Rhawn Gabriel Joseph, Ph.D.
(reprinted, updated from, Neuroscience, Academic Press, 2000)
In response to the threat of bodily injury or death, animals, including humans, may fight, take flight, or fight while attempt to flee--reactions mediated by the limbic system and limbic striatum / basal ganglia. Under conditions of overwhelming terror, or in reaction to prolonged and repetitive instances of severe stress and the subsequent secretion of stress hormones and excessive activation of limbic structures, victims may become so frightened they become paralyzed with fear (Galliano, Noble, Travis, & Puechl, 2013; Miller, 1951; Nijenhuis, Vanderlinden, & Spinhoven, 2011; Krystal, 2003), enter trance-like states, experience numbing and out-of-body "hallucinations" and may then suffer varying degrees of memory loss and/or an inability to forget coupled with flashbacks and intrusive trauma-related imagery (Fink, 1999; Foa, Riggs, & Gershuny, 1995; Joseph, 2011b, 1999d; Krystal, 2003; Moller, et al., 2011).
Cessation of movement, for example, may prevent a predator or a hunter, from spying their prey. Soldiers and Airmen who have found themselves behind enemy lines have commonly escaped capture by simply lying still. There have been numerous reports of enemy soldiers nearly stepping on these men, but nevertheless failing to see them. However, under some circumstances, such as a sinking ship, or burning aircraft, paralytic fear may instead result in death (Krystal, 2003).
Similarly, ruminating about the consequences of a catastrophe, or a miraculous escape from death, can have its benefits; that is, as a learning experience and as a form of analysis which enables one to avoid or to take more effective action if similar misfortune arises in the future. Of course, there is nothing adaptive about sudden flashbacks, nightmares, intrusive imagery, or an inability to forget, as the victim is simply being traumatized. In fact, it is the continual traumatization, that is, at the level of the central nervous system, coupled with the experience of trauma-related environmental stimuli, which provokes these flashbacks; a consequence of stress-induced injury to the amygdala, hippocampus, hypothalamus, and related structures and neural pathways.
THE NEURAL CIRCUITRY OF STRESS
During periods of extreme emotional stress, pain, and fear, and as part of the "fight or flight" response, the hypothalamic-pituitary-adrenal (HPA) axis prepares the brain and the body for possible catastrophic consequences by secreting large amounts of the amino peptide, corticotropin-releasing factor (CRF) which activates the andenohypophysis which begins secreting ACTH which stimulates the adrenal cortex which secretes glucocosteroids which interact with norepinephrine and other neurotransmitters (Fink, 1999; Moller, et al., 2011). Specifically, the anterior hypothalamus releases CRF, the pictuitary releases ACTH, and the outer layer of the adrenal glands secrete glucocorticoids and mineralalocorticoids. The primary glucocorticoid is cortisol (which mobilizes free fatty acids from adipose tissue, breaks down proteins, and increases blood pressure) and the primary mineralocorticoid is aldosterone.
These neurochemical events and stress stress hormones potentiate behavioral and autonomic reactions when confronted with or following bodily injury, and thus provide protective as well as activating influence which enables the organism to continue to function and thus escape or fight for their life. However, cortisol can also injure pyramidal cells, and can decrease the production of lymphocytes by the lymph nodes and thymus gland. Since a primary role of lymphocytes is immunological (e.g. to destroy invading bacteria), excessive cortical secretion can disrupt the immune response thus increasing the likelihood that one may get sick and die.
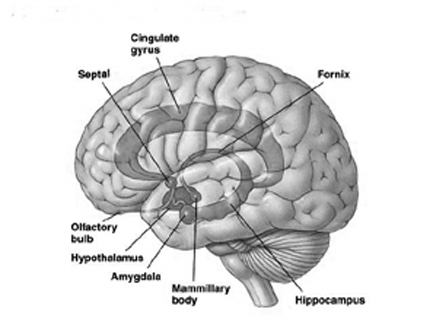
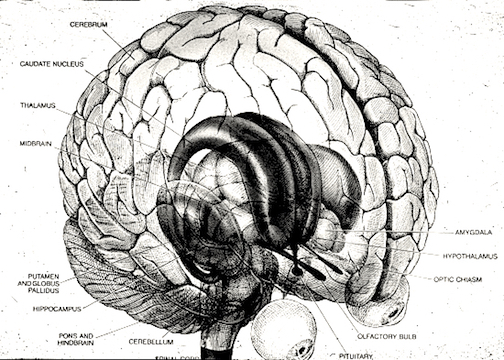
The neural circuitry of stress begins with peripheral receptors which directly transmit fear, pain, and stress-inducing signals to the brainstem locus ceruleus (LC) and periaqueductal gray, and the thalamus--structures which relay this information to the hypothalamus and amygdala, as well as to the entorhinal cortex (the "gateway to the hippocampus"), and the cingulate and orbital and medial frontal cortex (Fink, 1999; Moller et al.,2011). However, it is the hypothalamus and amygdala, as well as the autonomic nervous system, which play central roles in the stress response including the generation of fear and anxiety.
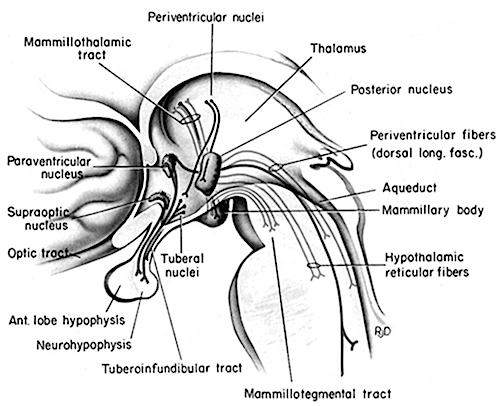
Specifically, in addition to influencing the HPA axis, the lateral and ventro-medial hypothalamus act on the sympathetic (SNS) and parasympathetic nervous system (PNS), respectively. Hence, with lateral hypothalamic/SNS activation, there is an increase in arousal as well as in blood pressure and heart rate--bodily changes which are also mediated by the amygdala. These neural circuits act directly on the brainstem, including the vagus nerve which controls heart rate. In addition, the amygdala projects to and is intimately interconnected with the limbic and corpus striatum (see chapter 16), and through these pathways can trigger running, kicking, punching, and flailing, and thus the motor components of the fight or flight response, including "freezing" and immobilization during periods of extreme fear.
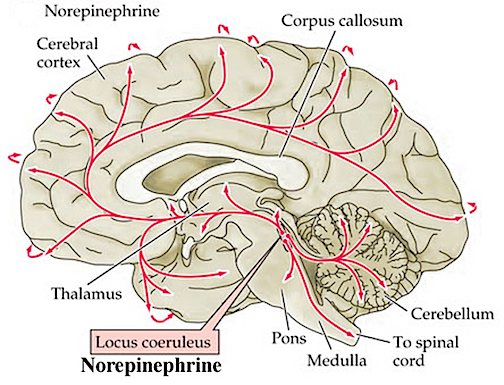
As noted, peripheral sensory receptors project directly to the LC, and under conditions of stress and in reaction to amygdala activation, LC neurons rapidly increase their rate of firing as well as their secretion of norepinephrine (NE). Thus, NE levels initially dramatically increase within the amygdala and hypothalamus (Bliss et al., 1968). Moreover, serotonin (5HT), dopamine (DA), corticosteroid, and enkephalin levels and blood flow to the temporal lobes also dramatically increase (Rosenblum, Coplan, & Friedman, 2009; Southwick et al., 2013). However, whereas subcortical NE levels initially increase, as stress becomes prolonged, NE (and 5HT) levels and NE receptor densities decrease in the amygdala as well as the hippocampus (Bliss et al., 1968), which exposes these structures to injury secondary to cortisol and enkephalins. In fact, these NE reductions trigger the activation of the HPA axis which results in the increased secretion of cortisol (Lupien & McEwen, 2007). Normally, cortisol secretion is subject to the tonic influences of NE; whereas cortisol can indirectly reduce NE synthesis.
