Rhawn Gabriel Joseph, Ph.D.

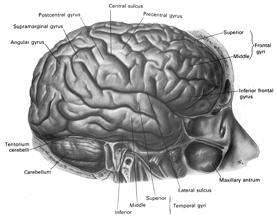
The functional neuroanatomy of the human brain is functionally localized, such that specific areas of the brain subserve specific functions. For example, if you were to suffer a severe injury to the left frontal-temporal region of your head and brain, you might lose the ability to talk. This is because, the left frontal tissues known as Broca's Area mediates the expression of speech. However, you would still be able to swear and sing because these abilities are mediated by the right frontal-temporal region of the brain
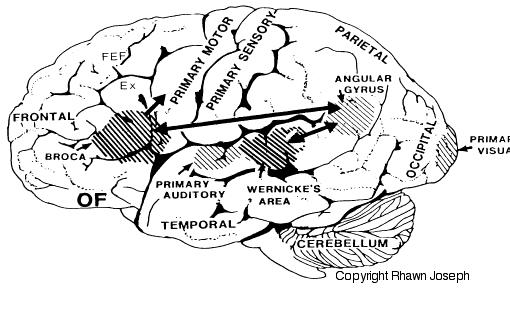
If instead, you suffered a massive stroke to the left temporal lobe (the brain area inside your skull near your ear), you would be unable to comprehend human speech. You would not understand what is being said to you. This is because the left temporal region, known as Wernicke's Area, subserves the ability to understand language.
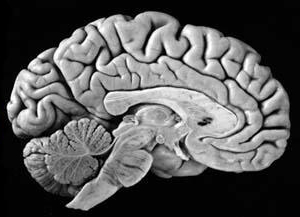

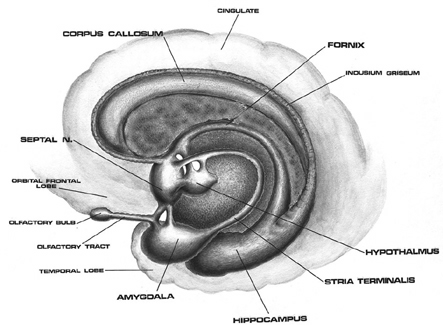
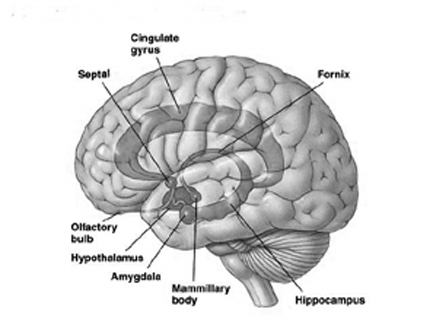
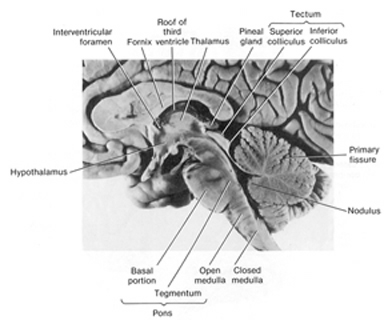
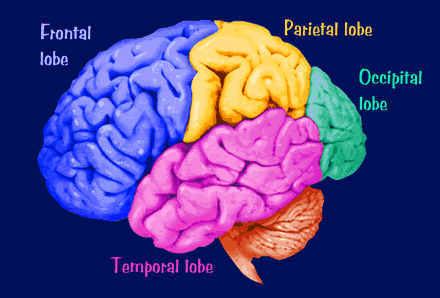
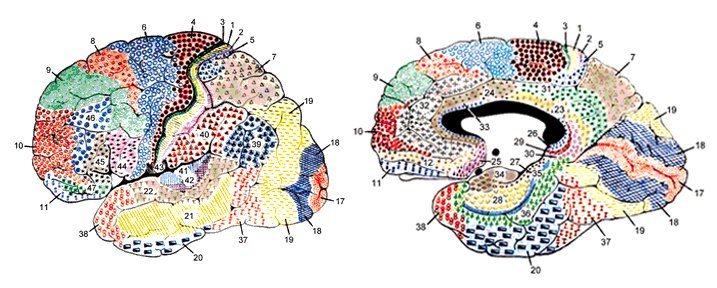
The human brain can be broadly described as consisting of 1) two cerebral hemispheres which are divided into frontal, parietal, temporal, and occipital lobes, 2) "gray matter" which covers the surface of the brain, and which consists of 6-7 major layers and several sublayers of specialized neurons, 3) subcortical structures consisting of 2-5 layers, and which include the striatum (caudate, putaman, globus pallidus), limbic system (hypothalamus, amygdala, hippocampus, septal nuclei, cingulate gyrus), thalamus, cerebellum, and brainstem (midbrain, pons, medulla).


The human is a heterogeneous composite of functionally specialized areas, structures, and subdivisions, many of which maintain multiple topological maps of the body and visual, auditory, somesthetic, olfactory, attentional, or personal space (Barbur et al., 2013; Graziano, et al., 1999; Joseph, 2006a, 1999a; Kaas, 2013; Lin et al., 1994; Passingham, 2012; Zeki, 2012). Consciousness, personal identity, and the capacity to experience love and hate, is made possible through these semi-independent mosaics of the mind, all of which perform unique and overlapping interactive functions such as those involving language, learning, and memory (e.g. Buchel, et al., 1999; Brewer et al., 1998; Frost, 1999; Gloor, 2012; Ojemann, 2001; Pujol et al., 1999; Wagner et al., 1998), including the maintenance and preservation of life.
The brain and the mind are synonymous and are hierarchically, vertically, and horizontally organized, with different regions engaging in both parallel and localized processing, with specialized neurons performing discrete functions, such as face recognition, and others participating in large neural networks to make complex cognitive functioning processing possible, including, for example, language and consciousness.
There are tissues of the mind which mediate language and complex cognition, such as the neocortex and lobes of the brain (Buchel et al., 1998; Evers et al., 1999; Frost et al., 1999; Price, 2012; Pujol, et al., 1999), those aspects associated with sex, love and war, such as the limbic system (Joseph, 1992a, 1994, 1998a, 1999b), and those areas which respond reflexively and completely unconsciously, laboring to maintain homeostasis, and the rhythmics of waking, sleeping, dreaming, and of the heart, the respiratory systems, and the vegetative mind. These latter unconscious activities are associated with the functional integrity of the brainstem (Aminoff, 1996; Blessing, 2012; Joseph, 1999c; Steriade & McCarley, 2010; Vertes, 2010), i.e., the medulla, pons, and midbrain, and to a much lesser extent, the adjacent diencephalon, i.e. the thalamus and hypothalamus.
The mind and brain are also functionally lateralized, with specific interactive subareas in the left hemisphere mediating the linguistic, temporal, sequential, arithmetical, and rhythmical aspects of consciousness and verbal thought (Buchel et al., 1998; Evers et al., 1999; Frost et al., 1999; Peterson et al., 2008, 2010; Price, 2012; Pujol, et al., 1999). It is the right half of the brain which subserves and provides the foundations for an interactive non-verbal awareness encompassing music, emotion, the body, prosodic language, and spatial thought (Joseph, 1982, 2008ab, 1999a; Parsons & Fox, 2012; Price, 2012; Ross, 2013).
Thus, the cerebrum and psyche are organized such that several semi-independent mental system coexist, literally one on top of the other, side by side, distributed and localized, with the limbic system and brainstem providing much of the "food for thought" that is received by the neocortex and reflected upon by the conscious mind.
THE BRAINSTEM
The brainstem is exceedingly ancient and complex structure (Aitkin, 2006; Vertes, 2010), and is concerned with sensory reception, arousal, and reflexive movements of the body, including stereotyped and routine motor acts such as yawning, opening and closing the mouth, wiping the face, or lifting the legs, taking steps, walking or running (Aminoff, 1996; Blessing, 2012; Cohen, et al. 2008; Klemm, 2010; Skinner & Garcia-Rill, 2010). Although capable of learning, forming simple memories, and displaying synaptic plasticity (Jones & Pons, 1998; Isukahara, 1985; Moore & Aitkin, 1975; Moore & Irvine, 1981), this portion of the brain cannot think, reason, or feel love or sorrow (Joseph, 1999c).
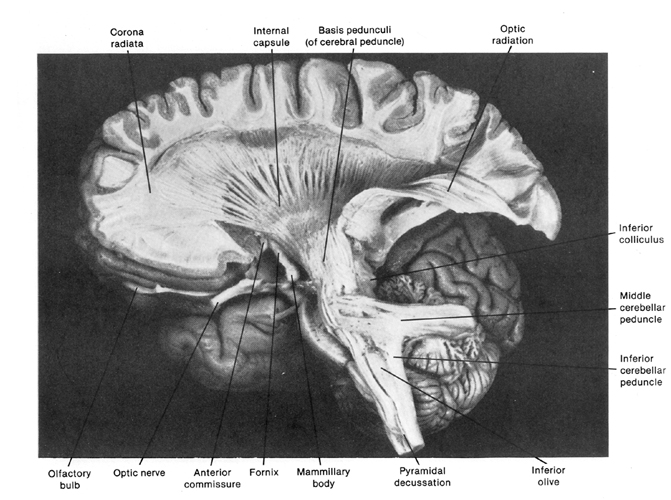
The brainstem is both diffusely organized and functionally specialized. When specific brainstem nuclei (and associated neural networks) are stimulated, sucking, chewing, swallowing, swimming, stepping, walking, and running movements can be induced (Barman, 2010; Cohen, et al. 2008; Klemm, 2010; Mogenson, 2010; Steriade & McCarley, 2010; Skinner & Garcia-Rill, 2010; Vertes, 2010). However, these movements are hierarchically organized with those requiring the least amount of organizational or conscious control being localized near the medulla-spinal border. It is noteworthy, however, that this areas also contains neurons which when stimulated can trigger female sexual posturing (Benson, 2008; Rose, 2010); i.e. the lordosis (or "doggie") position. These latter neurons are interconnected with the amygdala and ventromedial hypothalamus--nuclei which are also involved in sexual activities. At the level of the spinal cord, movement programs are exceedingly simplified and usually consist of only fragments of the entire motor display.
As one ascends from the spinal cord to the medulla, then the pons, and finally the midbrain, the degree, extent, and nature of these various motor programs and behaviors becomes increasingly complex. For example, when the most caudal regions of the medulla are stimulated stepping motions can be induced, whereas stimulation of the midbrain can initiate eye movements, head turning, controlled walking, running (Cowie & Robinson 1994; Cowie et al. 1994; Skinner & Garcia-Rill, 2010) and vocalization--which is produced by the periaqueductal gray (Jurgens, 1994; Larson et al. 1994; Zhang et al. 1994).
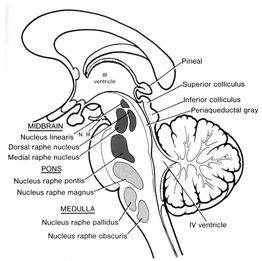
The periaqueductal gray coordinates the activity of the laryngeal, oral-facial, and principal and accessory muscles of respiration and inspiration (Zhang et al. 1994) and receives extensive input from the amygdala, the anterior cingulate, and the left and right frontal lobes--structures which are all implicated in vocalization, and/or speech and language. However, it is the coordinated activity of the periaqueductal gray which enables an individual to laugh, cry, or howl, even if the rest of the brain (excepting the brainstem) were dead

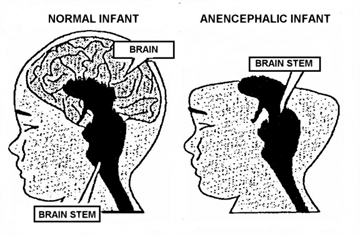
The brainstem is devoid of cognitive activity as it is designed to react immediately and reflexively to sensory stimuli. Consider, for example, infants born with only a brainstem, i.e. anencephalics, who are completely devoid of any semblance of conscious or cognitive activity; and the same is true of adults who suffer a brainstem-forebrain transection. Although they live and breath, these latter unfortunate souls are essentially forebrain dead.
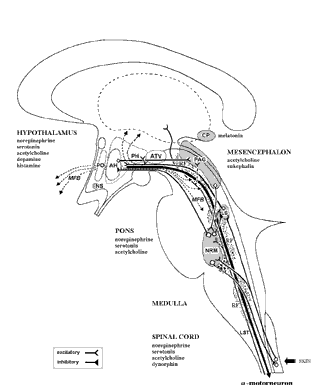
The brainstem, although almost wholly unconscious, maintains consciousness and promotes cognitive activity by feeding the forebrain with activating neurotransmitters such as norepinephrine (NE) Serotonin (5HT), and dopamine (DA) and by controlling arousal through the reticular activating system (Blessing, 2012; Steriade & McCarley, 2010; Usher et al., 1999). Because of its importance in these and other life-preserving functions, the stereotypical consequences of brainstem injury, therefore, are prolonged unconscious and death.
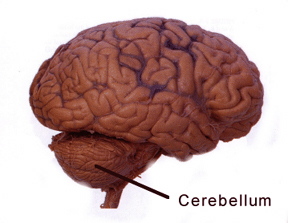
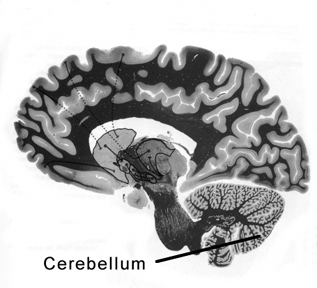
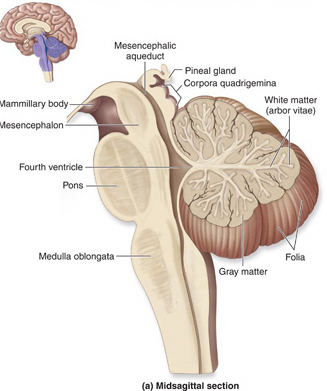
THE CEREBELLUM
The cerebellum sits atop the brainstem and accounts for approximately 25% of the brain. It communicates with almost all regions of the neuroaxis, with the single exception of the striatum, and has been implicated in cognitive, emotional, sensory, motor and speech processing (Llinas & Sotelo, 1992; Schmahmann, 2012; Silveri et al. 1994; Thach, 2012; van Dongen et al. 1994; Wallesch & Horn 2010) . The cerebellum has been shown to display neuroplasticity (Nimura et al., 1999) and learning and memory (Fiez et al. 1992; Lavond et al. 2010; Molinari, et al., 2012; Thompson et al., 2012) and may well serve as an integrative interface for cognition, emotion, motor functioning and memory.
