or following a head injury accompanied by a hematoma involving the white matter underlying the inferior parietal lobule (Greenblatt, 2005) and or the temporal-parietal cortex (Rumsey et al. 1992). In these instances the left angular gyrus is unable to receive visual input from the left and right visual cortex and visual input cannot be linguistically translated. The patient cannot gain access to the auditory equivalent of a written word.
Although unable to read written language, this syndrome is not always accompanied by a visual field defect (hemianopsia). Moreover, objects may be named correctly (Hecaen & Kremimin, 1977). However, patients may suffer from color agnosia (Benson & Geschwind, 1969), i.e. an inability to correctly name colors. There is often (but not always) an inability to copy written material (because of disconnection from the visual areas) and many patients have difficulty performing math problems. In some cases the patient suffers not only pure word blindness but blindness for numbers as well.
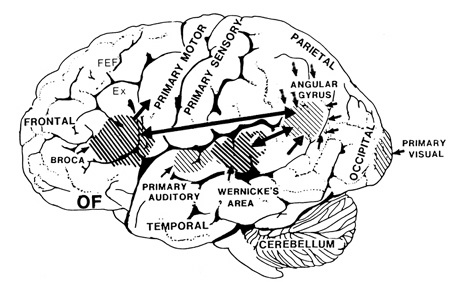
This condition has been described as Global Alexia by some authors. However, global alexia is a more pervasive disorder in which the ability to write (agraphia) and name objects is also compromised.
ALEXIA FOR SENTENCES
Patients suffering from alexia for sentences are able to read letters and single words but are unable to comprehend sentences. However, patients may have difficulty with unfamiliar or particularly long words whereas more familiar material is easily understood. Lesions are usually localized within the dominant occipital lobe but may extend into the inferior parietal area.
VERBAL ALEXIA
When patienta are able to read and recognize letters but are unable to comprehend or recognize whole words they are said to be suffering from verbal alexia. However, if presented with short words their reading ability improves. Hence, the longer the word the greater the difficulty reading (Hecaen & Albert, 2003). Verbal alexia usually results from lesion involving the dominant medial occipital lobe (Hecaen & ALbert, 2003; Hecaen & Kremin, 1977).
LITERAL/FRONTAL ALEXIA
When the patient is unable to recognize or read letters this is referred to as Pure Letter Blindness. Pateints are usually unable to read by spelling a word out loud, and the abilty to read numbers and even musical notion is often disturbed. Some authors have attributed literal alexia to left inferior occipital lesions (Hecaen & Kremin, 1977).
This disorder has also been referred to as Frontal Alexia. This is because individuals with Broca's aphasia or lesions involving the left frontal convexity have difficulty reading aloud, and have the most difficulty with single letters rather than whole words (Benson, 1977).
SPATIAL ALEXIA
Spatial alexia is associated predominantly with right hemisphere lesions. In part this disorder is due to visual-spatial abnormalities including neglect and inattention. That is, with right cerebral lesions the patient may fail to read the left half of words or sentences, and may in fact fail to perceive or respond to the entire left half of a written page.
Right posterior injuries may also give rise to spatial disorientation such that the patient is unable to properly visually track and keep place, their eyes darting half hazardly across the page. For example, they may skip to the wrong line. Spatial alexia may also result from left cerebral injuries in which case it is the right half of letters, words and sentences which are ignored (Marshall & Newcomb, 1973).
Regardless of which theory one adheres to, it appears that the angular gyrus plays an important role in writing. Presumably the angular gyrus provides the word images (probably via interaction with Wernicke's area) which are to be converted to graphemes. These representations are then transmitted to the left frontal convexity (i.e. Broca's and Exner's area) for grapheme conversion and motoric expression, However, it is possible that the initial graphemic representations are formed in the inferior parietal lobule (Roeltgen et al. 2005).
Nevertheless, it appears that there are at least two stages involved in the act of writing, a linguistic stage and a motor-expressive-praxic stage. The linguistic stage involves the encoding of information into syntactical-lexical units (Goldstein, 1948). This is mediated through the angular gyrus which thus provides the linguistic rules which subserve writing. The motor stage is the final step in which the expression of graphemes is subserved. This stage is mediated presumably by Exner's writing area (located in the left frontal convexity) in conjunction with the inferior parietal lobule.