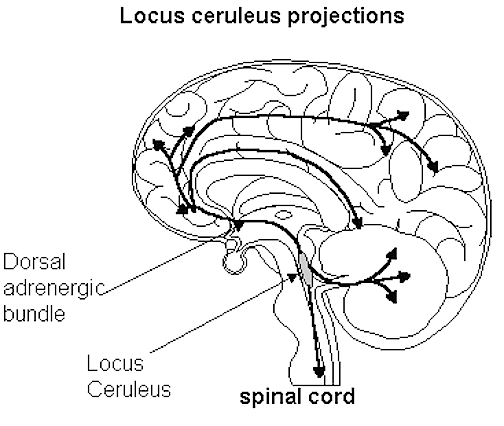
Thus a complex feedback system is maintained via the interaction of these substances and the amygdala, HPA axis and the LC; which when activated and secreted determine the stress response, and which provide protective as well as activating influence which enables the organism to continue to function and thus escape or fight for their life. Unfortunately, under conditions of chronic or repetitive stress, various subcomponents of this complex neural circuitry may be injured, thus disrupting the stress response and in some cases giving rise to profound disturbances of emotion, memory, and personality (Joseph, 2011b, 1999d). Indeed, under these conditions, neural tissue such as the amygdala and hippocampus may be injured, thereby giving rise to long term maladaptive reactions, such as memory loss, flashbacks, anxiety, and an enhanced startle reaction.
THE AMYGDALA AND FEAR
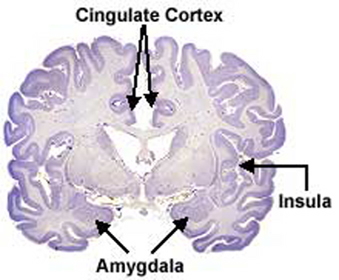
The amygdala is preeminent in the control and mediation of most higher order emotional and motivational activities (Davis et al., 2007; Fukuda, Ono, & Nakamura, 1987; Gaffan, 2007; Gloor, 2007; Halgren, 2007; LeDoux, 2005; Rolls, 2007; Rosen & Schulkin, 2011). Through massive interconnections maintained with the brainstem, thalamus, hypothalamus, septal nucleus, hippocampus, cingulate, medial forebrain bundle and the temporal, occipital, parietal and frontal lobes (Amaral, Price, Pitkanen, & Thomas, 2007; McDonald, 2007; Mehler, 1980; Russchen, 2002; Turner, Mishkin, & Knapp, 1980), the amygdala is able to sample and affect the perception of auditory, somesthetic, and visual stimuli as well as scrutinize this information for emotional significance (Amaral et al., 2007; O'Keefe & Bouma, 1969; Ono & Nishijo, 2007; Perryman, Kling, & Lloyd, 1987; Rolls 2007; Schutze, Knuepfer, Eismann, Stumpf, & Stock, 1987). This includes the ability to discern and express even subtle social-emotional nuances such as friendliness, fear, love, anger, or threat, and at a more basic level, determine if something might be good to eat.
However, the amygdala also promotes emotional and mood congruent behavior, including flight or fight. It is also becomes highly active when subjects think about or are presented with traumatic stimuli (Rauch et al., 2005, Shin et al., 2007) and mediates the stress response (Henke, 2007). In response to stress, the amygdala (in addition to other limbic nuclei) will secrete massive amounts of opiate-like substances (enkephalins) and will stimulate the secretion of corticosteroids. The amygdala is in fact rich in cells containing enkephalins, and opiate and corticosteroids receptors can be found throughout the amygdala (Atweh & Kuhar, 1977; Uhl, Kuhar, & Snyder, 1978). In this regard, the amygdala is implicated in the generation of extreme pleasure, or it can induce analgesic-opiate-induced numbing if injured during the course of fight or flight.
Extreme fear is the most common emotional reaction elicited with direct electrode stimulation of the human or non-human amygdala (Chapman, 1960; Davis et al., 2007; Gloor, 2007; Halgren, 2007; Rosen & Schulkin, 2011; Strauss, Risser, & Jones, 2002; Williams 1956). The pupils dilate and the subject will cringe, withdraw, and cower. This cowering reaction may give way to extreme panic and the animal will attempt to take flight.
Likewise, abnormal activity originating in the amygdala and/or the overlying temporal lobe can evoke overwhelming, terrifying feelings of "nightmarish" fear that may not be tied to anything specific, other than perhaps the sensation of impending death (Herman & Chambria, 1980; Strauss et al., 2002; Weil, 1956). With amygdala activation the EEG becomes desynchronized (indicating arousal), heart rate becomes depressed, respiration patterns change, the galvanic skin response significantly alters, the face contorts, the pupils will dilate, and the subject will look anxious and afraid (Bagshaw & Benzies, 1968; Davis, 2007; Kapp, Supple, & Whalen, 2009; Ursin & Kaada, 1960).
Unlike hypothalamic on/off emotional reactions, amygdala-fear reactions can last up to several minutes after the stimulation is withdrawn. Moreover, amygdala pathways may remain potentiated for minutes, hours, and even days following fear induced stimulation (Clugnet & LeDoux, 1990). Hence, the amygdala continues to process information in the abstract even when no longer observable (O'Keefe & Bouma, 1969). In consequence, the subject may continue to feel extreme terror long after the threat has been removed. In fact, amygdala-induced fear may be re-evoked even by otherwise neutral cues or stimuli associated with the original threat or trauma (Davis, 2007; LeDoux, 2005).
However, with extreme fear, the individual may not fight or take flight, but they may become literally petrified with fear. Likewise, with amygdala stimulation, there may be a complete arrest of movement (Applegate, Kapp, Underwood, & McNall, 2003; Gloor, 1960; Ursin & Kaada, 1960), and the subject may even briefly cease to breath, as if dead.
FIGHT, FLIGHT, AND FEAR PARALYSIS
Under conditions of extreme fear, victims may enter trance-like states, and become so paralyzed and numb with fear that they may appear catatonic, and may fail to make any effort to resist or to save their lives (Courtois, 1995; Galliano et al., 2013; Miller, 1951; Nijenhuis et al., 2011). Indeed, be it animal or human, a common response to extreme fear is to become motionless, and in the extreme, catatonic as if rigor mortis has set in; also referred to as "death-feigning" or "playing possum" (Kalin 2013; Krystal, 2003; Nijenhuis et al., 2011; Stern, 1951). In fact, the affected individual may become so stiff and rigid, that it may appear as if rigor mortis has set in. Moreover, the victim may not only become unresponsive, but psychologically and emotionally numb (Courtios, 1995; Foa, Riggs, & Gershuny, 1995; Krystal, 2003) and suffer a complete blocking off of cognition and memory such that they may resist and fail to respond even to attempts at assistance (Krystal, 2003).
For example, over 25% of rape victims report body stiffening (Galliano et al., 2013), whereas in air and sea disasters 10-25% of the victims may become frozen, stunned, and immobile (Krystal, 2003). Those afflicted will also fail to take any action to save their lives, such as attempting to evacuate a burning or sinking craft even though they have been uninjured. The airline industry has referred to these fear-induced stiffening reactions as "frozen panic states."
Fear-driven catatonic-paralytic states are prevalent in the animal kingdom (Kalin, 2013; Nijenhuis et al., 2011) and constitute an adaptive, life preserving reaction that appears to be largely mediated by the amygdala, supplementary motor area, and striatum (see below), coupled with the secretion of enkephalins and corticosteroids, and alterations in dopamine and norepinephrine turnover. Indeed, the amygdala not only mediates the behavioral reactions to fear including the fight or flight response (Davis et al., 2007; Gloor, 2007; Halgren, 2007; LeDoux, 2005; Rosen & Schulkin, 2011), but, with direct electrical stimulation the amygdala may become hyperactivated and there is a complete arrest of movement such that even breathing may temporarily cease (Applegate, et al 2003; Kapp, et al. 2007; Ursin & Kaada, 1960). Likewise, under conditions of overwhelming terror--an emotion mediated by the amygdala-- the victim may become catatonic and petrified with fear (Courtois, 1995; Galliano et al., 2013; Miller, 1951; Nijenhuis et al., 2011).