The cerebellum is typically thought of as a motor center. However, electrical-electrode stimulation or damage to this structure can trigger rage reactions (Bharos, et al. 1981), and hyperactivity (Carpenter, 1959), including "mania" (Cutting, 1976). Abnormalities in the cerebellum have also been implicated in the pathogenesis of schizophrenia and autism (Bauman & Kemper, 1985; Courchesne & Plante, 1996; Gaffeny, et al. 1987; Heath, 1977; Heath, et al. 1979, 1982; Taylor 2001; Weinberger et al. 1979, 1980). Although the notion that abnormal rearing conditions may contribute to autistic and schizophrenic behavior is no longer in fashion, it is noteworthy that Heath (1972) found that monkeys reared under deprived conditions displayed abnormal electrophysiological activity in the cerebellum (dentate gyrus) as well as the septal nuclei. These animals also displayed autistic behavior (Harlow & Harlow, 1965a,b). These findings are significant, for the cerebellum is an outgrowth of the vestibular system, and insufficient social-emotional or physical stimulation would also result in insufficient vestibular activation.
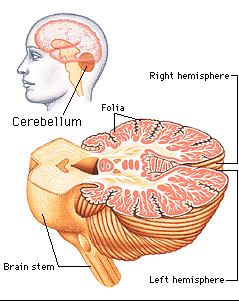
The cerebellum consists of a number of structures and distinct neural circuits associated with specific fiber systems, including climbing, parallel and mossy fibers. It also appears that different regions are concerned with different functions that also have a major motor component. These functions include speech (Silveri et al. 1994; van Dongen et al. 1994; Wallesch & Horn 2010), and visual processing, including the visual guidance of movement (Bloedel 1992; Stein & Glickstein 1992).
The cerebellum is tonically active, and presumably exerts a tonic and stabilizing influence on motor function (Llinas, 1981). Moreover, by altering its activity (e.g., Bloedel, et al., 1985; Thach, 1978), it can apparently coordinate, smooth, fine tune, as well as exert a timing influence on motor movements (Ivry, 2012; Llinas, 1981; Llinas & Sotelo, 1992). In fact, some cerebellar neurons become activated just thinking about making a movement (e.g., Dacety et al. 2010). Indeed, the cerebellum is associated not just with motor functioning, but classical conditioning and the learning of new motor programs (Llinas & Sotelo, 1992; Schmahmann, 2012).

For example, it has been suggested the initial acquisition of skilled movements, such as playing a guitar, requires neocortical and conscious control over motor functioning, with the cerebellum playing at best a minimal supplementary role. However, "practice makes perfect" and presumably the cerebellum immediately begins to increase its participation and slowly begins to learn the necessary movements. For example, the cerebellum becomes activated during initial learning stages (Watanabe, 1984) and as learning progress, the cerebellum may begin to acquire control over the task and may begin to associate the task and specific movements with specific changing contexts so that each context automatically triggers the movement (Thach, 2012). With time and practice, the cerebellum may slowly assume control over the associated movements, which become "automatic" and can then perform these movements with little or no help from the cerebrum which becomes free to do and think about other things.
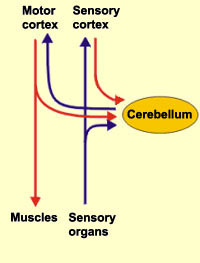
Conversely, lesions abolish the acquisition and retention of conditioned responses (Lavond et al. 2010), and compound movements are more severely effected that simple movements. These and other findings suggests that the cerebellum may act to integrate and combine different movements, and movement sequences. Moreover, it has been proposed that climbing fibers may act to learn the task, mossy fibers may learn the "context," whereas parallel fibers integrate the context with the actual motor activity, and even correcting errors (Thach, 2012).
THE DIENCAPHALON
The Diencephalon consists of the hypothalamus and thalamus.
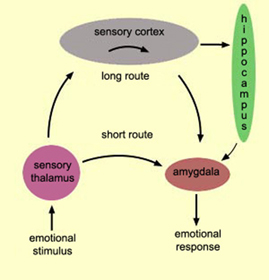
With the notable exception of olfaction (Gloor, 2012), all sensory input is first projected to the brainstem (Blessing, 2012; Vertes, 2010) and is then relayed to the immediately adjacent thalamus and hypothalamus--collectively referred to as the diencephalon ("between brain"). The diencephalon represents that rudimentary aspect of the unconscious mind that generates vague sensory impressions and diffuse emotions (Dreifuss, Murphy, & Gloor, 1968; Joseph, 1992a; Olds, 1956), including pain (the thalamus), and hunger, thirst, sexual arousal, or depression and rage (the hypothalamus).
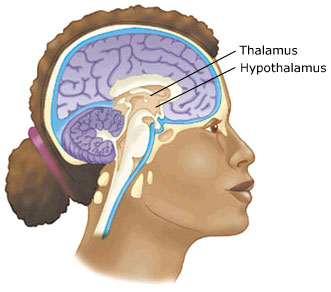
Like the brainstem, the hypothalamic portion of the diencephalon does not think or reason, but reflexively reacts--often in response to amygdala input, in which case it remain active for an extensive period of time (Dreifuss, et al., 1968; Rolls 1992). Nor are the emotions generated by this portion of the brain well differentiated. The hypothalamus may feel pleasure in general, or depression in general, or enraged in general with no differentiation, specificity, or concern for consequences other than the satisfaction of internal needs. .
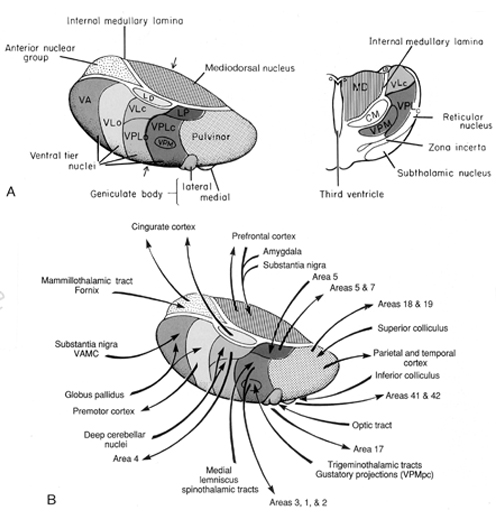
By contrast, considerable information and cognitive pre-processing occurs within the various subdivisions of the thalamus, such as the the lateral and medial geniculate nucleus (LGN & MGN), the pulvinar, the motor and subthalamus (M/ST), and the dorsal medial nucleus (DMN). These structures play a significant role in processing auditory (MGM) and visual input (LGN), the guidance of motor functions (M/ST), and the regulation of attention and arousal (DMN). For example, the LGN receives visual input directly from the retina, and then transfers this information to the visual cortex (Casagrande & Joseph, 1978). The MGN receives auditory input from the midbrain and transfer this information to the auditory cortex (Amaral et al., 1983; Pandya & Yeterian, 1985). The dorsal medial and reticular thalamus exert regulatory influences over neocortical, striatal, and limbic system arousal and influence memory and attentional functioning (Joseph, 1999a; Skinner & Yingling, 1977; Yingling & Skinner, 1977). .
In addition, the subthalamus, which is intimately associated with the amygdala, the motor thalamus and the striatum, participates in the organization and expression of gross purposeful affective-motoric behaviors (Crossman, Sambrook, & Mitchell, 1987; Parent & Hazrati, 1995). Thus the subthalamus (and striatum with which it is also intimately associated) can trigger running, kicking, punching, flailing, and a variety of oral and emotional facial expressions, or conversely "freezing" in reaction to extreme fear. The striatum and subthalamus act as an emotional-motor interface which enables humans (and other animals) to express their emotions through body language and facial expression.
Because of their role in guiding and controlling motor activities, if the subthalamus or motor thalamus is injured, patients may demonstrate a variety of hypo- or hyperactive motor abnormalities including rigidity, catatonia, and catalepsy, or conversely tremor and uncontrolled ballistic movements such as kicking, flailing, and so on (Crossman, et al., 1987; Parent & Hazrati, 1995; Royce, 1987).
Although capable of experiencing pain, the mental and perceptual functioning of the thalamus occurs outside of conscious awareness. Rather, the functioning of these various thalamic subdivisions occurs in a mental realm best described as the "preconscious;" acting to provide the conscious mind with its sensory and perceptual contents by relaying data from the brainstem to the neocortex as well as to the limbic system.
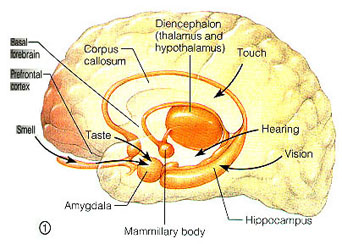
THE LIMBIC SYSTEM
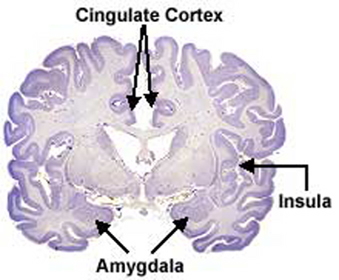
The principle structures of the limbic system include the amygdala, hippocampus, septal nuclei, anterior cingulate gyrus, as well as the hypothalamus (Joseph, 1992a, 1994, 1998a; LeDoux, 1996; Gloor, 2012; MacLean, 2010). These structures represent a truly interactive system and are intimately interconnected by a number of interactive pathways, e.g. the stria terminalis, medial forebrain bundle, fornix-fimbria, amygdalofugal, and so on.


Collectively, the limbic system subserves all aspects of emotional, social, motivational and sexual functioning (Gloor, 2012; Halgren, 1992; Leutmezer et al., 1999; MacLean, 2010; Olds, 1956; Olds & Forbes, 1981), as well as learning and memory (Eichenbaum et al. 1994; Mishkin, 2010; Nunn et al., 1999; Squire, 1992), and homeostatic, endocrine, and hormonal activities (Smith et al. 2010), including the stress response (Fink, 1999; Joseph, 1998b, 1999b,d), and even the craving for pleasure-inducing drugs (Childress, et al., 1999). With the exception of the hippocampus, electrical stimulation of each of these structures has induced feelings of extreme pleasure, and extremely negative emotions, such as fear, anger, and rage (Gloor, 2012; Heath, 1974; Olds, 1956; Olds & Forbes, 1981). Activation of these structures, the amygdala and hypothalamus in particular, have also induced fighting, fleeing, and sexual behavior--affective motor actions made possible through the basal ganglia (e.g., corpus and limbic striatum) and the brainstem; structures which are partly under the control of the amygdala.

The limbic system, the amygdala and cingulate in particular, also play significant and important roles in the evolution, development and expression of language and emotional speech (Joseph, 1982, 1992a, 1999b; MacLean, 2010). It is these early maturing structures which provide the neurological foundations (that is, in association with the periaqueductal gray) for the development of infant speech and what has been termed, "limbic language" (Joseph, 1982, Jurgens, 2010).
LIMBIC SYSTEM SEXUALITY
The limbic system is sexually differentiated such that there is a male, female, and even homosexual limbic system. In humans, sexual differentiation is initiated around 3 months after conception, and is triggered by the presence or absence of testosterone which also effects cellular development. For example, glia cells, which manufacture certain neurotransmitters and which nourish and even guide immature migrating immature neuroblasts to their terminal substrate, develop unique male-specific patterns before birth. Hence, testosterone effects neural migration and thus the organization and neural growth of the limbic system as well as the neocortex and spinal cord.
For example, the presence of fetal testosterone promotes the development of spinal motor neurons which project to the penis. Moreover, the total brain volume of the human male is about 7% larger than that of the female, and much of this differences is due to the greater volume of white matter in the male cerebrum (glia and axons), the only exceptions being the human hippocampus which is larger in the female, and the amygdala which is 16% larger in the male in total volume (Filipek, et al., 1994).
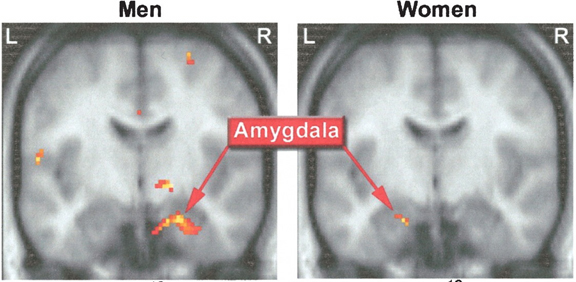
The female and male primate amygdala are sexually differentiated and have their own unique patterns of dendritic growth and organization (Nishizuka & Arai, 1981). As noted, in humans the male amygdala is 16% larger, and in male rats the medial amygdala is 65% larger than the female amygdala (Breedlove & Cooke, 1999), and the male amygdala grows or shrinks in the presence of testosterone--findings which may be related to sex differences in sexuality and aggression.
Moreover, female amygdala neurons are smaller and more numerous, and densely packed than those of the male (Bubenik & Brown, 1973; Nishizuka & Arai, 1981), and smaller, densely packed neurons fire more easily and frequently than larger ones--which may be related to the fact that females are more emotional and more easily frightened than males (chapters 7,13,15), as the amygdala is a principle structure involved in evoking feelings of fear (Davis et al., 2012; Gloor, 2012; LeDoux, 1996).