Hence, disturbances involving the ability to write can occur due to disruptions at various levels of processing and expression and may arise secondary to lesions involving the left frontal or inferior parietal cortices. Thus, similar to alexia, there are several subtypes of agraphia which may become manifest depending upon which level and anatomical region is compromised. These include Frontal Agraphia, Pure Agraphia, Alexic Agraphia, Apraxic Agraphia, and Spatial Agraphia.
EXNER'S WRITING AREA
Within a small area along the lateral convexity of the left frontal region lies Exner's Writing Area (see Hecaen & Albert, 2003). Exner's area appears to be the final common pathway where linguistic impulses receive a final motoric stamp for the purposes of writing. Exner's area, however, also seems to be very dependent on Broca's area with which is maintains extensive interconnections. That is, Broca's area acts to organize impulses received from the posterior language zones and relays them to Exner's area for the purposes of written expression.

Exner's Area (EX) is above Broca's Area and adjacent to the motor area
Lesions localized to this vincinity have been reported to result in disturbances in the elementary motoric aspects of writing, i.e. Frontal Agraphia (Penfield & Roberts, 1959), sometimes referred to as Pure Agraphia. However, Pure Agraphia has also been attributed to left parietal lesions (Basso et al. 2003; Strub & Geschwind, 2005). In general, with frontal agraphia, grapheme formation becomes labored, incoordinated, and takes on a very sloppy appearance. Cursive handwriting is usually more disturbed than printing. The ability to spell, per se, may or may not be effected, whereas with parietal lesions spelling is often abnormal. Rather, there are disturances in grapheme selection and the patient may seem to have "forgotten" how to form certain letters, and/or they may abnormally sequence or even add unnecessary letters when writing (see Hecaen & Albert, 2003).
With severe damage, the left hand is more effected than the right (due to disconnection from the left hemisphere Language Axis). However, with either hand patient's may not be able to write or spell correctly even if given block letters.
In addition, Frontal Agraphia can result from lesions involving Broca's area. These individuals are unable to write spontaneously or to dictation, and when they do write their samples may be contaminated by perseverations or the addition of extra strokes to letters (e.g. such as when writing an "m"). Often such patients are able to write their name or other well learned (automatic) items without difficulty. Hence, when tested they should be required to write spontaneously as well as to dictation.

PURE AGRAPHIA
In pure agraphia, patients have difficulty in the motor control of grapheme selection and production, frequently misspell words, and may insert the wrong letters or place them in the wrong order or sequence when attempting to write. (Marcie & Hecaen, 2012) In contrast, reading, oral speech and the ability to name objects or letters are usually unimpaired.
Most commonly, pure agraphia is associated with lesions involving the superior and mid parietal regions of the left hemisphere; areas, 5 and 7 (Basso, et al. 2003; Vignolo, 2005), and/or the inferior parietal region (Strub & Geschwind, 2005). Patients are unable to write because the area involved in organizing visual-letter organization (i.e. the inferior parietal lobe) is cut off from the region controlling hand movements in the frontal lobe (Strub & Geschwind, 2005). However, according to Vignolo (2005), the posterior superior left parietal lobule (area 7) is crucial for the sensorimotor linguistic integration needed for writing.
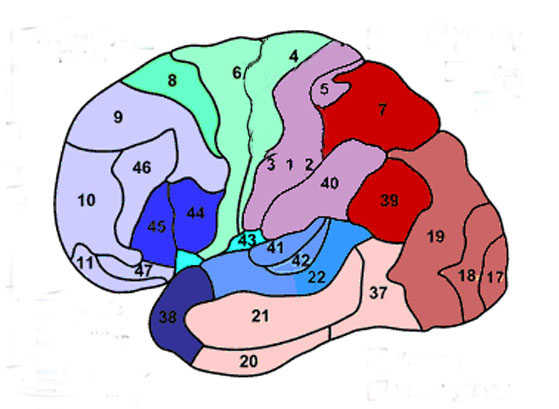
ALEXIC-AGRAPHIA
Alexic agraphia is a disturbance involving the ability to read and to write. It is a disruption of both the ability to decode and to encode written language. However, reading and writing may not be equally affected. Usually recognition of letters is better than words, and the longer the word, the greater is the difficulty with recognition. Patients have severe difficulty forming graphemes and spelling, even when block letters are employed (Marcie & Heilman, 2012). Although words are misspelled, syntactical sequencing is preserved such that what is written remains grammatically correct. Nevertheless, patients often display some degree of anomia and apraxia (due to inferior parietal involvement). Verbal-auditory comprehension is generally intact as is expressive speech.