Again, however, under certain conditions fear paralysis may be exceedingly adaptive. Since movement and motion typically alerts a predator to potential prey and thus triggers attacking behavior, lack of movement sometimes eliminates it. Hence, by freezing and not-moving, predators may cease to respond or even fail to note the presence of potential prey. Moreover, unless exceedingly hungry, some predators avoid food that appears to be already dead, though just as commonly the hapless victim is ripped to pieces and readily consumed.
Fortunately, even if this stratagem fails, hyperactivation of the amygdala may induce a numbing reaction thus ensuring a merciful death. Indeed, the amygdala not only mediates the fight, flight, or fear-induced freezing reaction (Applegate et al., 2003; Kapp et al., 2007; Ursin & Kaada, 1960), but under high levels of stress, fear and arousal, it secretes and triggers the secretion of massive amounts of enkephalins as well as corticosteroids and other stress hormones via the hypothalamic- pituitary-adrenals (Hakan, Eyle, & Henriksen, 2009; Roozendall, Koolhaas & Bohus, 2007). In general, these stress hormones and neurotransmitters initially facilitate motor responsiveness and the narrowing of attention. However, high levels of enkephalins and corticosteroids are also associated with numbing, analgesia, a reduction in pain perception coupled with a blocking of cognition and memory loss.
It is the massive secretion of opiates which likely produce the narcotic-like bliss associated with "near death experiences," and the numbing which enables a severely wounded warrior to keep fighting, or a hunted and wounded beast to lie down and calmly allow itself to be eaten alive. Similarly, the trauma or fear-induced release of enkephalins and corticosteroids may account for the psychological numbing reported by victims of catastrophe, or sexual or physical trauma (Courtios, 1995; Foa et al., 1995) as well as the tendency of some victims to become so numb with fear that they fail to take any action to save their life.
Moreover, the continued secretion of corticosteroids and enkephalins are directly associated with the development of immobility, cataplexy, paralysis, and catatonia (Amir, Brown, Amit, & Ornstein, 1981; Fenselow, 1986; Kalin, Shelton, Richman & Davidson, 2011). Catatonia, coupled with emotional and physical (enkephalin-induced) numbing, also represents a total surrender reaction, a complete loss of will, where the victim enters a trance-like state and simply loses the will to resist or to live. Hence, prey may cease to run or fight and may simply stand still or lie down and allow predators to literally eat them alive. Under life threatening conditions, some humans, in fact become so numb with fear that they passively allow themselves to be raped, robbed, assaulted or marched into a ditch and murdered (Galliano et al., 2013; Krystal, 2003). During World War II, tens of thousands of European Jews obeyed orders in an automaton, trance-like fashion, and would take of their clothes, and together with their husbands, wives and children, descend into pits and passively allow themselves to be shot. A fear-induced trance-like paralysis of will, therefore, coupled with emotional and enkephalin-induced numbing and cognitive blocking, gives rise, therefore, a total surrender reaction, usually as a prelude (and hopeful guarantee) of a "merciful" death.
THE AMYGDALA-STRIATUM AND FEAR PARALYSIS
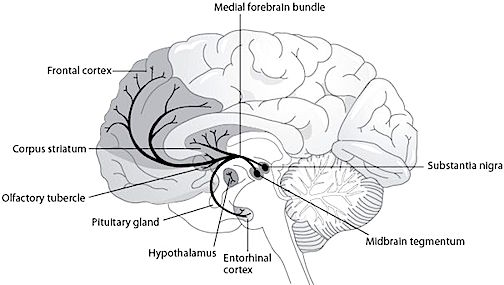
The amygdala is buried within and is contiguous with the anterior-inferior-medial temporal lobe and consists of several major nuclear groups including what has been referred to as the "extended amygdala" (Heimer & Alheid, 1991). The extended amygdala is essentially contiguous with the limbic striatum: the olfactory tubercle, nucleus accumbens, and substantia innominata (ventral globus pallidus). In addition, the corpus striatum (caudate and putamen) forms a bulbous extension of and in fact evolved from the amgydala. The human amygdala arches in a posterior-dorsal-anterior loop and becomes the corpus striatum and is the first portion of the striatal complex to appear during embryological development. The caudate nucleus is in fact dominated by axons from the inferior temporal lobe and amygdala (Amaral, Price, Pitkanen, & Thomas, 2007; Heimer & Aheid, 1991). However, the caudate is also able to indirectly influence and even inhibit amygdala activity; accomplished through the nigrostriatal dopamine system (Amato et al. 2002).
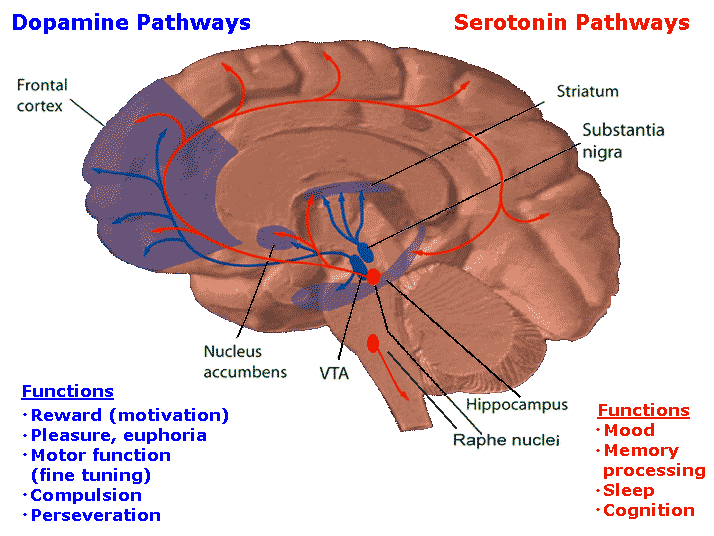
In part, the striatum has evolved in order to serve as an emotional-motor interface so that limbic needs and amygdala-triggered impulses may be acted on in a stereotyped and efficient manner so as to facilitate escape, attack, or communication (MacLean, 1990; Mogenson, 1987). In response to amygdala activation, the striatum (including the subthalamic nucleus) may trigger a variety of stereotyped and ballistic motor actions such as running, kicking, and punching, or conversely "freezing" in reaction to extreme fear. Likewise, because all humans possess a striatum and limbic system that developed, evolved, and is organized in an identical manner, when happy, sad, angry, and so on, the facial and body musculature assumes the same readily identifiable emotional postures and expressions regardless of culture or racial orgins (Ekman, 2013; Eible-Ebesfedlt, 1995; Joseph, 2013).
Hence the amygdala and corpus and limbic striatum constitute a functional unit and interact in producing and coordinating gross, or whole body motor activity in reaction to emotional and motivational concerns (Everitt & Robbins, 2007; Heimer & Alheid, 1991; MacLean, 1990; Mogenson & Yang, 1991). However, the amygdala and striatum are also intimately associated with the medial frontal lobe and the supplementary motor areas (Amaral et al., 2007; McDonald, 2007); structures which similarly contain high levels of dopamine. Presumably, it is through its striatal, motor-neocortical and dopamine interconnnections that the amygdala is able to guide, direct, or trigger a variety of emotionally-motivated movement programs, or conversely, a complete cessation of movement, including fear-induced rigidity and catatonia.