Dendritic spine density in the female rat hippocampus also increases and decreases by as much as 30% during each estrus cycle (Woolley, et al., 2010) which in turn may influence memory. Indeed, in humans it has been shown that estrogen replacement therapy slows memory loss in women. In fact, it has been reported that women who take this hormone have a 54% lower chance of developing Alzheimers (see chapter 16). On the other hand, dendritic spine density can rapidly change within a few seconds (regardless of gender), as these spines can rapidly grow or disappear in response to varying experiences or lack thereof.
In addition, the human anterior commissure which connects the right and left amygdala/temporal lobe is sexually differentiated (Allen et al. 1989), as is primate/mammalian hypothalamus (Bleier et al. 1982; Dorner, 1976; Gorski et al. 1978; Rainbow et al. 1982; Raisman & Field, 1971, 1973)--with which the amygdala is intimately interconnected. That is, the anterior commissure is thicker in women which, coupled with her more densely packed amygdala neurons (Bubenik & Brown, 1973; Nishizuka & Arai, 1981) may account for her greater social-emotional sensitivity (see chapters 8, 10, 13, 15).
Thus, different structures of the limbic system have sex specific patterns of neuronal and dendritic organization and perform different functions depending on if one is a man or a woman.
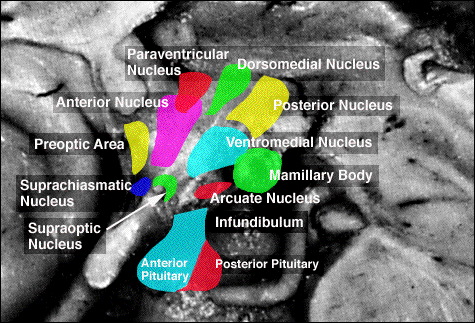
For example, chemical and electrical stimulation of the sexually dimorphic preoptic and ventromedial hypothalamic nuclei triggers and/or increases sexual behavior in males and females (with each taking their respective sexual positions), and significantly increases the frequency of erections, copulations and ejaculations, as well as pelvic thrusting followed by an explosive discharge of semen even in the absence of a mate (Hart et al., 1985; Lisk, 1967, 1971; Maclean, 1973). In female primates, activation of these areas can also trigger maternal behavior (Numan, 1985). Conversely, lesions to the preoptic and posterior hypothalamus eliminates male sexual behavior and results in gonadal atrophy.
Likewise, activation of the sexually dimorphic amygdala--which is larger in males (Filipek, et al., 1994)-- can produce penile erection and clitoral engorgement (Kling and Brothers, 1992; MacLean, 2010; Robinson and Mishkin, 1968; Stoffels et al., 1980), and trigger sexual feelings (Bancaud et al., 1970; Remillard et al., 1983), extreme pleasure (Olds and Forbes, 1981), memories of sexual intercourse (Gloor, 2006), as well as ovulation, uterine contractions, lactogenetic responses, and orgasm (Backman and Rossel, 1984; Currier, Little, Suess and Andy, 1971; Freemon and Nevis,1969; Warneke, 1976; Remillard et al., 1983; Shealy and Peel, 1957).
Moreover, these sexually dimorphic structures also play different roles among females depending on if a woman is sexually receptive, pregnant, or lactating. For example, in a lactating female, the sexually dimorphic supraoptic and paraventricular hypothalamic nuclei (which projects to the posterior lobe of the pituitary) may trigger the secretion of oxytocin--a chemical which can trigger uterine contractions as well as milk production and which makes nursing a pleasurable experience. In fact, dendritic spine density of ventromedial hypothalamic neurons varies across the estrus cycle (Frankfurt et al., 2010) and thus presumably during pregnancy and while nursing.
Hence, the core of our personal and emotional being, the limbic system, is sexually differentiated. There is a male vs a female limbic system, and even a "homosexual" limbic system (Levay, 2001; Swaab, 2010); structures which are organized in unique sex specific dendritic and neuronal patterns and which govern sex-specific behaviors. Coupled with evolutionary (Joseph, 1999e) and early environmental influences (Joseph, 1979; Joseph & Gallagher, 1980), the sex differences in these and other structures account for many of the stereotypical sex differences in thinking, sexual orientation, aggression, and cognitive functioning (Barnett & Meck, 2010; Beatty, 1992; Dawson et al. 1975; Harris, 1978; Joseph, et al. 1978; Stewart et al. 1975) which characterized the mind of woman and man, including their sexual behaviors.
SOCIAL BEHAVIOR & THE LIMBIC SYSTEM
The amygdala are involved in all aspects of social and emotional functioning. If the right and left amygdala are destroyed, the neocortex will be denied all related social-emotional and affective input and the patient will no longer be able to recognize or feel affection for family, friends or loved one's (Lilly et al., 1983; Marlowe et al., 1975; Terzian & Ore, 1955). Although the ability to speak, think, reason, and read and write is preserved, the personal-affective contents of consciousness will have been erased.
Humans and animals subject to bilateral amygdala destruction avoid all contact with others, preferring to sit alone in isolation, and will withdraw if approached. Similarly, primates who are subject to bilateral amygdala removal lose all interest in social activity and persistently attempt to avoid contact with others. If approached they withdraw. If followed they flee. Even maternal behavior is severely affected following bilateral amygdala destruction. According to Kling (1972) mothers will bite off fingers or toes, break arms or legs, and behave as if their "infant were a strange object to be mouthed, bitten and tossed around as though it were a rubber ball".
LIMBIC SYSTEM MEMORY
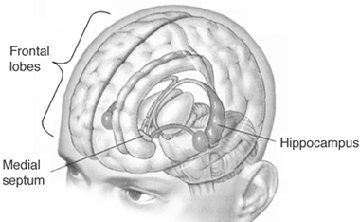
Although the hippocampus is not associated with emotion per se, stimulation of this structure and/or the amgydala, can trigger recent and even long forgotten memories, especially those that the patient feels to be exceedingly emotionally meaningful or personally profound, such as traumas or the recollection of the first time they had sexual intercourse (Gloor, 2012; Halgren, 1992; Penfield & Perot, 1963). The hippocampus, in conjunction with the amygdala (e.g. Chapman et al., 2010; Gloor, 2012; Mishkin, 2010) has been implicated in the regulation of neocortical arousal, and long term memory storage and recall including the ability to remember words, conversations, and visualize one's self and the surrounding environment (Eichenbaum, et al., 1994; Nishitani, et al.,, 1999; Nunn et al., 1999; Penfield & Milner, 1958; Scoville & Milner, 1957; Squire, 1992; Xu et al., 1998).
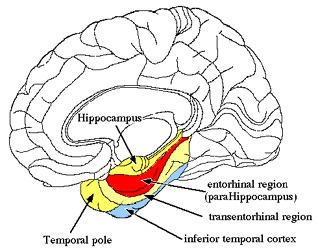

The hippocampus and amygdala displays synaptic plasticity and dendritic proliferation, and will grow additional dendritic spines in response to new learning (Engbert & Bonhoeffer, 1999), and interacts with the amygdala in the storage of the cognitive and emotional attributes of memory (Gloor, 2012; Halgren, 1992), including dreaming and what has been referred to as the primary process (Joseph, 1992a). As noted above, dendritic spines can grow and change position in response to new experiences or lack therefore, thus forming innumerable new synapses and creating vast neural networks supporting complex memories.
THE CINGULATE & ENTORHINAL CORTEX
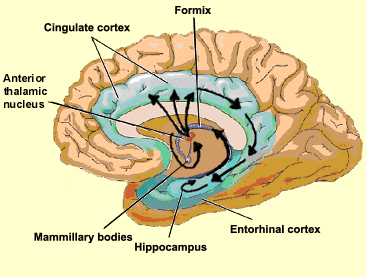
The different structures of the limbic system subserve different as well as overlapping functions ranging from primitive emotions (e.g., hypothalamus) to the spiritually profound (e.g., the amygdala). From an evolutionary perspective, some structures are exceedingly ancient and have a pedigree extending almost a half billion years backwards in time, e.g. the amygdala/striatal/hippocampus, hypothalamus, brainstem.
Others of more recent vintage, such as the cingulate gyrus, may have first begun to evolve around 200 million years ago (MacLean, 2010). This more recent evolutionary origin is also reflected by those functional capacities it mediates, including complex cognitive-affective activities such as maternal-infant behavior and emotional speech (Devinsky et al., 1995; Joseph, 1999b; MacLean, 2010; Slotnick, 1967; Smith, 1945, Stamm, 1955; Ward, 1948).
That the cingulate has evolved more recently is also evident structurally. For example, with the exception of the cingulate, the structures of the limbic system are comprised of allocortex. Allocortex has three layers with pyramidal cells sandwiched between layers I and III. The cingulate consists of mesocortex (also referred to as "paleocortex" and "transitional cortex"), which consists of five layers.
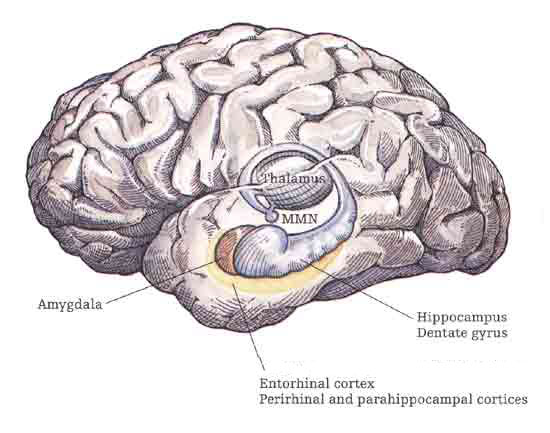
Although the entorhinal cortex --the "gateway to the hippocampus-- may have begun to evolve at the same time as the cingulate, unlike this latter structure, the entorhinal cortex appears to have continued to "evolve" and add new layers. The entorhinal cortex, which receives and relays information to and from the neocortex and hippocampus, consists of between 7 and 8 layers (Braak & Braak, 1992; Ramon y Cajal, 1902/1955; Rose, 1926). This seven to eight layer organization may well partly explain the unique importance of the entorhinal cortex in complex cognitive processing, for it receives input from the hippocampus and all neocortical association areas information which it apparently integrates, and in conjunction with the amygdala and in particular the hippocampus (which it partly coats), stores in memory (Gloor, 2012; Squire, 1992). The entrohinal cortex, which partly surrounds the hippocampus and is interconnected with the amygdala, appears to be a supra-modal memory center.
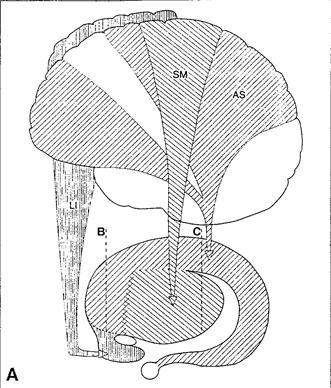
THE LIMBIC AND CORPUS STRIATUM
Within 50 million years of the close of the Cambrian "Explosion" (500 million years ago), cartilaginous sharks began to swim and patrol the primeval seas. Sharks are considered a "living fossil" and dissection of the shark brain reveals a brainstem, diencephalon, and a forebrain consisting of a dorsal/ventral amygdala-striatal mass with a primordial hippocampal-striatum centered at its dorsal core. Together the amygdala, striatum, and hippocampus, formed the forebrain, the dorsal pallium (Gloor, 2012; Haberly 2010; Herrick, 1925; Stephan & Andy, 1977; Ulinksi, 2010).
Although the recipient of visual input, transferred from the brainstem, the primordial amydala-striatal-hippocampus, was dominated by the olfactory lobe--a dominance which was exaggerated further when animals left the sea and began to wonder upon dry shores. With the evolution of amphibians and reptiles, the forebrain expanded and the amygdala, striatum, and hippocampus began to differentiate, and were pushed apart (Gloor, 2012; Haberly 2010; Herrick, 1925; Nieuwenhuys & Meek, 2010ab; Smeet, 2010; Stephan & Andy, 1977; Ulinksi, 2010). Nevertheless, the amygdala remains intimately associated with the striatum, which in turn responds to amygdala concerns.
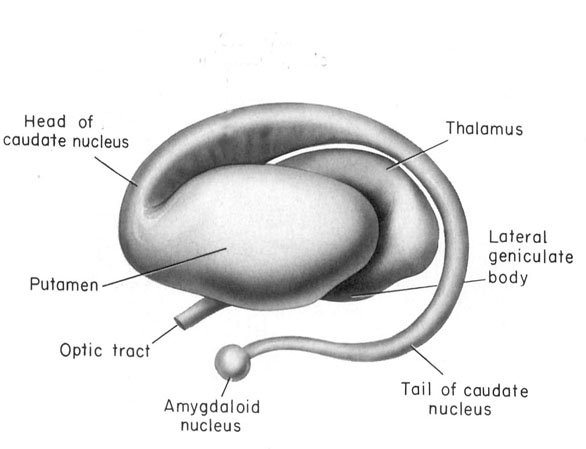
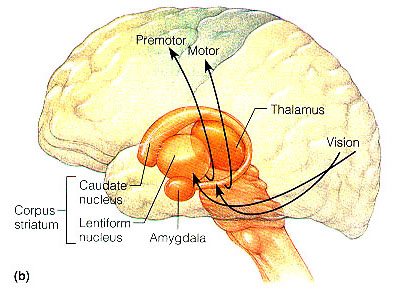
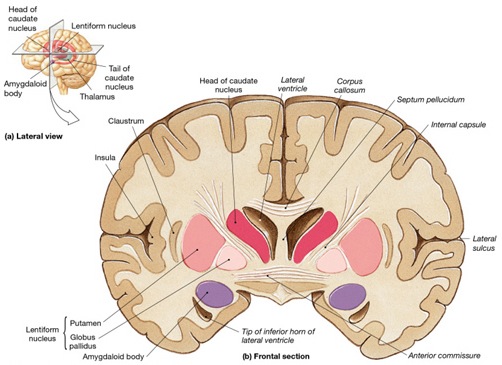
In the human brain, the striatum is located anterior to the thalamus and is a major component of the basal ganglia. Considered broadly, the basal ganglia consists of the subthalamic nucleus, portions of the midbrain, the limbic striatum, the amygdala, and the globus pallidus and putamen (the lenticular nucleus) and the caudate nucleus (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Mink & Thach, 2001; Parent & Hazrati 1995). The putamen and the caudate are referred to as the corpus striatum, with the head of the caudate extending deep into the frontal lobe and its tail merging with the amygdala. Beneath the corpus striatum is the limbic striatum (or extended amygdala) which is comprised of the substantia innominata, the nucleus accumbens, and the olfactory tubercle.