Often alexia with agraphia is due to a lesion involving the left inferior parietal lobule and angular gyrus (Benson & Geschwind, 1969; Hecaen & Kremin, 1977), and for this reason has also been called parietal alexia. Hence, because the angular gyrus is destroyed there results a massive disconnection such that the auditory areas cannot completely communicate with the visual areas. Nor can the frontal areas interact with the inferior parietal lobe. There is thus two separate disconnections. Patients are unable to correctly perceive visual- linguisitic symbols and are unable to reproduce them; i.e. they cannot read or write.
APRAXIC AGRAPHIA
It has been argued that the sensory motor engrams necessary for the production and perception of written language are stored within the inferior parietal lobule of the left hemisphere (Strub & Geschwind, 2005). When this region is damaged, patients sometimes have difficulty forming letters due to an inability to access these engrams (Strub & Geschwind, 2005). Writing samples are characterized by misspellings, letter omissions, distortions, temporal-sequential misplacements, and inversions (Kinsbourne & Warrington, 1964). This type of agraphia is often seen in conjunction with Gerstmans syndrome (to be explained).
Because the patient is also apraxic, the ability to make gestures or complex patterned movements such as those involved in writing is also deficient. That is, the ability to correctly temporally sequence hand movements, independent of writing, is affected (see section on Apraxia). The patient no linger knows how to correctly hold or manipulate a pen, or how to move their hand when writing.
SPATIAL AGRAPHIA
Right cerebral injuries can secondarily disrupt writing skills due to generalized spatial and constructional deficiencies. Hence, words and letters will not be properly formed and aligned even when copying. There may be difficulty keeping lines straight and letters may be slanted at abnormal angles. In some cases the writing may be reduced to an illigible scrawl. In addition, patients may write only on the right half of the paper such that as they write, the left hand margin becomes progressively larger and the right side smaller (Hecaen & Marie, 1974). If allowed to continue patients may end up writing only along the edge of the right hand margin of the paper.
Patients with right hemisphere lesions may tend to abnormally segment the letters in words when writing cursively (i.e. cu siv e ly ). This is due to a failure to perform closure as well as a release over the left hemisphere (i.e. left hemisphere release). That is, the left acting unopposed begins to abnormally temporally-sequence and thus produce segments unnecessarily. It is also important to note that Hecaen and Marcie (1974) report that the insertion of gaps or blanks into words can also occur following left hemisphere lesions.
APHASIA & AGRAPHIA
Although every patient with agraphia does not necessarily suffer from aphasic abnormalities, every patient who has aphasia has some degree of agraphia. In some instances, the aphasia may be mild and the agraphic disturbance may be severe or conversely quite subtle and only revealed through special testing. In general, it is best to require the patient to write cursively rather than to print. Moreover, the more complex the writing task, the more likely will an agraphic disturbance be demonstrated. Hence, in testing one should not be satisfied with the writing of simple words or sentences if agraphia is truly to be ruled out.
The left hemisphere, therefore, serves all aspects of the conscious mind associated with language. Thinking in words, reading, writing, talking, and comprehending the verbal, numeral, and temporal sequential aspects of reality, including control over the right hand and right half of the body, are all the domain of the left half of the brain.
When the left half of the brain has been injured, not just the verbal aspects of consciousness, but the linguistic aspects of emotion can also be disrupted. Patients may become profoundly depressed, or develop severe psychotic disorders, also known as "formal thought" disorders, and appear to be suffering from schizophrenia. In many cases, instead of a mental breakdown, the patient has instead suffered a severe injury to the left half of the brain, which normally controls all aspects of language-dependent conscious experience.