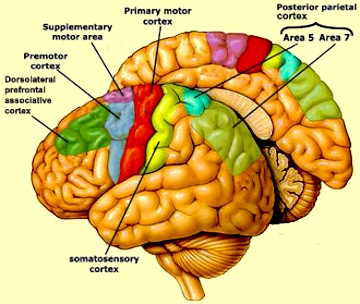
The amygdala, supplementary motor areas (SMA) within the medial frontal lobes and corpus striatum are an integral component of a very elaborate motor-feedback loop which includes the motor thalamus, motor neocortex, and the brainstem (Alexander & Crutcher, 1990a,b; Alexander, Crutcher, & DeLong, 1990; Mink & Thach, 1991; Parent, 2005); motor structures which project to and mutually influence one another as well as rely on the dopamine neurotransmitter system. However, these structures also interact in a step-wise fashion, such that activation of the SMA precedes activity in the association (secondary) motor cortex which is then followed by primary motor activity and finally the activation of the cranial or spinal nerves, thus inducing specific movements (Alexander & Crutcher, 1990a,b; Alexander et al., 1990; Mink & Thach, 1991).
Likewise, when frightened, these areas act in a coordinated fashion so as to mediate purposeful motor movements, with the exception that activation of the amygdala, SMA and striatum take precedence over the primary motor cortex which is concerned with fine motor movements involving the hands and fingers. As noted, the striatum and SMA (including the subthalamic nucleus) are exceedingly important in producing stereotyped motor acts, including running, biting, or ballistically flailing with arms and legs so as to hit, kick, and punch any predator or aggressor.
Petrified with Fear: Catatonia, The Amygdala, SMA, and Striatum
As noted, in response to extreme stress, prolonged terror or depth electrode amygdala stimulation, the subject may cower, run away or become petrified with fear. These behaviors appear to be mediated by the dopamine neurotransmitter system, and the SMA and caudate which are very susceptible to the disruptive influences of stress. In fact, hyperactivation or depth electrode stimulation of these structures can induce a catatonia coupled with a complete surrender reaction and a loss of "will" (Hassler, 1980; Joseph, 1999a; Laplane, Talairach, Meininger, Bancaud, & Orgogozo, 1977; Luria, 1980; Penfield & Jasper, 1954; Penfield & Welch, 1951); that is, unless the amygdala has first been destroyed (Spiegel & Szekely, 1961).
In fact, as based on regional cerebral blood flow and EEG studies that there activity significanly increases in the medial frontal (right caudal anterior cingulate sulcus) and inferior frontal lobes during hypnotic induction suggestion (Rainville, et al., 1999). This is significant in that hypnotic states are also associated with a complete surrender reaction and a loss of "will."
Under stressful conditions or those involving direct electrical or chemical activation of the SMA or caudate, the individual will become rigid, stiff, and unmoving, and will cease to speak or react to questions, threats, or external stimulation (Goto et al., 1990; Hassler, 1980; Joseph, 1999a; Kish et al., 2003; Laplane et al., 1977; Luria, 1980; Spiegel & Szekely, 1961). After the stimulation is removed, patients have reported that their mind essentially had become a blank, a void, and that thoughts no longer entered their head.
Identical disturbances, including the gegenhalten and waxy flexibility of fear-induced catatonia and death feigning, are produced with tumors, strokes, or lesions of the medial SMA and adjoining tissues (Feeman & Watts, 1942; Gasper et al., 1991; Hornykieciz, 2002; Playford et al., 2007). For example, if a physician attempts to move the patient's arm, it will become stiff and rigid (i.e. gegenhalten). Freeman & Watts (1942, pp 46-47) described one individual who developed waxy flexibility, catatonia and related symptoms after a gunshot wound that passed completely through the medial frontal lobes and SMA. "The patient lay in a catatonic-like stupor for two months, always upon one side with slightly flexed arms and legs, never changing his uncomfortable position. He did not obey commands, but if food and drink were given to him, he swallowed them naturally. He was incontinent, made no complaints, gazed steadily forward, showed no interest in anything and could not be persuaded to talk."
However, in other instances (i.e. waxy flexibility), the patient display an inability to resist and will behave like an automaton that is completely under the control of the examiner. If the arm is placed or forced into a new or awkward position, the arm will either remain in that posture or very slowly return to a normal resting position. Such patients might remain in odd and uncomfortable positions for exceedingly long time periods and make no effort to correct the situation. Moreover, they may not respond to painful stimulation.
TRAUMATIC STRESS AND TRANCE STATES
In some respects, waxy flexibility is reminiscent of and magnetic apraxia in which there is also a loss of will and where the individual responds to external stimulation and display a complete inability to resist. Moreover, these behaviors are reminiscent of the loss of will and others behaviors induced while under hypnotic trance.
For example, under hypnosis, a highly hypnotizable subject will display a complete surrender of will, will allow their arms and legs to be placed in odd and uncomfortable positions which they will maintain, and they may be unresponsive to cold and hot stimulation. Moreover, medial frontal (right anterior cingulate) and bilateral inferior frontal activity has been shown to significantly increase among subjects who have been hypnotized, as based on regional cerebral blood flow and EEG studies (Rainville, et al., 1999).
Moreover, these conditions are reminiscent of the trance-like states and he loss of will displayed by those who have been profoundly emotionally traumatized. As noted, uring World War II, tens of thousands of European Jews obeyed orders in an automaton, trance-like fashion, and would take of their clothes, and together with their husbands, wives and children, descend into pits and passively allow themselves to be shot.
It is also noteworthy that individuals who have a history of severe and profound emotional trauma are easily hypnotized (Hilgard, 1974; Nash & Lynn, 1985; Nemiah, 1979; Putnam, 1986). In fact, even "normal" non-abused children tend to be susceptible to hypnosis (London & Cooper, 1969). In fact, because children are also easily hypnotized and can in fact induce self-hypnosis, coupled with this neurological immaturity, they may thus be more at risk for entering trance-like states and developing dissociative disturbances of personality.
In the hypnosis literature there have also been reports of a "hidden observer;" a dissociated portion of consciousness that passively observes all that occurs (Hilgard, Hilgrad, Macdonald, Morgan, & Johnson, 1978; Spanos, deGroot, Tiller, Weekes, & Betrand, 1985). As noted, dissociated "observers" have also been produced during the course of electrode stimulation and seizure activity originating in the temporal lobe. Patients will report that they have left their bodies and are hovering upon the ceiling staring down at themselves (Daly, 1958; Penfield, 1952; Penfield & Perot 1963; Williams, 1956). That is, their consciousness and sense of personal identity appears to split off from their body, such that they experience themselves as two different people, one passively watching, the other being observed. One female patient claimed that she not only would float above her body, but would sometimes drift outside and even enter into the homes of her neighbors. Possibly, if this dissociative reaction is induced under conditions of abuse and during the development of a trance-like state, the dissociated aspect of consciousness may develop as a separate personality.
Hence, traumatic conditions may injure the medial frontal lobes and hippocampus and amygdala-striatum, thus giving rise to trance-like states, a loss of will, and a complete fear paralysis.
DOPAMINE, TERROR, CATATONIA, AND FEAR-PARALYSIS,
Paralysis and disturbances of movement are also associated with profound dopamine (DA) depletion or following neurochemical lesions within the striatum or the SMA (Goto, Hirano, & Matsumoto, 1990; Kish, Shannak, & Hornykiewicz, 2003; Spiegel & Szekely, 1961). The body becomes stiff and rigid, the face may become frozen and mask-like, and the patient may have extreme difficulty initiating speech or responding to questions.
As noted, the striatum, SMA, and amygdala are richly innervated with DA. One of the principles effects of DA is inhibition (Ellison, 2009; Le Moal & Simon 1991), and the amygdala has a higher concentration of DA than the caudate nucleus or SMA (Stevens, 2007). In part, DA acts to modulate amygdala as well as striatal and SMA activity (Amato et al., 2002; Ben-Ari & Kelly, 1976).