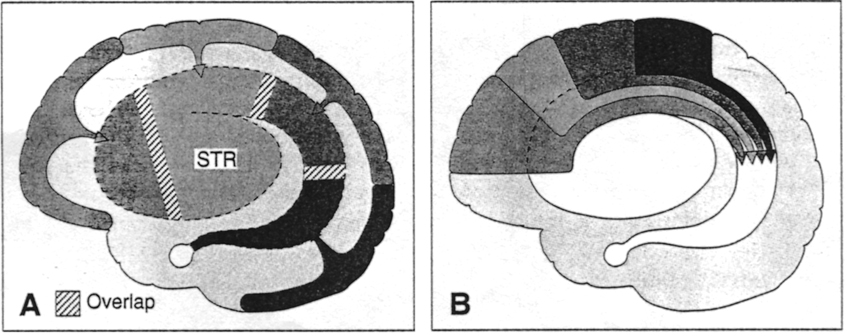
The limbic and corpus striatum serve, in part, as the motor-component of the limbic system, and receive input directly from the amygdala. Hence, the striatum reacts to certain types of visual and olfactory stimuli which are deemed by the amygdala to be emotionally significant, with gross motor movements, e.g., kicking, hitting, running, flailing.
Because of its importance in movement, if the corpus striatum were injured, the patient may become stiff and rigid, such as occurs with Parkinson's disease, or they may involuntarily twitch, jerk, kick, and flail about with their limbs--depending on which aspects of the basal ganglia are injured (see chapter 16). Moreover, patients with abnormalities and reduced functional activity of the striatum may develop obsessive compulsive motor movements, often involving the hands, or increasingly lose inhibitory control over motor functioning such that may display signs of mania. In fact, children with hyperactivity and impulse control disorder, have been shown to display reduced striatal activity, which is increased when treated with Ritalin. However, in "normal" children, Ritalin decreases striatal activity.
Although the dorsal and ventral aspects of the striatum play roles in motor functioning, the limbic striatum is more concerned with emotion and memory. Hence, damage to this structure is associated with loss of memory including Alzheimer's disease.
The Limbic System Vs Neocortex: Consciousness
The contents of consciousness, that is, the contents of the neocortex, are initially derived from the limbic system and thalamus, which provide sensory, perceptual, and emotional input. Structures such as the amygdala and thalamus project to almost every region of the neocortex, and if denied limbic and thalamic input, consciousness would be extinguished and the ability to become conscious of the external or internal world would be denied.
However, although "conscious" the limbic system does not appear capable of self-consciousness or self-reflection. The brain of the shark, amphibian, and reptile, consist of limbic and brainstem tissue, and there is no evidence of self-consciousness, or thinking or thought among these creatures. Rather, they reflexively react. Of course, the limbic system has also continued to evolve with the evolution of mammals and then humans--and in part, the neocortex is the consequence of the evolutionary expansion.
Hence, whereas the neocortical aspects of the conscious human mind are concerned with the more rational and linguistic aspects of experience, the limbic aspects of the human mind are associated with the emotional and even the hallucinatory aspects of experience, including those features associated with what has been described as the primary process. By contrast, the brainstem, controlling waking and sleeping, as well as the rhythmic aspects of vegetative functioning, could be identified with the most primitive regions of the psyche as its functional activity is for the most part, completely beyond conscious scrutiny or control.
THE NEOCORTEX
Perception, cognition, fine-motor expression, and computational processing, is made possible by neurons, the majority of which are pyramidal neurons. Most pyramidal neurons are located in the neocortical mantle of the lobes of the brain, which gives this outercoating its grayish appearance. Over 90% of the gray matter is located in the neocortex. The neocortex ("new cortex") likely first began to evolve between 100 million to 150 million years ago (MacLean, 2010).
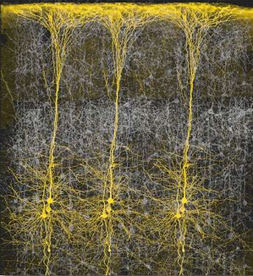
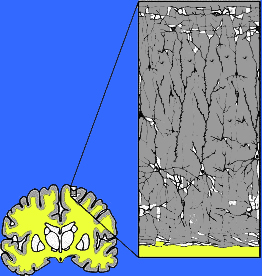
Neocortex has also been referred to as isocortex and is organized into vertical columns and horizontal layers (Mountcastle, 2012). Each layer and each column contains cells which perform specific functions (Ferster et al., 1996; Hubel & Wiesel, 1968, 1974; Mountcastle, 2012; Peters & Jones, 1984) and which receive or transmit information to or from adjacent cells or distant regions of the brain (Kaas & Krubitzer 2001; Peters & Jones, 1984; Sereno et al. 1995). For example, each column contains neurons that may respond to the same frequency of sound, or to tactile input to the thumb, or to a visual input on the same region of the retina (Hubel & Wiesel, 1968, 1974; Mountcastle, 2012), depending on if the column is located in the temporal, parietal, or occipital lobe.
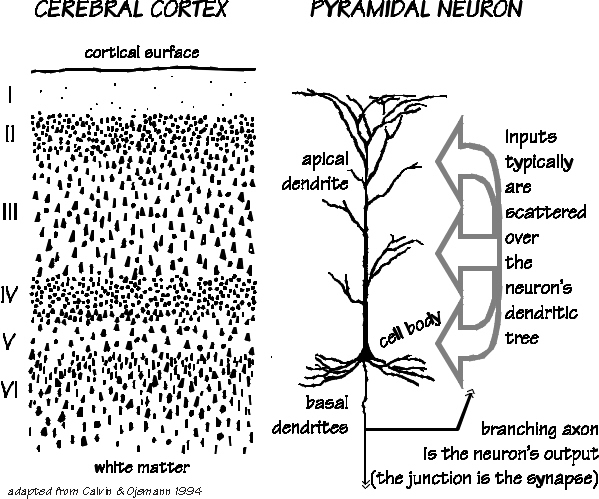
Those neurons which project to neurons in the next column or to those neurons in an upper or lower layer, are referred to as local circuit neurons. Those which project from the neocortex to the brainstem, or from one half of the brain to the other, are referred to as long distance neurons (Peters & Jones, 1984).
Neocortical Layers
Classically, the neocortex is said to consist of six to seven layers when in fact it consists of numerous layers which vary depending on brain area (Braak & Braak, 1992; Peters & Jones, 1984; Ramon y Cajal, 1902/1955; Rose, 1926). For example, the deepest layer, neocortical layer VI, consists of two distinct layers (VIa and VIb). In the occipital lobe, three additional layers (i.e. sublayers) can be distinguished within layer IV (which also receives considerable thalamic input and is very thick). By contrast, within the motor areas of the frontal lobe, layer IV is exceedingly thin (as there is comparatively minimal thalamic input), whereas layer V is exceedingly thick, It is layer V of the frontal motor areas which contribute the bulk of axons that form the descending corticbulbar, corticopontine, and corticorubral brainstem pathways which establish contact with cranial nerve and sensory and spinal motor neurons (Brodal, 1981; Kuypers & Catsman-Berrevoets, 1984). Likewise in the temporal neocortex layer V is relatively thick as are layers I and VI (since much of the temporal lobe is association and assimilation cortex). As noted, the entorhinal cortex, the "gate way to the hippocampus" and which is located along the medial surface of the temporal lobe, consists of between 7 and 8 layers (Braak & Braak, 1992; Ramon y Cajal, 1902/1955; Rose, 1926).
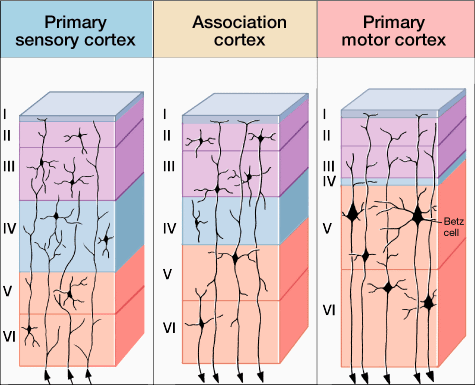
Hence, the thickness, layering, and composition of the human neocortex varies from lobe to lobe and actually consists of from 7 to 9 (or more) layers rather than 6. For our purposes (and throughout this book) we will described the neocortex as having 7 layers.
Specifically, layer I is referred to as the Molecular Layer and consists of Golgi II cells and horizonal cells. Layer I receives innumerable dendrites from local circuit neurons located in the lower layers. Layer I, however, actually contains few neurons and is mostly made up of tangentially running axons and horizontally running birfucating apical dendrites received from the pyramidal cells of the lower layers (Peters & Jones, 1984). Layer II is referred to as the External Granular Layer, and consists of densely packed small pyramidal, stellate, and granule cells. Most of the neurons in layer II are local circuit neurons which project to adjacent columns and adjacent layers.
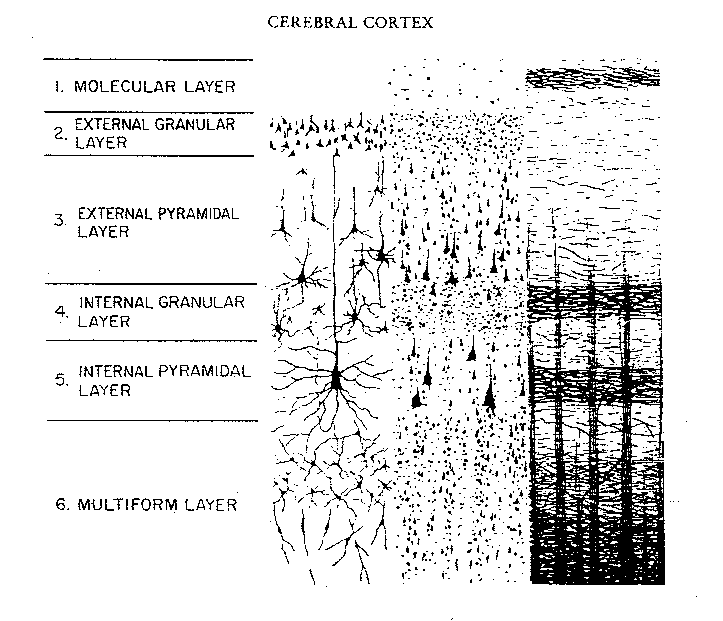
Layer III is the Pyramidal Layer and consists of medium pyramidal cells which project axons to distant areas of the neocortex. Hence, the neurons of layer III can be considered long distance neurons.
Layer IV, the Internal Granular Layer, has a granular appearance and consists of small pyramidal, granule, and stellate (starshaped) cells and receives massive axonal projections from the thalamus. These neurons are predominantly local circuit, and project to adjacent columns and layers. That is, upon receiving and analyzing thalamic input, the neurons of layer IV transfer this data to adjacent neurons for additional analysis. Because the primary, secondary and association sensory areas receive considerable thalamic input, layer IV is relatively thick--except in the motor cortex.
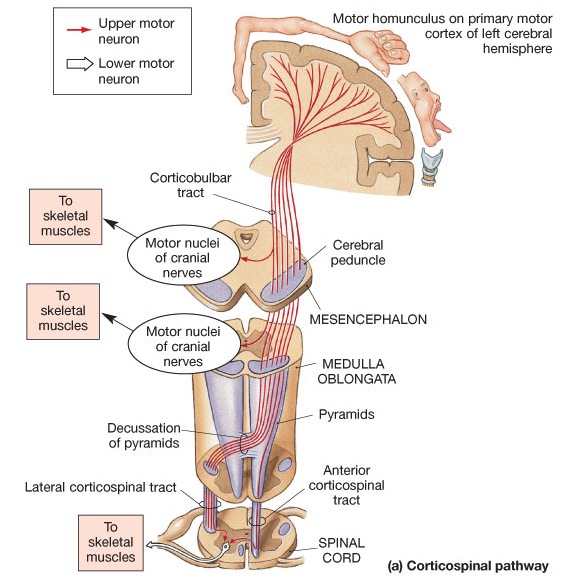
Layer V is the Ganglionic Layer and consists of large and medium size pyramidal cells, including, in primary motor cortex (Brodmann's area 4) the giant cells of Betz. The pyramidal neurons of layer V are long distance neurons, and give rise to descending axons which form the corticospinal, pyramidal, corticobulbar, corticopontine, and corticorubral brainstem pathways which establish contact with cranial nerve and sensory and spinal motor neurons (Brodal, 1981; Kuypers & Catsman-Berrevoets, 1984). It is these "pyramidal" and cortico-spinal neurons which make purposeful, fine motor movement possible. Approximately 31% of the corticospinal tract arises from the pyramidal cells located in the primary motor areas 4, with the remainder arising from the frontal motor associations areas 6, 8, and the primary somesthetic areas 3,1,2, with a scattering of fibers being contributed by the occipital and temporal lobe, as well as limbic system structures.
Layer VIa is the Multiform Layer and contains pyramidal, fusiform, and spindle shaped cells, whereas Layer VIb consists of predominantly of spindle shaped cells. These are predominantly local circuit neurons, and receive considerable input from the brainstem.
The Cytoarchitextural Neuronal, & Chemical Organization of the Neocortex
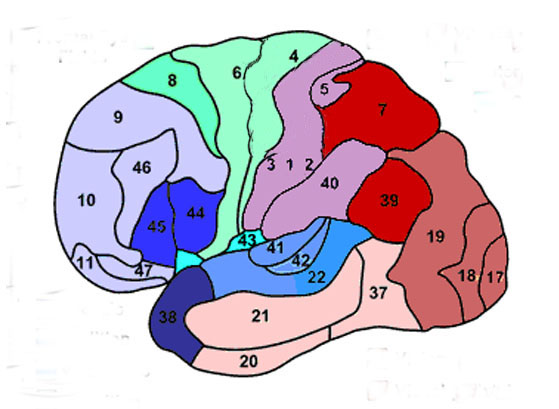
Korbinian Brodmann detailed the regional variation in the cytoarchitectural organization of the cortex, and conducted detailed comparative studies of numerous species, each of which displays common as well as varying patterns of cytoarchitexture and gyral folding. Based on these cytoarchitextural differences and commonalities, Brodmann divided the cortex into distinct regions and created cytoarchitextural maps of the brains of a variety of species, including humans. For examples, Brodmann's area 17 is synonymous with the primary visual cortex, whereas Brodmann's areas 3,1,2, denote and identify the primary somesthetic receiving areas.

However, although these area differ in regard to organization, what they share in common is a preponderance of pyramidal cells. As noted, pyramidal cells are also the largest and are more numerous than any other neocortical neuron (Peters & Jones, 1984). Pyramidal neurons account for up to 3/4 of all neocortical cells. Pyramidal neurons also serve as both local-circuit and long-distance neurons and generally receive two types of synaptic contacts referred to as Gray types I and II which differ in synaptic morphology and (respectively) excitatory vs inhibitory influences (Peters & Jones, 1984). However, almost all pyramidal cell are excitatory and use glutamate and aspartic acid as transmitters (Tsmoto, 2010).
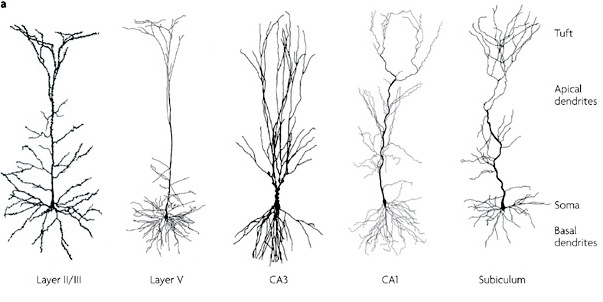
Pyramidal cells can also be classified as Golgi I and II cells. However, of all neocortical neurons, only 10% are Golgi type I neurons, the main source of long-distance (excitatory) axons, the majority are interneurons, i.e. local circuit neurons which in turn provide almost 90% of cortical axons and dendrites. Approximately 95% of Golgi type I long distance axons interconnect distant regions within the same hemisphere and only about 5% cross the corpus callosum, the fiber pathways which link the right and left hemishere (Peters & Jones, 1984).
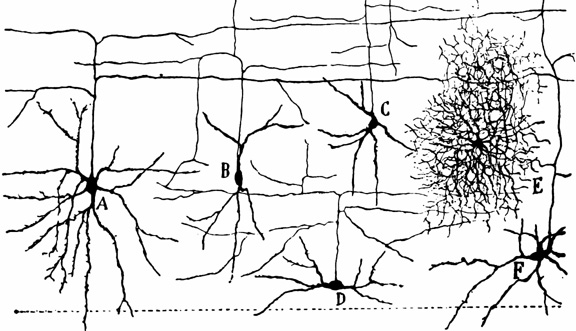
Non-pyramidal cells also function as local-circuit (inter-) neurons, and connect adjacent cells, layers, and cell columns, and display the greatest degree of morphological diversity. These include stellate cells, bipolar cells, chandelier cells, and basket cells. Many of these cells are inhibitory and may use GABA as a neurotransmitter, though the majority in fact appear to be excitory and use glutamate (Peters & Jones, 1984). Presumably, non-pyramidal, local circuit (interneurons) act to fine tune information processing via inhibitory filtering and selective excitatory transmission. They also serve to integrate and assimilate information received in adjacent regions of the neocortex.
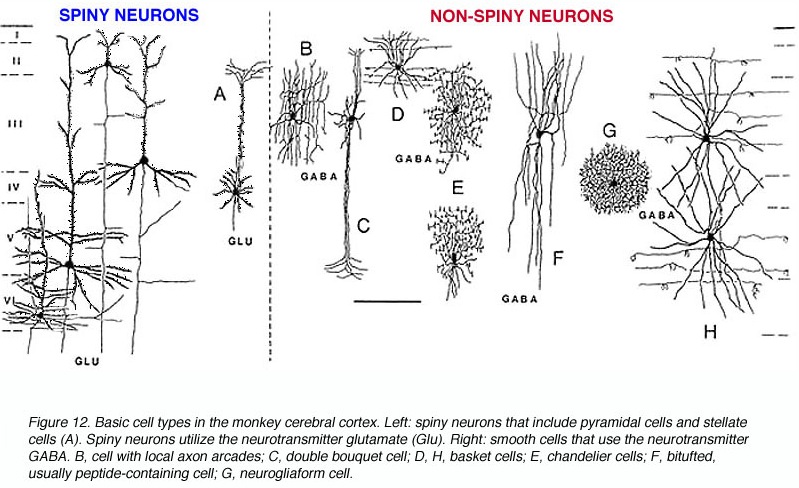
In addition to glutmate and GABA, neocortical neurons contain and respond to peptides, including substance P, corticotropin releasing factor, and opiates. The peptide containing neurons tend to congregate in layers II, III, and IV (Jones & Hendry, 2006).
Information processing throughout the brain is also dependent on glia and non-neuroglia elements. Glia serve a supportive and nurturing role, and may also act to store information. During embryonic brain development, radial glia fibers act to guide migrating neurons to the neocortex, and some glia also form myeline sheaths which surround axons, thus serving as a form of insulation which promotes information transmission.
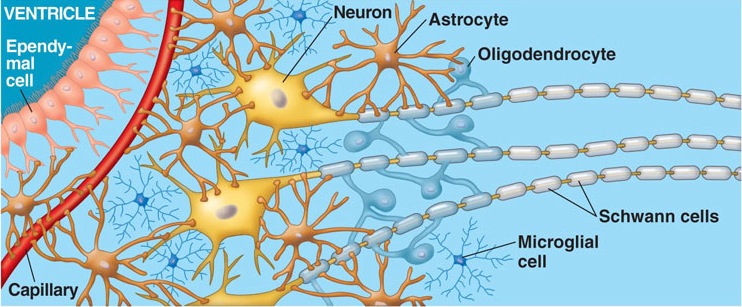
Glia and non-neuroglia elements make up almost 70% of the volume of the neocortex. Of the remainder, 22% consists of axons and dendrites, with the body (soma) of the neuron comprising only 8% (Peters & Jones, 1984).
THE CONSCIOUS AND UNCONSCIOUS (EMOTIONAL) MIND
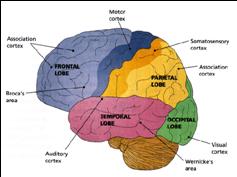
The human cerebrum can be subdivided into frontal, temporal, parietal, and occipital lobes, as well as the limbic system, striatum and diencephalon. The brainstem and cerebellum are not considered part of the cerebrum, but instead comprise the "hind brain."
Neocortex & Consciousness
The cerebrum constitutes nearly 90% of the volume of the brain, and is 50 times larger than the brainstem and 8 times larger than the cerebellum (Filipek, et al., 1994). More than 60% of the cerebrum consists of gray matter, and less than 40% consists of white matter. The amygdala, basal ganglia, diencephalon and hippocampus, make up less than 3% of the central gray, whereas more than 90% of the gray matter is found within the neocortex, "a proportion that highlights the central and dominant role that the neocortex plays in virtually all fully-integrated functions of the central nervous system (Caviness et al., 2012, p. 5).
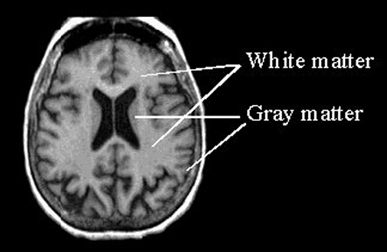
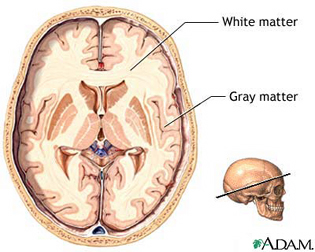
The neocortex is clearly associated with what is classically considered the conscious mind--a consciousness that knows it is conscious, and which includes the capacity to think and reason, to abstract and consciously reflect upon the self and the world, as well as the ability speak grammatically and read, write, compose, and recite the music of poetry. Consciousness, however, is modular, with different regions of the neocortex engaging in both localized as well as parallel processing.
For example, as based on functional imaging, it has been demonstrated that language processing, and silent mental activities, such as thinking, or generating inner speech, activates the neocortex of the frontal lobes (Paulesu, et al., 2013; Peterson et al., 2008; Demonet, et al., 1994). Reading and language processing also activates the neocortex of the temporal lobes (Bookheimer, et al., 1995; Bottini, et al., 1994; Fletcher et al., 1995; Howard et al., 199; Shaywitz, et al., 1995; Warburton, et al., 1996) and the left inferior parietal lobule (Bookheimer, et al., 1995; Price, 2012). Moreover, during language processing there is activity in the brainstem, the cerebellum, and various limbic structures, especially the mesocortical anterior cingulate gyrus.

When considered very broadly, it could be argued that the neocortex is associated with the conscious, rational mind, and conscious-awareness that we identify as distinctly human--a consciousness that knows it is conscious (Joseph, 1982, 2008a). By contrast, it has been argued (Joseph, 1992ab) that the limbic system represents that aspect of the mind classically referred to as the "unconscious" (Freud, 1900) and the "collective unconscious" (Jung, 1945). And just as it has been theorized that the unconscious continually supplies the conscious mind with all manner of impulses, imagery, and ideas (e.g. Freud, 1900), it is now apparent that the limbic system does likewise, and that those aspects of consciousness associated with the neocortex are often driven by these unconscious limbic mental realms and associated impulses.
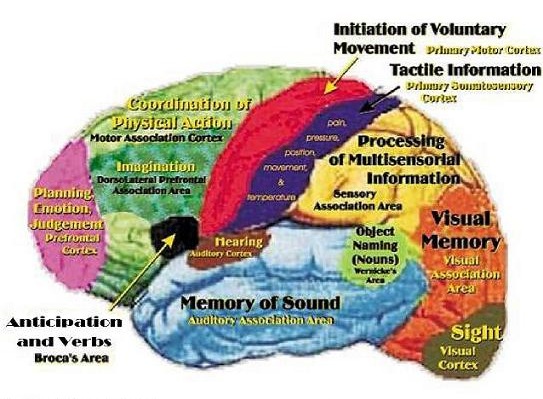
THE FRONTAL, PARIETAL, TEMPORAL AND OCCIPITAL
LOBES The rational, logical, linguistic, and self-reflective aspects of consciousness, therefore, are associated with the neocortical shroud which envelops the brain. Neocortical consciousness, however, also appears to be somewhat modular with the different lobes of the brain (and the right vs left hemisphere) subserving different aspects of consciousness and perceptual and personality functioning.
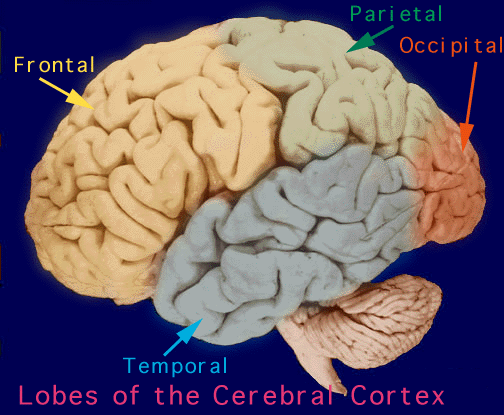
As is common knowledge, the six to seven layered neocortical shroud which encompasses and envelopes the old brain, can be divided into the frontal lobes which comprise the anterior half of the human telencephalon, and the parietal, occipital and temporal lobes which are located in the posterior half of the cerebrum, each of which contributes differently to the mosaic of mind including personality.
The frontal lobes has been referred to as the "senior executive" of the brain and personality (Fuster 2012; Joseph 2006a, 1999a; Koechlin et al., 1999; Milner & Petrides 1984; Passingham 2013; Selemon et al. 1995; Shallice & Burgess 2001; Stuss 1992) and is associated with goal formation, long term planning skills, the ability to consider multiple alternatives and consequences simultaneously, as well as memory search and retrieval. Because it is interlocked with the thalamus, limbic system, brainstem, and the parietal, occipital, and temporal lobes, the frontal lobes are provided multiple streams of input and are constantly informed as to the processing which takes place in other regions of the brain (Cavada 1984; Fuster 2012; Jones and Powell 1970; Goldman-Rakic 1995, 1996; Passingham, 2013, 2012; Pandya & Yeterian 2010; Petrides & Pandya 2008). Moreover, it can act to inhibit, suppress, or enhance perceptual and information processing, including learning and memory through inhibitory and excitatory influences directed to the thalamus, brainstem, or the different lobes of the brain (Joseph, 1999a).
For example, the frontal lobes interact directly with the temporal lobe (which contains the hippocampus and amygdala) in order to ensure that certain percepts are attended to, committed to memory, and later remembered (Dolan et al., 2012; Kapur et al., 1995; Passingham, 2012; Posner & Raichle, 1994; Tulving et al., 1994). As demonstrated through functional imaging, it has been determined that the ability to remember a visual or a verbal experience, is directly correlated with activation of the right or left frontal lobe and the temporal lobes during that experience (Brewer et al., 1998; Wagner et al., 1998). According to Wagner et al., (1998, p. 1190), "what makes a verbal experience memorable partially depends on the extent to which left prefrontal and medial temporal regions are engaged during the experience." The degree of this activity, therefore, can be used to predict which experiences will be "later remembered well, remembered less well, or forgotten" (Brewer et al., 1998, p. 1185). Thus the frontal lobes appear to be acting directly on and in concert with the amygdala and hippocampus in regarding to memory functioning.