Initially, in reaction to fear or emotional (or amygdala) arousal, DA production is increased (Le Moal & Simon 1991). Increased DA production enables the amygdala-striatal-SMA motor centers to filter irrelevant movements or actions and to selectively engage life-preserving ballistic motor acts such as running, kicking, and failing with the extremities (chapter 16).
With continued stress DA levels may continue to increase unabated, thus inducing complete motor inhibition, and/or (in some individuals) DA may eventually become reduced inducing motor hyperactivation and hypertonia (Gasper et al., 1991; Hornykieciz, 2002; Le Moal & Simon, 1991; Playford et al., 2007); i.e. a paralysis of movement. Hence, in either case the organism may freeze, fall to the ground, and cease to move, blink or even breathe--or breathe only shallowly and slowly. The creature therefore appears to be in a state of rigor mortis and thus dead, i.e. catatonic.
Under conditions of prolonged and excessive stress, significant alterations in DA turnover occur within the amygdala and the medial frontal lobes and SMA (Gasper et al., 1991; Hornykieciz, 2002; Playford et al., 2007), thereby disrupting this emotional-motor feedback loop and creating amygdala and striatial hyperactivity (Amato et al., 2002; Ben-Ari & Kelly, 1976); conditions which also promote corticosteroids and enkephalin production (see below).
Loss of DA is directly associated with the development of tonic EMG activity, motor neuron hyperactivity and thus excessive tonic excitation of the musculature, thereby producing limb and facial rigidity (chapter 16). These conditions also afflict those with Parkinson's disease--a disturbance characterized by extreme rigidity. Indeed, Parkinson's disease is associated with a massive loss of DA neurons in the substantia nigra and an 80% decrease in corpus striatal DA (Goto et al., 1990; Kish et al., 2003); disturbances which also induce amygdala hyperactivity (Amato et al., 2002; Ben-Ari & Kelly, 1976).

Individuals suffering from chronically low levels of DA, including those with Parkinson's disease, are sometimes described as excessively aroused and to suffer from heightened autonomic nervous system activity (Stacy & Jankovic, 2007). Moreover, many Parkinson's patients experience not only rigidity and akinesia, but an episodic freezing of movement (Dietz, Goetz, & Stebbins, 1990; Pascual-Leone, Valls-Sole, & Brasil-Neto, 2009), a tendency to easily fall (Stacy & Jankovic, 2007) an impairment of "righting reflexes" (Calne, 2009), and a reduced capacity to blink (Freedman, 2007) and even to breathe (Stacy & Jankovic, 2007); conditions similar to frozen-panic states and fear-induced catatonia. Like those who are severely frightened, Parkinson's patients also sometimes "shake like a leaf," and display a 4-8 c/sec. ("pill rolling") resting tremor which is exacerbated by stress (Freedman, 2007; Stacy & Jankovic, 2007). In addition, just as those who become catatonic and "petrified with fear" experience an eclipsing of cognition, sigificant cognitive deficits are common among those with reduced DA and/or Parkinson's disease (Freedman, 2007). Thus, DA is a major factor in the catatonic-fear reaction.
Norepinephrine, Enkephalins, Corticosteroids, and Traumatic Stress
The amygdala and limbic striatum are rich in cells containing enkephalins, norepinephrine (NE), and DA. Moreover, opiate, NE, DA, and corticosteroid receptors can be found throughout these structures, especially the amygdala (Atweh & Kuhar, 1977; Ellison, 2009; Hakan, Eyle, & Henriksen, 2009; Roozendall, Koolhaas & Bohus, 2007; Uhl, Kuhar, & Snyder, 1978). If threatened, frightened, or injured, the amygdala (and other limbic nuclei) become exceedingly aroused and secrete massive amounts of enkephalins (Atweh & Kuhar, 1977; Uhl et al., 1978) and corticosteroids (via the hypothalamic- pituitary-adrenals) as well as NE, DA and serotonin. In general, these stress hormones and neurotransmitters initially facilitate motor responsiveness, selective attention, and inhibit feelings of pain (Bliss et al., 1968; White & Neuman, 1980; Rosenblum et al., 2009; Southwick et al., 2013). However, whereas the initial release of NE and DA promote motor reactivity, the continued secretion of corticosteroids and enkephalins are directly associated with the development of immobility, cataplexy, paralysis, and catatonia (Amir, Brown, Amit, & Ornstein, 1981; Fenselow, 1986; Kalin, Shelton, Richman & Davidson, 2011).
Specifically, as fear and stress levels increase, corticostereoid, enkephalin, NE, and DA turnover are increased, especially in the amygdala (Bliss et al., 1968; Krystal, 1990; Tanaka, Kohno, Nakagawa,2002). However, with continued and prolonged stress, NE (Bliss et al., 1968; Spoont, 2007) and (in some individuals) DA levels may be temporarily reduced, whereas corticosteroid and enkephalin secretion may continue unabated.
However, whereas corticosteroids increase NE turnover in the amygdala and other forebrain tissues (Dunn & Berridge, 1987), opiates exert an inhibitory influence on the release of NE (Izquierdo & Graundenz 1980) and stimulate corticostereoid production. Hence, as opiate and cortisol production increases, NE (and DA) levels may begin to drop propitiously due to depletion and inhibition, especially in the amygdala (Bliss et al., 1968; Tanaka et al., 2002) thus producing amygdala-striatal-SMA hyperactivation and a catatonic dissociative reaction.
Moreover, increased and high levels of enkephalins and corticosteroids are associated not only with catatonia, but with numbing, analgesia, a reduction in pain perception (Amir et al.,1981; Auerbach, Fornal, & Jacobs, 1985; Fenselow, 1986; Kalin, Shelton, Richman & Davidson, 2011; Roberts, 2004; Spoont, 2007), and the inhibition of hippocampal and pyramidal neural activity (Packan & Sapolsky, 1990; Uno, Tarara, Else, & Sapolsky, 1989; Roozendall, Koolhaas & Bohus, 2007). Thus, at high levels, corticosteroids and enkephalins may disrupt all aspects of cognition and memory functioning and produce a catatonic paralysis of movement.
DISSOCIATION, FEAR & OUT-OF-BODY EXPERIENCES
As the individual becomes increasingly fearful, and then paralyzed and numb with fear, consciousness and memory functioning may be completely, albeit temporarily eclipsed. Although the amygdala is largely responsible for fear-induced catatonic numbing, the hippocampus may play a more significant role in regard to any subsequent loss of memory and alterations in consciousness. For example, in response to high levels of stress, whereas the amygdala becomes excessively active, the hippocampus displays a reduction in activity and may come to be suppressed (Joseph, 2011b, 1999d; Lupien & McEwen, 2007). Moreover, hippocampal suppression appears to contribute to the immobilization response, including the syndrome of "learned helplessness" (Henke, 2007), a condition that may arise following repeated episodes of inescapable stress. The amygdala and hippocampus appear to interact in this regard, with high levels of amygdala arousal initially acting to excite the hippocampus, even producing hippocampal long term potentiation (Henke, 2007), whereas with continued arousal the hippocampus may become hyperactivated only to eventually come to be suppressed which disrupt memory. However, under certain conditions, recollections from the past may be involuntarily produced and give rise to complex hallucinations, including visions of seeing one's self engaged in certain acts.
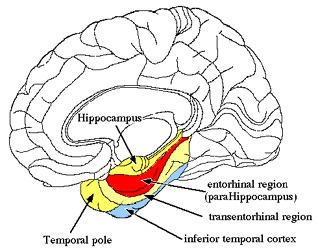

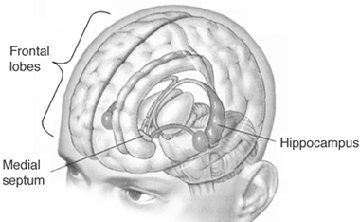
As is well established, the hippocampus is implicated in the storage of long term memories. The hippocampus (and associated structures including the amygdala) also apparently enables an individual to visualize and remember themselves engaged in various acts. That is, the hippocampus and overlying temporal (and parietal) lobe, apparently enable individuals to visualize and remember themselves engage in various activities, as if viewing their behavior and actions from afar. In fact, it has been demonstrated that the hippocampus enables an individual to remember their surroundings, and contains "place neurons" which cognitive map one's position and the location of various objects within the environment (Nadel, 1991; O'Keefe, 1976; Wilson & McNaughton, 2013).
Specifically, O'Keefe, Nadel, and colleagues (Nadel, 1991; O'Keefe, 1976), found that hippocampal pyramidal cells were able to become attuned to specific locations within the environment, as well as particular objects and their location in that environment, thereby creating cognitive maps of visual space. Moreover, as the subject moves about in that environment, entire populations of cells would fire but only when in a particular spot, whereas other cells would fire when in a different location. Moreover, some cells respond not just when moving about, but in reaction to the speed of movement, or when turning in different directions. Moreover, some cells are responsive to the movements of other people in that environment and will fire as that person is observed to move around. (Nadel, 1991; O'Keefe, 1976; Wilson and McNaughton, 2013).
Given these hippocampal attributes, it thus appears that fear-induced alterations or abnormalities in hippocampal-temporal lobe activity presumably triggers the involuntarily visualization of one's surroundings and their personal image similar to what occurs during normal remembering. That is, they experience a dissociated hallucination and see themselves as well as others as if floating above the scene due to abnormal activity in these hippocampal neurons. In fact, hyperactivation of this structure can produce exceedingly vivid hallucinations and dissociative experiences such that the individual may involuntarily see themselves engaged in various acitivities. For example, during the course of electrode stimulation and seizure activity originating in the temporal lobe or hippocampus, patients may report that they have left their bodies and are hovering upon the ceiling staring down at themselves (Daly, 1958; Penfield, 1952; Penfield & Perot 1963; Williams, 1956). That is, their consciousness and sense of personal identity appears to split off from their body, such that they experience themselves as two different people, one passively watching, the other being observed. One female patient claimed that she not only would float above her body, but would sometimes drift outside and even enter into the homes of her neighbors. Likewise, during episodes of severe traumatic stress personal consciousness may be fragmented and patients may dissociate and experience themselves as splitting off and floating away from their body, passively observing all that is occurring.
Penfield and Perot (1963) describe several patients who during a temporal lobe seizure, or neurosurgical temporal lobe stimulation, claimed they split-off from their body and could see themselves down below. One woman stated: "it was though I were two persons, one watching, and the other having this happen to me." According to Penfield (1952), "it was as though the patient were attending a familiar play and was both the actor and audience."
A patient described by Williams (1956) claimed she was lifted up out of her body, and experienced a very pleasant sensation of elation and the feeling that she was "just about to find out knowledge no one else shares, something to do with the link between life and death." Another patient reported that upon leaving her body she not only saw herself down below, but was taken to a special place "of vast proportions, and I felt as if I was in another world" (Williams, 1956). Other patients suffering from temporal lobe seizures or upon direct electrical activation have noted that things suddenly became "crystal clear" or that they had a feeling of clairvoyance, of having the truth revealed to them, of having achieved a sense of greater awareness such that sounds, smells and visual objects seemed to have a greater meaning and sensibility. Similar claims are made by those who have "died" and returned to tell the tale.
Given that under conditions of extreme stress and fear, the amygdala, hippocampus and temporal lobe become exceedingly active, it is thus perhaps not surprising that some victims will experience a "splitting of consciousness" and have the sensation they have left their body and are hovering beside or above themselves, or even that they floated away (Courtois, 1995; Grinker & Spiegel, 1945; Noyes & Kletti, 1977; Summit, 2003; van der Kolk & Fisler 1995). That is, out-of-body dissociative experiences appear to be due to fear induced hippocampa (and amygdala) hyperactivation.
Noyes and Kletti (1977) described several individuals who experienced terror, believed they were about to die, and then suffered an out-of body dissociative experience: "I had a clear image of myself... as though watching it on a television screen." "The next thing I knew I wasn't in the truck anymore; I was looking down from 50 to 100 feet in the air." "I had a sensation of floating. It was almost like stepping out of reality. I seemed to step out of this world."
One individual, after losing control of his Mustang convertible while during over 100 miles per hour on a rain soaked freeway, reported that "time seemed to slow down and then... part of my mind was a few feet outside the car zooming above it and then beside it and behind it and in front of it, looking at and analyzing the respective positions of my spinning Mustang and the cars surrounding me. Simultaneously I was inside trying to steer and control it in accordance with the multiple perspectives I was given by that part of my mind that was outside. It was like my mind split and one consciousness was inside the car, while the other was zooming all around outside and giving me visual feedback that enabled me to avoid hitting anyone or destroying my Mustang."
Individuals reporting "near death" mental phenomenon may also report exceedingly vivid out-of-body "hallucinations" (Eadie 2007; Rawling 1978; Ring 1980). Consider for example, the case of Army Specialist J. C. Bayne of the 196th Light Infantry Brigade. Bayne was "killed" in Chu Lai, Vietnam, in 1966, after being simultaneously machine gunned and struck by a mortar. According to Bayne, when he opened his eyes he was floating in the air, looking down on his burnt and bloody body: "I could see me... it was like looking at a manikin laying there... I was burnt up and there was blood all over the place... I could see the Vietcong. I could see the guy pull my boots off. I could see the rest of them picking up various things... I was like a spectator... It was about four or five in the afternoon when our own troops came. I could hear and see them approaching... I looked dead... they put me in a bag... transferred me to a truck and then to the morgue. And from that point, it was the embalming process. I was on that table and a guy was telling jokes about those USO girls... all I had on was bloody undershorts... he placed my leg out and made a slight incision and stopped... he checked my pulse and heartbeat again and I could see that too... It was about that point I just lost track of what was taking place.... [until much later] when the chaplain was in there saying everything was going to be all right.... I was no longer outside. I was part of it at this point" (reported in Wilson, 1987, pp 113-114; and Sabom, 2002, pp 81-82).
Approximately 37% of patients who are resuscitated report similar out-of-body experiences (Ring, 1980). In fact, these dissociative out-of-body "hallucinations" are detailed in the 1,300 year-old Tibetan Book of the Dead (the Bardo Thodol) and the Egyptian Book of the Dead which was composed over 6,000 years ago. Nevertheless, be it secondary to the fear of dying, or depth electrode stimulation, these experiences all appear to be secondary to excessive amygdala, hippocampal, temporal lobe activity.
SEX, SATAN, AND ALIEN ABDUCTIONS
Compared to other cortical areas, the most complex, vivid hallucinations, including out-of-body dissociative experiences, have their source in the temporal lobe and the hippocampus and amygdala appear to be the responsible agents. Moreover, the amygdala, hippocampus, and temporal lobe play a significant role in the production of REM sleep and dream activity (Hodoba, 1986; Meyer, Ishikawa, Hata, & Karacan, 1987), and become disinhibited in reaction to hallucinogens such as LSD, and are thus implicated in the production of LSD-induced hallucinations (Baldwin, Lewis, & Bach, 1955; Chapman, Walter, & Ross, 1963; Serafetinides, 1965). Hence, under conditions of extreme emotional stress and trauma, the hippocampus, amygdala and temporal lobe may become abnormally active, and victims may experience extreme sensory distortions and hallucinations, including out-of-body phenomenon.
Due to the development of these fear-induced sensory-distorting hallucinatory states, trees, animals, and inanimate objects may even assume demonic likenesses and/or be invested with satanic intent. These horrible hallucinations and sensory distortions may also be committed to memory. Later, the victim may recall the "hallucination" and believe they were set upon by demons, witches and the like, and/or that they were abused in Satanic rituals, or abducted and painfully probed by demonic aliens.