In addition to influencing arousal, memory, and perceptual actiivty, the right and left frontal motor areas control fine motor activity, including the emotional and non-emotional aspects of expressive speech (Joseph, 1982, 2008a, 1999a,e; Ross, 2013). Both the right and left frontal lobes in fact become functionally active during language tasks (Peterson et al., 2008), and receive auditory-linguistic input that has been organized and transmitted from the posterior speech zones, the inferior parietal lobule, and in the left hemisphere, Wernicke's area in the superior temporal lobe. Damage to the left frontal lobe can thus result in expressive aphasia, whereas right frontal injury may disturb the melody and prosodic aspects of speech.
Because the frontal lobes serve the "senior executive" of the brain and personality, if the frontal lobes are injured, all aspects of personality functioning may become severely abnormal (Fuster, 2012; Joseph, 2006a, 2008a, 1999a; Stuss & Benson, 1984). Patients may become lethargic, apathetic, or conversely, disinhibited, impulsive, aggressively sexual, and display what has been classically referred to as the frontal lobe personality.
One individual who was described as quite gentle and sensitive prior to his injury, subsequently raped and brutalized several women. Similar behavior has been described following frontal lobotomy. As stated by Freeman and Watts (1943, p. 805): "Sometimes the wife has to put up with some exaggerated attention on the part of her husband, even at inconvenient times and under circumstances which she may find embarrassing. Refusal, however, has led to one savage beating that we know of, and to an additional separation or two" (p. 805). Curiously, in these situations Freeman and Watts (1943, p. 805) have suggested that "spirited physical self-defense is probably the best strategy of the woman. Her husband may have regressed to the cave-man level, and she owes it to him to be responsive at the cave-women level. It may not be agreeable at first, but she will soon find it exhilarating if unconventional."
However, since the frontal lobes serve so many functions, patients may display different symptoms depending on if the lesion impacts the right, left, orbital, or medial aspects of the frontal lobes (Joseph, 1999a). Oatients may display severe apathy, depression, schizophrenia or aphasic speech (left frontal), mania, disinhibition, and confabulation (right frontal), obsessive compulsions (orbital-striatal) or catatonia (medial frontal).
For example, a soldier who received a gunshot wound that passed completely through the frontal lobes, "layed in a catatonic-like stupor for two months, always upon one side with slightly flexed arms and legs, never changing his uncomfortable position; if he were rolled into some other position, he would quickly get back into his former one. He did not obey commands, but if food and drink were given to him, he swallowed them naturally. He was incontinent, made no complaints, gazed steadily forward and showed no interest in anything. He could not be persuaded to talk, and then suddenly he would answer quite correctly about his personal affairs and go back to mutism. From time to time he showed a peculiar explosive laugh, especially when his untidiness was mentioned". Incredibly, the patient "was eventually returned to active duty" (Freeman & Watts 1942, pp 46-47).
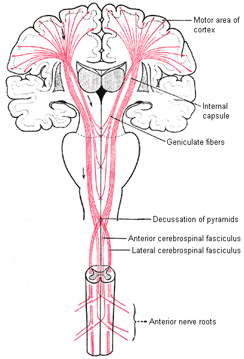
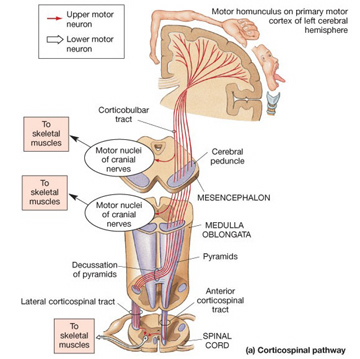
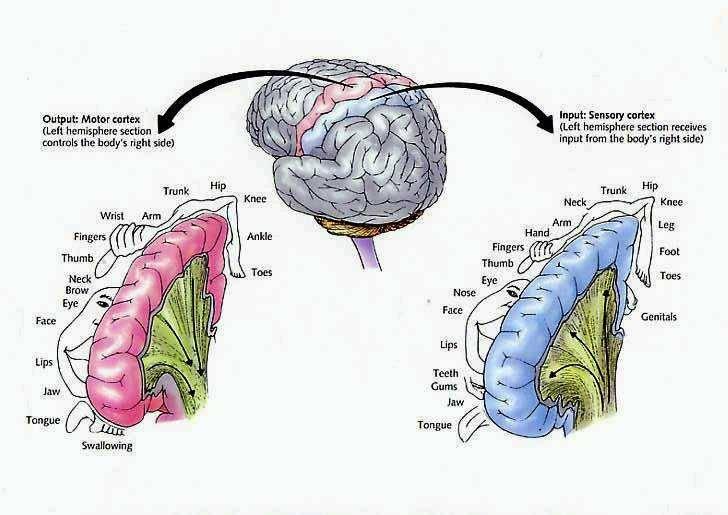
The motor areas of frontal lobes controls body movement and fine motor activity. A motor map of the body is maintained within the neocortex of the frontal lobes, and if damaged, the individual will become paralyzed on one half of the body; the extent of the paralysis depending on the extent and depth of the damage.
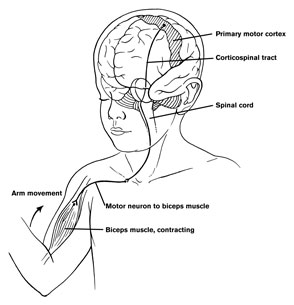
The frontal lobes control muscular activity and movement through the pyramidal tract (also known as the cortico-spinal tract). Pyramidal neurons project to the motor centers in the striatum, thalamus, brainstem, and the spinal cord. In this way, gross and fine motor movement can be controlled by the senior executive of the brain and personality.
THE TEMPORAL LOBES
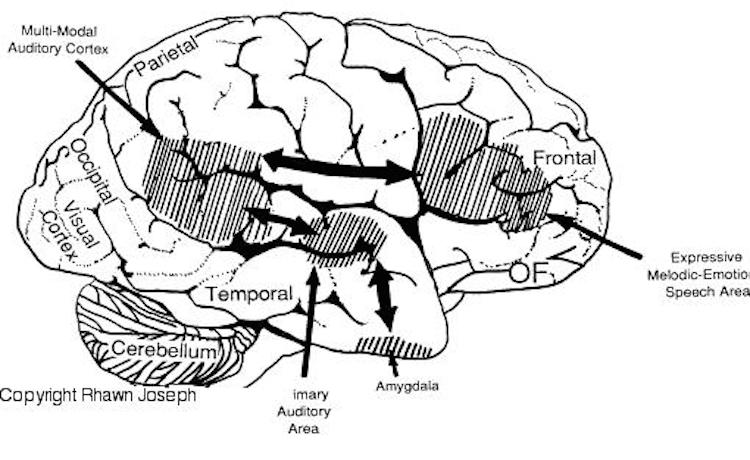
Whereas the frontal lobe act to regulate personality and emotion, such as through inhibitory projections it maintains with the limbic system, the temporal lobes are the source of one's personal subjective and emotional identity (Gloor, 2012) and appears to be the main storage site for spatial, verbal (Brewer et al., 1998; Squire, 1992; Wagner et al., 1998), personal and even sexual memories (e.g., Gloor, 2012; Halgren, 1992). The temporal lobes, in fact, contain the core structures of the limbic system, the amygdala and hippocampus, and of all brain regions, only stimulation or activation of the temporal lobe or the underlying limbic structures, gives rise to personalized, subjective, emotional, and sexual experiences (Gloor, 2012; Halgren, 1992; Penfield & Perot, 1963). Stimulation of the temporal lobe can give rise to profound auditory or visual hallucinations, including the sensation or having left the body, as well as spiritual and religious feelings such as having the "truth" revealed and of receiving knowledge regarding the meaning of life and death.
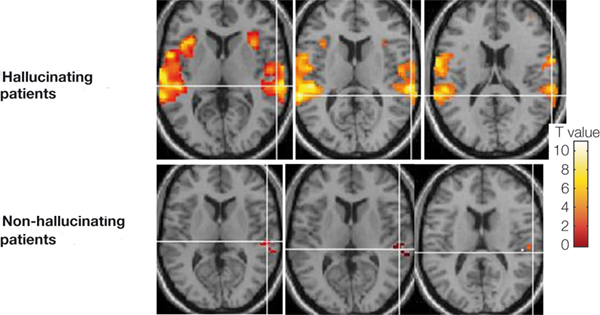
In addition to memory and personal and spiritual identity, the temporal lobe are responsive to complex auditory and visual stimuli (Binder et al., 1994; Gross & Graziano 1995; Nakamura et al. 1994; Nelken et al., 1999; Nishimura et al., 1999; Price, 2012; Rolls 1992; Tovee et al. 1994), and subserve the ability to comprehend complex and emotional speech. Functional imaging studies have repeatedly demonstrated activity in the superior and middle temporal lobe when engaged in language tasks (Bookheimer, et al., 1995; Bottini, et al., 1994; Fletcher et al., 1995; Howard et al., 1996; Shaywitz, et al., 1995; Warburton, et al., 1996). The temporal lobes, however, are functionally lateralized. Specifically, whereas the right temporal lobe subserves the ability to perceive and comprehend emotional, animal, environmental, and musical sounds (Joseph, 2008a; Parsons & Fox, 2012; Ross, 2013) the left temporal lobe, including Wernicke's area is directly responsible for the capacity to comprehend complex human speech. The left superior temporal lobe, i.e. the planum temporal, which contains the auditory area, is in fact significantly larger than its counterpart on the right.
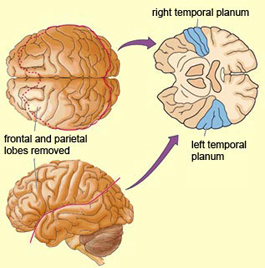
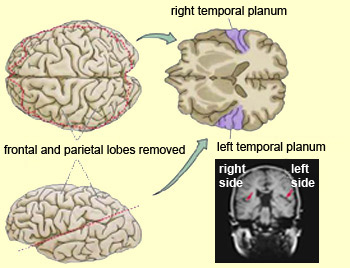
It has been determined, as demonstrated through lesion and physiological studies, that Wernicke's area (in conjunction with the inferior parietal lobule) acts to organize and separate incoming sounds into a temporal and interrelated series so as to extract linguistic meaning via the perception of the resulting sequences. When damaged a spoken sentence such as the "big black dog" might be perceived as "the klabgigdod." Patients develop what has been referred to as Wernicke's receptive aphasia. Presumably this disorder is due in part to an impaired capacity to discern the individual units of speech and their temporal order. That is, sounds must be separated into discrete interrelated linear units or they will be perceived as a meaningless blur.
Patients with damage to Wernicke's area are nevertheless, still capable of talking due to the preservation of Broca's area which continues to receive lingusitic-related ideational material via the arcuate fasciculus pathway which originate in the temporal lobe and passes through the inferior parietal lobule--a structure which acts as a phonological storehouse that becomes activated during short-term verbal memory and word retrieval (Demonet, et al., 1994; Paulesu, et al., 2013; Price, 2012). However, because Wernicke's area is injured, speech output also becomes abnormal, a condition referred to as fluent aphasia. Broca's area keeps talking, but what is says is nonsense.

The speech output of a patient with "fluent aphasia" in many respects resembles the acutely psychotic speech of patients suffering from certain subtypes of schizophrenia. In fact, certain subtypes of schizophrenia have been repeatedly associated with abnormalities and irritative lesion involving the left temporal lobe (DeLisi et al. 2001; Dauphinais et al. 2010; Flor-Henry 1983; Perez et al. 1985; Rossi et al. 2010, 2001; Sherwin 1981). Conversely, given the role of the left temporal lobe in language, patients diagnosed with schizophrenia and who show signs of left temporal lobe dysfunction, also tend to demonstrate aphasic abnormalities in their thought and speech (Chaika, 1982; Flor-Henry, 1983; Hoffman, 2006; Hoffman, Stopek & Andreasen, 2006; Rutter, 1979).
The right temporal lobe also participates in language--as demonstrated through functional imaging (Bottini et al., 1994; Price et al., 1996; Shaywitz, et al., 1995)--and is especially responsive to sounds conveying emotion, melody, prosody, including those made by animals and those arising from the natural environment such as wind and rain (e.g. Fujii et al., 2010; Joseph, 1982, 2008a; Ross, 2013; Schnider et al. 1994; Zatorre & Halpen, 2013). It is the right temporal lobe which enables an individual to determine if someone is speaking sincerely, or with anger, happiness, and so on, whereas the left temporal lobe listens to the words being said.
Hence, if the right temporal lobe is severely injured, patients may suffer from a non-verbal social-emotional auditory agnosia (Joseph, 1982, 2008a, Ross, 2013; Schnider et al. 1994), also referred to as phonagnosia (van Lancker, et al., 2008), in which case they can no longer perceive social-emotional vocal nuances and may misperceive what others are saying or implying, and may be no longer capable of hearing "sincerity" or mirth or even "love" in which case they may become paranoid. Hence, although the ability to comprehend the non-emotional, denotative aspects of language is preserved (Fujii et al., 2010), patients with right superior temporal injuries may lose the capability to correctly discern environmental sounds (e.g. birds singing, doors closing, keys jangling), emotional-prosodic speech, as well as music (Nielsen, 1946; Ross, 2013; Samson & Zattore, 2008; Schnider et al. 1994; Spreen et al. 1965; Zatorre & Hapern, 2013).
The loss of the ability to appreciate music is due to the right temporal lobe being dominant for the perception of melodic and musical stimuli. For example, while listening to or performing scales, activity increases in the left temporal lobe, whereas when listening to Bach (the third movement of Bach's Italian concerto), the right temporal lobe becomes highly active (Parsons & Fox, 2012). Likewise, Evers and colleagues (1999) in evaluating cerebral blood velocity, found a right hemisphere increase in blood flow when listening to harmony (but not rhythm), among non-musicians in general, and especially among females. In fact, right temporal lobe activity increased when pianists were playing from memory Parsons & Fox, 2012). Conversely, right temporal injuries can disrupt the ability to remember musical tunes or to create musical imagery (Zatorre & Halpen, 2013).
Unfortunately, individuals with right temporal injuries not only lose the ability to appreciate music, but may misperceive and fail to comprehend a variety of paralinguistic social-emotional messages. This includes difficulty correctly identifying the voices of loved ones or friends, or discerning what others may be implying, or in appreciating emotional and contextual cues, including variables such as sincerity or mirthful intonation. In consequence, a patient may complain that his wife no longer loves him, and that he knows this from the sound of her voice. Or, because of difficulty discerning nuances such as humor and friendliness the patient may even become paranoid or delusional as they realize that friends and loved ones sound different, and may entertain delusions that they've been replaced by imposters.
The temporal lobes, therefore, are crucially important in memory, language, social emotional relations, maintaining personal identity and the ability to comprehend speech. If injured, patients may suffer memory loss, and/or language-related ideational activity may become abnormal, thus producing a formal thought disorder or delusions or paranoia, and personality functioning may become fractured and thus schizophrenic.
THE PARIETAL LOBES
The parietal lobes maintain the body image and also consists of cells which are responsive to a variety of divergent stimuli, including movement, hand position, objects within grasping distance, audition, eye movement, pain, heat, cold, as well as complex and motivationally significant visual stimuli (Aoki et al., 1999; Cohen et al. 1994; Deibert et al., 1999; Dong et al. 1994; Kaas, 2013; Lam, et al., 1999; Remy et al., 1999). The parietal lobes assimilate this information in order to coordinate reaching and the movements of the body in space, particularly the hand. The parietal lobe is in fact considered a "lobe of the hand." Hence, because of its importance in controlling hand movement, one consequence of parietal lobe injury may be apraxia--an inability to perform coordinated step-wise and sequential movements of the hands (Barrett et al., 1998; Buxbaum, et al., 1998; De Renzi and Lucchetti, 2008; Heilman et al., 1982; Kimura, 2013; Strub and Geschwind, 1983).
Patients with apraxia demonstrate gross inaccuracies as well as clumsiness when making reaching movements or when attempting to pick up small objects in visual space. They may also be impaired in their ability to acquire or perform tasks involving sequential changes in the hand or upper musculature including well learned, skilled, and even stereotyped motor tasks such as lighting a cigarette, using a key, or making a cup of coffee. That is, they may perform the various steps in the wrong order; e.g., pretending to stir the coffee, then pretending to pour it.
In addition, patients may no longer be able to perform constructional tasks, such as drawing or copying or performing puzzles or block designs, a condition referred to as constructional apraxia. They may leave out parts, grossly distort the figure, or even fail to draw half the object--particularly with right parietal injuries.
Apraxic disorders are most common with injuries involving the inferior parietal lobule (IPL), a structure that engages in the motor programming of hand movements, such as those involved in drawing, constructing, building, and which require sequential and orderly movements (De Renzi and Lucchetti, 2008; Heilman et al., 1982; Kimura, 2013; Strub and Geschwind, 1983). As noted above, the IPL (the angular and supramarginal gyrus) is also coextensive with Wernickes area and acts to assimilate auditory, visual, and tactile impressions, and provide names for these associated assimilation (which also makes reading and writing possible). Once this is accomplished, the IPL then injects this material, temporal sequential fashion, into the stream of language and thought all of which is transmitted to Broca's area and which is then expressed as grammatical speech (Joseph, 1982, 2006a, 1999e,f; Joseph et al., 1984).
This is not merely a hypothesis based on lesion studies, for as based on functional imaging, others have come to the same conclusion; i.e. the inferior parietal lobule acts as a phonological storehouse that becomes activated during short-term memory and word retrieval (Demonet, et al., 1994; Paulesu, et al., 2013; Price, 2012). For example, viewing words activates the left supramarginal gyrus (Bookheimer, et al., 1995; Vandenberghe, et al., 1996; , Menard, et al., 1996; Price, 2012) which also becomes active when performing syllable judgements (Price, 2012), and when reading (Bookheimer, et al., 1995; Menard, et al., 1996; Price, et al., 1996). Injuries to the IPL, therefore, can result in word finding difficulty (anomia) as well as a loss of the ability to read or write.
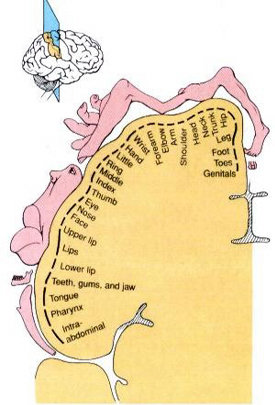
The parietal lobes also subserve and maintain a unique aspect of one's personal identity, the body image (Joseph, 2006a, 2008a). The parietal lobe actually maintains multiple maps of the body (Kaas, 2013; Lin et al., 1994), and in this regard, it is responsible for the ability to remember and recognize the body as an extension of one's personal self--particularly the right parietal lobe which appears to maintain multiple maps of both halves of the body and a bilateral image of body-image visual space (Joseph, 2006a, 2008a). Presumably, it is because the body image is maintained in the parietal lobe, that individuals who have suffered amputations continue to experience a phantom limb. Although the body part has been eliminated, the neural representation for the body may remain intact.
Because the parietal lobes maintain the body image, and as the entire body is multiply represented along the surface of the parietal lobes, massive injuries may result in a destruction of the body image. Memories of the body may be erased.
As noted, the right parietal lobe maintains a bilateral body image, the left parietal lobe appears to maintain a memory of only half the body (Joseph, 2006a, 2008a). Inf the left parietal lobe is injured, the right parietal area may continue to monitor both halves of the body and both halves of visual space and the body image will remain intact. If the right parietal area is severely injured, the left half of the body image, and in fact all memories of the left half of the body and the left half of space, may be abolished; a condition referred to as unilateral neglect. Patients may dress or groom only the right half of their body, eat only off the right half of their plates, and fail to read the left half of sentences and words, and so on. The left parietal lobe, having a memory of only the right half of the body, is unable to recognize the left half of the body, and ignores it and denies its existence.
Following a massive right parietal injury, and when confronted with their unused or immobile limbs patients may (at least initially) deny that it belongs to them and instead claim it must belong to the doctor or a patient in the next bed. For example, Gerstmann (1942) describes a patient with left-sided hemiplegia who "did not realized and on being questioned denied, that she was paralyzed on the left side of the body, did not recognize her left limbs as her own, ignored them as if they had not existed, and entertained confabulatory and delusional ideas in regard to her left extremities. She said another person was in bed with her, a little Negro girl, whose arm had slipped into the patient's sleeve" (p. 894). Another declared, (speaking of her left limbs), "That's an old man. He stays in bed all the time."
With right parietal injuries coupled with unilateral neglect, patients may develop a dislike for their left limbs, try to throw them away, become agitated when they are referred to, entertain persecutory delusions regarding them, and even complain of of strange people sleeping in their beds due to their experience of bumping into their left limbs during the night (Bisiach & Berti, 1987; Critchley, 1953; Gerstmann, 1942). One patient complained that the person tried to push her out of the bed and then insisted that if it happened again she would sue the hospital. A female patient expressed not only anger but concern least her husband should find out; she was convinced it was a man in her bed.
THE OCCIPITAL LOBES AND VISION
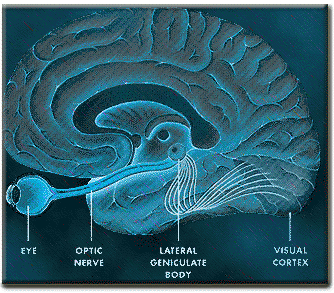
The occipital lobes are the smallest lobes of the brain, but like other tissues of the mind, they process information from a number of modalities and contain neurons which respond to vestibular, acoustic, visual, visceral, and somesthetic input (Beckers & Zeki 1994; Ferster, et al., 1996; Horen et al., 1972; Pigarev 1994; Sereno et al. 1995; Zeki. 2012). Predominantly, however, the neocortex of the occipital lobes are the main receiving stations for visual stimuli transmitted from the retina to the thalamus (Barbur et al., 2013; Ferster et al., 1996). Simple and complex visual and central/foveal analysis is one of the main functions associated with the occipital lobe (Kaas & Krubitzer 2001; Sereno et al. 1995; Zeki, 2012).
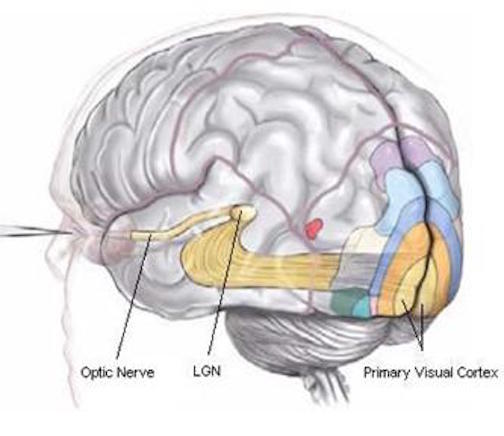
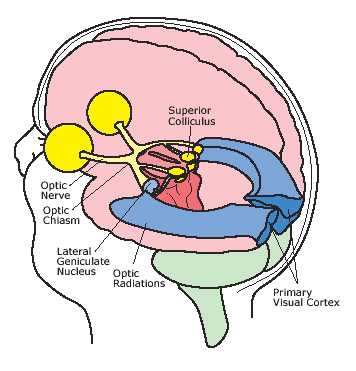
Specifically visual information is shunted from the lateral geniculate nucleus (LGN) of the thalamus to the primary visual receiving areas, striate cortex, area 17. Area 17 is referred to as "striate cortex" due to the striped appearance of layer IV, which is also directly innervated by the LGN. Layer IV is divided into three sublayers, with the middle layer containing a rather thick band of cortex, the band of Baillarger/Gennari, which is visible to the naked eye.
Throughout the striate cortex neurons with similar receptive properties are stacked in columns, with all the neurons within one column responding, for example, to a certain visual orientation, and the cells in the next column to an orientation of a slightly different angle. Columns exist for color, location, movement, etc, with some columns responding to input from one eye, i.e. ocular (eye) dominance columns (Hubel & Wiesel, 1968, 1974). In general, a strict topographical relationship is maintained throughout the visual projection system and the visual cortex. Within the visual cortex immediately adjacent groups of neurons respond to visual information from neighboring regions within the retina (Kaas & Krubitzer 2001).
Information received in the visual areas of the occipital lobe are then projected dorsally and ventrally to a variety of visual association areas, including the parietal and temporal lobes (Kaas & Krubitzer 2001; Nakamura et al. 1994; Sereno et al. 1995; Tovee et al. 1994). The dorsal stream of visual information flows to and is assimilated by the parietal lobes and is incorporated for the purposes of coordinating body and arm and leg movements in visual space. The parietal (dorsal) visual stream, therefore is most sensitive to objects in the periphery and lower visual fields (Motter & Mountcastle, 1981; Previc 2010); i.e. where the hands, feet, and ground are more likely to be viewed, and which thus enables the parietal lobe to guide and observe the hands in motion.

The ventral visual stream flows from the occipital lobe to the inferior temporal lobe (ITL) where it is assimilated and also serves to activate feature detecting neurons which are sensitive to faces, objects, and other complex geometric stimuli. ITL neurons are sensitive to color, contrast, size, shape, orientation and are involved in the perception of three dimensional objects including specific shapes and forms including the human face (Eskander, et al. 1992; Gross & Graziano 1995; Gross, et al. 1972; Nakamura et al. 1994; Rolls, 1992; Sergent, et al.2010). In consequence, if injured, patients may suffer from an inability to recognize the faces of friends, loved ones, or pets (Braun et al. 1994; DeRenzi, 2006; Hanley et al. 2010; Hecaen & Angelergues, 1962; Landis et al., 2006; Levine, 1978); a condition referred to as prosopagnosia. Some patients may in fact be unable to recognize their own face in the mirror.
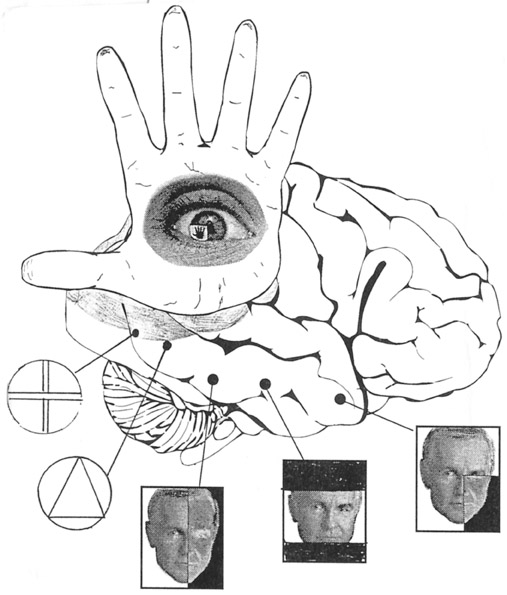
The ventral occipital/temporal and the dorsal occipital/parietal visual areas also interact. For example, the ventral stream tends to focus on objects, faces, and so on, whereas the dorsal stream focuses on the hands in visual space. By interacting, the hands (parietal lobes) can be directed at specific objects identified and targeted by the temporal lobes, thus making coordinated hand-eye coordination possible.
The mosaic which can loosely be defined as consciousness, consists of multiple and parallel streams of information which are processed hierarchically, horizontally, vertically, and in modular fashion, with the brainstem, cerebellum, diencephalon, striatum, limbic system, and neocortex providing their own unique contributions. Conscious processing within the neocortex also occurs hierarchically, vertically, horizontally, as well as in parallel, even within the separate lobes of the brain.
For example, in addition to their layered and columnar organization (Mountcastle, 2012), each of the four lobes of the brain consists of multiple cellular cytoarchitectonic subdivisions which perform unique and/or overlapping functions. For example, each lobe can be subdivided into primary, secondary, association, and assimilation areas. In general, information processing in the sensory receiving areas of the parietal, temporal, and occipital lobes begins in the primary receiving area and then flows to the secondary receiving area and then to the association and multi-modal assimilation areas (Jones & Powell, 1970; Pandya & Yeterian, 1985). However, since the various subdivisions of the thalamus also project to the association areas, these tissues of the mind may receive specific types of visual, or auditory, or somesthetic input in advance of the primary areas (e.g. Zeki, 2012).