Consider the Walt Disney version of "Snow White." When the woodcutter, who been ordered to cut out her heart, urged Snow White to flee for her life, she panicked and ran into the darkening forest in near hysteria. And, as she ran and stumbled darting in tears here and there, the trees became demonic, growing eyes and wicked mouths, and gnarled arms and hand which stretched out threateningly toward her. Overcome with terror, she collapsed to the forest floor, sobbing uncontrollable.
Now, perchance, had Snow White later recalled this frightening misadventure, she may well have explained to skeptical listeners that demons had emerged from the forest, and threatened to snatch her away. and, she may truly believe this happened, for it is what she truly experienced and what she now truly remembers. Of course, the skeptics, charlatans, foolish psychologists from academic settings who have never examined a patient, and the defenders of pedophiles and those who abuse children, would have us believe that some evil therapist implanted a "false memory;" even though in fact, there is absolutely no credible evidence that complex "false memories" can be implanted in a normal individual in the absence of drugs, hypnosis, or severe duress. Unfortunately, just as there are charlatans conjuring up evidence for "false memories" there are also exceedingly poorly trained psychotherapists, who are likely to mistake the remembered hallucination as a real Satanic event, or an alien abduction, and may then provide treatment for what they believe to be an otherworldly condition, when, at least in the above instance, this is clearly not the case.
It is noteworthy that many of those reporting alien abductions, have a history of sexual molestation or severe emotional trauma, or temporal lobe epilepsy (Mack, 2009). And, as detailed in chapters 28, 29, and/or above, stress, sexual trauma and sexual activity activates the amygdala as well as the temporal lobe--structures which are associated with the production of complex and exceedingly frightening hallucinations, including those of a demonic, religious, and sexual nature including naked women, demons, ghosts and pigs walking upright dressed as people.
Hence perhaps it is not surprising that some children who were sexually abused, sometimes report that they were subject to bizarre sexual rituals that involved demonic (Satanic) activities (which is not to say that in some cases this may in fact be the case). And perhaps it is not surprisingly that those with histories of severe abuse, and/or who suffer from seizure disorders, may believe that demonic-like aliens lifting them into the air and took them to a room of vast proportions--descriptions which are identical to those of at least some patients during temporal lobe stimulation, and at least some individuals who are severely frightened and/or who died only to return to tell the tale.
Of course many come back to life and can remember absolutely nothing that may have occurred after they died. Loss of memory following extreme stress and severe emotional trauma is not at all uncommon.
STRESS-INDUCED MEMORY LOSS AND AMNESIA
During periods of severe stress, initially the hippocampus may become increasingly aroused and activated, even displaying long term potentiation (Henke, 2007). However, as arousal and stress levels increase, hippocampal functioning becomes increasingly abnormal. In consequence, individuals may dissociate, hallucinate, and experience themselves as having left their body. Victims may also suffer varying degrees of memory loss, and in some cases, an amnesia so profound that even personal identity is forgotten (Joseph, 2011b, 1999d). Indeed, is not uncommon for memory to be disrupted under conditions of extreme arousal, fear, terror, and prolonged emotional stress. In one study, 38% of women who had been examined at a hospital following a sexual assault or after being sexually abused, were found later to have no memory of the assault or their early abuse (Williams 2007). Similarly, victims of rapes and physical assaults tend to provide poorer descriptions of their assailants than those who were victims of robberies (Kuehn 1974).
In both animal and human studies it has been repeatedly demonstrated that as arousal levels dramatically increase, memory progressively deteriorates, such that fewer details are recalled, and an inverted-U shaped learning curve is produced (Christianson, 2007; Conrad, Galea, Kuroda, & McEwen, 2005; Diamond et al., 2007, 2009; Easterbrook 1959; Joseph, Forrest, Fiducia, Como, & Siegel, 1981; Kebeck & Lohaus 1986; Kirchbaum, et al. 2005; Kramer, Buckout, & Eugenio 1990; Newcomber, et al. 2009; Yerkes & Dodson, 1908). Moreover, "memory for information associated with negative emotional events, that is, information preceding and succeeding such events...is found to be less accurately retained" (Christianson,2007, p. 193).
Christianson and Nilsson (2004), for example, found that high states of emotional arousal (as indicated by cardiac activity) was associated with reduced recall and poorer recognition memory when asked to remember names, occupations and so on listed below pictures of horribly disfigured vs neutral faces. Moreover, amensia persisted even with cued recall but not with cued recognition. Similarly, Eysenck (1979), in his review of studies examining the relationship between performance and arousal level (as measured by anxiety) indicated that those who were highly aroused showed impaired memory and learning on complex and difficult tasks.
In fact, these same U-shaped relationships between memory and arousal have been demonstrated even in regard to non-threatening emotional events and following the administration of arousal inducing drugs, or the injection of stress-related neurotransmitters and hormones (Diamond et al., 2007, 2009; Kovacs, Telgdy, & Lissak, 1976; Lupien & McEwen, 2007; Warembourg, 1975). For example, epinephrine will improve learning and memory at low and intermediate doses but high doses result in an inverted U shaped learning curve such that memory functioning rapidly deteriorates (Berlyne 1960; McGaugh 1989; Gold 2007).
Even so called "flashbulb" memories are subject to considerable decay and forgetting (Neisser & Harsch, 1987). Specifically, Neisser and Harsh had subjects fill out a questionnaire regarding the Challenger space craft explosion. When questioned 32-34 months later, 75% could not recall filling out the questionnaire and many had forgotten considerable detail regarding the accident. According to Neisser and Harsch (1987), "As far as we can tell, the original memories are just gone."
Moreover, the younger the subject when first exposed to this high impact emotional information, the greater is the degree of verbal forgetting and distortion. For example, in examining memory for national traumatic events such as the death of JFK and Robert Kennedy, Winograd and Killinger (2003) found a steep gradient of forgetting which became more profound for memories formed between the ages of 1-7. Hence, for those adults who had been 1-7 years of age when Kennedy had been killed, only approximately 50% of those who had been 4.5 years old or older, could verbally recall the news and provide at least one detail. Those adults who were younger than 3 had no verbal recollections regarding context or associated events or information sources, and only a few who were younger than 5 were able to demonstrate detailed verbal knowledge or memories when questioned as adults.
In yet another study regarding memory for the Challenger space craft explosion, Warren & Swartwood (1987) found that children age 5 were also less accurate and more likely to delete features over time in describing their recollections. When tested two years later, those who had been the youngest as well as the most emotionally upset at the time were less likely to provide extensive narratives and deleted more features.
Children in general, appear to become more easily overwhelmed by high impact emotional events. For example, Peters (1991) found that children who observed a theft (vs those who did not), and who became the most excited and upset (as measured by pulse rate) experienced a considerable degree of memory loss. That is, as pulse rate went up eyewitness recognition memory declined such that those with the highest pulse rate had the most difficulty correctly identifying the face of the thief. This was particularly evident when the children were later placed face to face with the "thief" (i.e. live line up condition), thus inducing high levels of arousal, such that 58% made incorrect identifications.
Given this susceptibility of children to the adverse effects of trauma, in some instances trauma-induced memory loss may in fact result in a complete dissociative amnesia. Consider, for example, Tara Burke who was kidnapped at age-3, held captive in a van for 10 months, and repeatedly sexually assaulted by two men who filmed her ordeal. Despite a subsequent court trial and intense media scrutiny, 18-year old Tara has no memory of her trauma, and continues to display classic symptoms of dissociative amnesia: "It's like a story that has happened to someone else" (Joseph, 2011b). Likewise, at 18-months of age, "Baby Jessica" McClure was trapped 22 feet deep below the Earth's surface in a narrow hole for 58 terrifying and painful hours. Although this incident was internationally televised and subject to intense media attention, as well as one television movie and several books, and despite the skin grafts and amputation of one toe, 10-year old Jessica cannot remember anything of her ordeal (Babineck, 2007).