In general, however, the flow is from primary to secondary to association area. For example, somatosensory information is first transmitted from the thalamus to the primary receiving areas of the parietal lobe (Brodmann's cytoarchitectonic areas 3,2,1). Here the individual sensory elements are analyzed and localized, e.g., cold, wet, hard, small, cubicle, palm. These sensory impressions are then shunted to the secondary areas (Brodmann's area 5) where these impressions may be combined, e.g. an ice cube held in the hand. These association areas are then transferred to the association-assimilation areas (Brodmann's areas 7, 39, 40), i.e. the inferior parietal lobule where they may then be visualized and even named.
As per the motor areas, the opposite sequence of events takes places, with information flow beginning in the supplementary motor areas (SMA) prior to movement, and prior to activation of the secondary-association areas-- cytoarchitectonic areas 8,6 (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010). The SMA may well initiate movements as this part of the brain has also been associated with what has been described as the "will."
The secondary motor areas also become activated prior to movement and precedes cellular activation of the primary region (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Weinrich et al. 1984). The premotor area appears to program various gross and fine motor activities, and becomes highly active during the learning of new motor programs (Porter 2010; Roland et al. 1981).
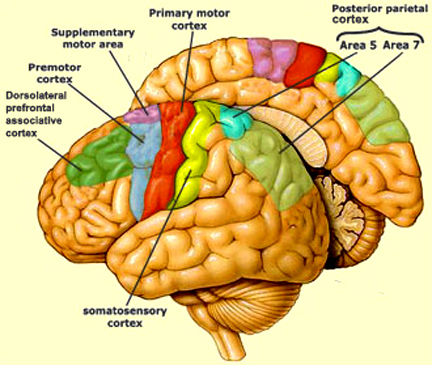
Depending on the task, the primary motor areas may show low level activity prior to movement, but then becomes highly active during movement (Passingham, 1999)--a reflection of the fact that it supplies at least a third of the axonal trunk which makes up the corticospinal tract and whose cells maintain multiple musculature connections (via the spinal cord), as well as a one to one correspondence with specific muscles. In fact, direct electrical stimulation of the frontal primary motor cortex can induce twitching of the lips, flexion or extension of a single finger joint, protrusion of the tongue or elevation of the palate (Penfield & Boldrey, 1937; Penfield & Jasper, 1954; Penfield & Rasmussen, 1950; Rothwell et al. 1987), even though patients never claim to have willed these movements. Again, however, like the sensory areas, different motor subareas may become activated simultaneously, including the primary motor area which may become aroused prior to movement, only to increase in activity during specific movements (Passingham, 2013).
However, the motor areas also interact with the sensory areas in the parietal lobe. In this way, motor movement can be coordinated with sensory impressions arising from the skin, joints, muscles, and body.
Incoming and outgoing signals, therefore, are processed sequentially and in parallel. Beginning in the primary receiving areas these signals are relayed via a variety of separate branches to the secondary receiving areas and to association areas further downstream which are also simultaneously receiving thalamic input, processing this data, and then relaying this information back to the primary areas as well as laterally to other association areas (Kaas, 2013; Panya & Yeterian, 1985; Zeki, 2012) and back to the thalamus and to the frontal lobes. A similar stepwise and parallel process occurs; albeit in reverse, within the motor areas. That is, the primary motor areas in the frontal lobe may also become active prior to movements, but then increase their activity when the movement is actually performed (Passingham, 2012).
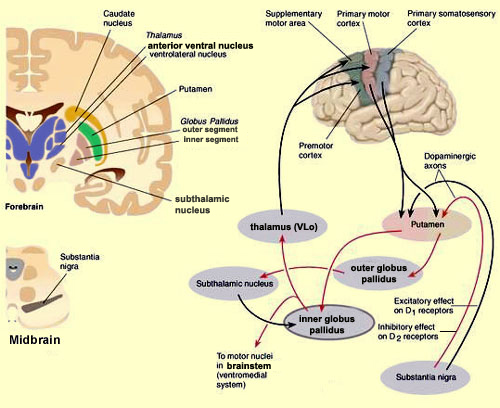
Hence, these divergent cytoarchitectonic areas do not initially act in isolation as multiple areas in diverse regions of the brain become activated simultaneously and interact in regard to perceptual and motor functioning. That is, functions are localized as well as distributed throughout the neuroaxis with different regions making unique and overlapping contributions to the mosaic of the mind.
Those aspects of the mind that we associate with consciousness, language, and rational thought, are clearly maintained and propagated by the neocortical mantle of the frontal, temporal, parietal, and occipital lobes and is divisible horizontally, vertically, hierarchically, cytoarchitecturally, and in accordance with the specialities of the different lobes of the brain including the limbic system and diencephalon. These different temporal and parallel streams of mental activity, including those generating by the structures of the limbic system, coalesce to create a conscious composite, a semi-integrated whole.
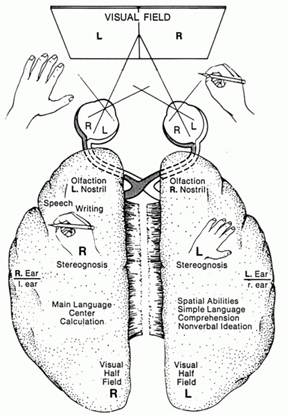

The cerebrum including its striatal, limbic, and diencephalic components, is divisible not only horizontally, vertically, hierarchically, and in regard to its different lobes, but laterally. That is, two semi-independent streams of conscious awareness coexist literally side by side. Each half of the cerebrum, which includes neocortical, striatal, limbic and diencephalic structures, has its own memories, goals, personal identity, and social emotional orientation and capabilities or lack-thereof, as has now been repeatedly demonstrated following surgical sectioning of the thick interhemispheric axonal pathway, the corpus callosum (Akelaitis, 1945; Bogen, 1969, 1979; Joseph, 2008b; Levy, 1974, 1983; Sperry, 1966, 1982).
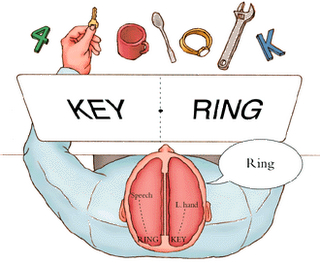
As described by Nobel Lauriate Roger Sperry (1966, p. 299), "Everything we have seen indicates that the surgery has left these people with two separate minds, that is, two separate spheres of consciousness. What is experienced in the right hemisphere seems to lie entirely outside the realm of awareness of the left hemisphere. This mental division has been demonstrated in regard to perception, cognition, volition, learning and memory."
In consequence, following the surgical sectioning of the corpus callosum, each half of the brain and mind may act independently. For example, Akelaitis (1945, p. 597) describes patients with complete corpus callosotomies who experienced extreme difficulties making the two halves of their bodies and the two halves of their brains cooperate, as each half of the brain apparently had its own desires, goals and intentions. A recently divorced male "split-brain" patient noted that on several occasions while walking about town he found himself forced to go some distance in another direction by the left half of his body. Specifically, whereas the speaking, language dominant left half of his brain just wanted to go for a walk, the right half of his brain was trying to walk toward the new home of his former wife.
A "split-brain" patient I examined, who had recently broken up with his girlfriend, indicated thumbs down and stated matter-of-factly that he had absolutely no desire to see her again. However, when his (non-verbal) right hemisphere was asked how it felt about the situation, and was told to give a thumbs up or down, it gave a thumbs up (with the left hand) when asked if he still wanted to see her.
The right hemisphere of another split-brain patient apparently felt considerable animosity for his wife, for it slapped her several times----much to the embarrassment of his left (speaking) hemisphere. In another case, a patient's left hand attempted to choke the patient himself and had to be wrestled away. The right and left hemisphere of yet another split-brain patient enjoyed different television programs, different foods, and had different attitudes even about mundane activities such as going for a walk (Joseph, 2008b).
For example, while watching television one afternoon, this patient "2-C" reported that to the dismay of his left (speaking) hemisphere, he was dragged from the couch by his left leg, and that the left half of his body dragged him to the TV where his left hand then changed channels even though he (or rather his left hemisphere) was enjoying the program. On another occasion, it simply turned the TV off and tried to leave the room. Once, after he had retrieved something from the refrigerator with his right hand, his left hand took the food, put it back on the shelf and retrieved a completely different item, "Even though that's not what I wanted to eat!" his left hemisphere complained. On at least one occasion, his left leg refused to continue "going for a walk" and would only allow him to return home.
2-C was so annoyed with the independent actions of the left half of his body that he frequently expressed hate for it, even striking it angrily with the right hand. Nevertheless, the right hemisphere knew exactly what it was doing, as was demonstrated experimentally, and thus had its own goals, desires, intentions, and favorite foods and TV shows--even showing the good sense of turning off the TV and leaving the room (Joseph, 2008b).
THE NEUROANATOMY OF MIND
In summary, the human mind and brain are functionally lateralized, sexually differentiated, and hierarchically, vertically, and horizontally organized, and are significantly shaped and effected by experience. Considered broadly, it can be said that the most unconscious and reflexive aspects of the "unconscious" mind are associated with the brainstem.
The diencephalon, which is immediately anterior and adjacent to the brainstem, is associated with a vague sensory affective unconscious, such that pre-conscious cognitive-sensory processing (thalamus) and reflexive emotional processing (hypothalamus) takes place in this region--information which may be relayed to the neocortex as well as to the limbic system.
The limbic system, which is anterior to the diencephalon, and is both dorsally and ventrally situated, is capable of exceedingly complex and sophisticated mental activity and can process, analyze, and learn and remember complex cognitive, linguistic, visual-spatial, and affective material, as well as generate complex emotions ranging from love to hate. As will be detailed in chapters 13 and 15, the limbic system can also vocalize and it can think, and it can transfer this information to the overlying neocortex which it may impel to act on its desires and its fears.
The neocortex is associated with the more rational and logical aspects of the mind. It is this neocortical shroud which envelops and coats the cerebrum with six to seven layers of gray matter, which cogitates, speaks in words and sentences, and can reason, rhyme, plan for the future, as well as ponder and analyze its own brain and mind.
The mind, however, is also a continuum, and the human brain is a composite of interacting structures which are intimately interconnected and which perform a variety of unique and overlapping functions often in parallel. And yet, although these structures interact and often engage in parallel processing, they are also functionally specialized, with some areas, such as the brainstem and limbic system, often acting completely independently of the neocortex or what is classically referred to as the conscious mind.
OVERVIEW: CONSCIOUSNESS, AWARENESS, AND THE NEUROANATOMY OF MIND
The brain and the mind are synonymous, for if the brain is damaged, so too is the mind. The brain and associated mental activity are functionally lateralized and hierarchically organized, such that specific cognitive and emotional functions may be localized to specific regions of the brain. There are phylogenetically older, sensory-motor non-emotional mental systems associated with the brainstem, and a highly complex emotional and memory system associated with the limbic system.
Whereas the brainstem is incapable of conscious or cognitive activity, and instead mediates reflexive motor acts, including breathing, heart rate and arousal, it is the limbic system which mediates the ability to feel love or sorrow or to determine if something is good to eat. Indeed, it is the limbic system which enables humans to form memories of the long ago, as well as to recall these memories in order to dream of the future, as specific limbic nuclei become active not only when learning and remembering, but when dreaming.
In fact, the limbic system provides humans (and perhaps non-humans) with the capacity to experience the most profound of emotions, from love to spiritual ecstasy and religious awe, serving, at its most profound, as perhaps even a transmitter to god.
By contrast, the more recently evolved neocortex of the right hemisphere is the domain of a highly evolved social-emotional, visual-pictorial, spatial, body centered awareness and employs emotional and melodic sounds for expression (Bogen, 1969, 2013; Borod et al., 1992; Cimino et al., 2001; Freeman & Traugott, 2013; Galin, 1974; Heilman & Bowers 1996; Hoppe, 1977; Joseph, 2008ab; Levy, 1974, 1983; Ross, 2013; Sperry, 1966, 1982). The right half of the brain is responsible for discerning distance, depth and movement, and recognizing environmental and animal sounds such as a chirping bird, a buzzing bee, a babbling brook, or a thunderstorm, as well as the capacity to sing, dance, chase or throw something with accuracy, and run without falling or bumping into things. Whereas the left hemisphere is concerned with logic and grammatical rules of organization and expression, including the analysis of details, sequential units, and parts, it is the right hemisphere which is able to perceive events or stimuli as a whole and which can see the forest as well as the trees. Whereas right hemispheric activity is associated with the production of the visual, emotional, hallucinatory, hypnogogic aspects of dreaming, the left hemisphere tends to immediately forget the dream upon waking (Joseph, 2008a).
Hence, in the right hemisphere we deal with a non-verbal form of awareness that accompanies in parallel the temporal-sequential, language dependent stream of consciousness associated with the functional integrity of the left cerebrum. In fact, and as has been repeatedly demonstrated, the right and left cerebral hemisphere are each capable of self-awareness, can plan for the future, have goals and aspirations, likes and dislikes, social and political awareness, and can independently and purposefully initiate behavior, guide responses choices and emotional reactions, as well as recall and act upon certain desires, impulses situations or environmental events --sometimes without the aid, knowledge or active (reflective) participation of the other half of the brain.
Thus, in summary, the right and left hemisphere subserve almost wholly different mental systems--a function of evolutionary metamorphosis and the evolution of language and tool making capabilities-- whereas the limbic system is synonymous with the most archaic regions of the emotional, sexual, unconscious mind. However, because the brain and mind are hierarchically organized and lateralized, and as the limbic system retains the capacity to completely overthrow and hijack the neocortically equipped "rational" and emotionally intelligent mind, conscious-awareness, as well as the brain, are subject to fracture, with each isolated segment acting on its own wishes and desires, independent of and sometimes conflicting with those mental systems that may remain intact such as those associated with the language-dependent conscious mind.