FUNCTIONAL AMNESIA
Some victims are unable to forget and suffer flashbacks and intrusive imagery. Others may be unable to remember.
Amnesia and memory loss may be secondary or related to a number of different factors, including post traumatic stress disorders, degenerative disturbances and neurological disease or stroke, disconnection syndromes (such as corpus callosum immaturity) as well as head injury, intense fear, emotional shocks and physical traumas, including rape, assault, sexual molestation or the terror of war in which case even hardened soldiers and aviators may lose their memory (Donaldson & Gardner, 1985; Grinker & Spiegel, 1945; Parson, 2003; Southard, 1919). These disturbances have been referred to as "functional" as well as "dissociative" and "hysterical amnesias." In some instances, loss of memory is only for the event itself and may last from seconds to minutes to hours and even days, weeks, and years.
For example, Donaldson and Gardner (1985) describe a woman who was kidnapped and repeatedly raped over a period of weeks. She developed a dissociative reaction and was able to recall only bits a pieces of what occurred until nightmares and flashbacks and therapeutic assistance enabled her to remember.
Fisher (2002) reports that a druggist who had been terrorized, handcuffed and robbed by two thieves subsequently became amnesic and lost all memories of what occurred. In another case, a woman became amnesic for 18 hours after seeing her husband die right before her eyes (Fisher, 2002).
In some cases, particularly where there are predisposing factors, even personal identity may be forgotten. For example, Schacter, Wang, Tulving, & Freedman (2002) describe a 21 year old patient (PN) suffering from functional retrograde amnesia. He had no idea as to his name, address or any other personal information and had been wondering the streets of Toronto until he approached a police officer and was taken to a hospital. It was only when his picture was placed in a newspaper that a cousin recognized him, although PN claimed not to recognize her. According to his cousin, his grandfather had died the week before. However, PN did not remember the death or the funeral or anything about his grandfather who his cousin said he loved dearly. However, "P.N.'s amnesia cleared on the next evening while he watched an elaborate cremation and funeral sequence in the concluding episode of the television series Shogun. PN reported that as he watched the funeral scene, an image of his grandfather gradually appeared in his mind. He then remembered his grandfather's death as well as the recent funeral. During the next few hours, the large sections of his personal past that had been inaccessible for the previous 4 days returned" (p. 524).
Similarly, Christianson and Nilsson (1989) describe a 23 year old female who was raped and beat while out jogging. When found by a police man she had no memory of her identity or that of her relatives, friends, boyfriend, or place of work and all recollection of her past life, including all aspects of the rape were forgotten. However, after approximately two weeks, when she returned with police to the jogging path so as to discover the scene of the rape (which she still could not recall) she became extremely anxious and upset... and "thereafter, isolated memory-pictures from the traumatic evening started to return in a nonchronological manner, and within a time space of 10-20 minutes she was able to reconstruct the whole episode," (p. 291), including the fact that she was attacked near the crumbled bricks and raped in the nearby meadow.
HIPPOCAMPAL AMNESIA
Stress and arousal-induced memory loss appear to be directly due to disruptions in the functional integrity of the hippocampus and amygdala. Under highly arousing conditions, or following repetitive and prolonged episodes of extreme stress, the hippocampus and amygdala may be injured, and suffer profound dendritic death and atrophy (Diamond et al., 2007, 2009; Lupien & McEwen, 2007; Sapolsky, 2005). These structures may also develop abnormal seizure-like activity (Cain, 2007; Gahwiler 2003; Goelet & Kandel, 1986; Henriksen, Bloom, & McCoy, 1978); all of which significantly impacts learning and memory. Moreover, reductions in the size of the amygdala, and hippocampal dendritic death and atrophy, have been induced by social deprivation and impoverished rearing conditions (Diamond, 1985; Greenough, West, & Devoogd, 1978; Walsh, Budtz-Olsen, Penny, & Cummins, 1969); conditions which are exceedingly stressful and which disrupt learning and memory (Joseph, 1979; Joseph & Gallagher, 1980).
In fact, hippocampal atrophy and memory disturbances have been documented among adults emotionally traumatized in front line combat (Bremner, Randall, Southwick, Krystal, Innis, & Charney, 1995b, Gurvitz, Shenton, Hikama, Ohta, Lasko, & Gilbertson, 2005) and those sexually abused as children (Bremner, Randall, & Scott, 1995a; Stein, Hannah, Koverloa, & McClarty, 1995). Be it animal or human, neglect, abuse, and emotional trauma negatively impacts the hippocampus, the limbic system, as well as learning and memory.
Indeed, it is well established that injury to the hippocampus and overlying temporal lobe is associated with significant memory loss and amnesia (Eichenbaum et al., 2009; Milner, 1970; Reed & Squire, 2007). Moreover, it has been experimentally demonstrated that direct electrical activation of the amygdala or hippocampus can induce a profound amnesia such that minutes, hours, days and even weeks may be erased from memory (Brazier, 1966; Chapman, 1958; Chapman, Markham, Rand, & Crandall, 1967; Halgren, et al., 1991; Jasper & Rasmussen, 1958); memory distrubances which may slowly begin to shrink such that some memory is recovered; i.e. "shrinking retrograde amnesia." In cases of transient global amnesia there is also evidence for temporary mesial temporal inactivation (Hodges & Warlow, 1990), incuding bilateral hypofusion of the hippocamus (Evans et al., 2013), structures which also become inactivated following the seizure- or electrode induced- postictal anterograde amnesia (Brazier, 1966; Halgren, et al., 1991). Similarly, with bilateral hippocampal ablation patients suffer from a profound anterograde as well as retrograde amnesia which may extend backwards several years in time.
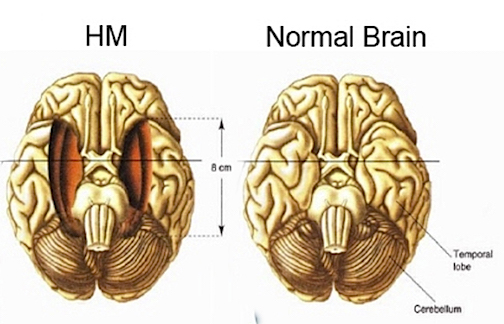
Consider, the famous case of H.M. who can recall almost nothing following bilateral hippocampal amputation (Milner, 1970; Penfield & Milner, 1958; Scoville & Milner, 1957). Dr. Brenda Milner has worked with H.M. for over 25 years and yet she is an utter stranger to him. Every time he discovers his favorite uncle died he suffers the same grief as if he had just been informed for the first time. Nevertheless, H. M. is aware of his deficit and frequently apologizes for his loss of memory. "Right now, I'm wondering" he once said, "Have I done or said anything amiss? You see, at this moment everything looks clear to me, but what happened just before? It's like waking from a dream. I just don't remember...Every day is alone in itself, whatever enjoyment I've had, and whatever sorrow I've had...I just don't remember."
CORTICOSTEROIDS, LTP, MEMORY LOSS, AND HIPPOCAMPAL-AMYGDALA INJURY
Stress-induced hippocampal and amygdala hyperactivation and/or dendritic death and atrophy and significant memory loss is directly due to the stress-induced secretion of enkephalins, norepinephrine, and especially corticosteroids (Diamond et al., 2007, 2009; Kovacs, Telgdy, & Lissak, 1976; Lupien & McEwen, 2007; Sapolsky, 2005; Warembourg, 1975), substances which are released as part of the fight or flight response. As noted, prolonged, or repeated episodes of emotional distress or severe emotional trauma can also alter the secretion of NE, DA, and 5HT (Bliss et al., 1968; Rosenblum et al., 2009; Southwick et al., 2013; Spoont,2007; Witvliet 2007). These neurotransmitter fluctuations negatively impact amygdala and hippocampal neurons, axons, dendrites, and their pre and post synaptic substrates (Cain, 2007; Goelet & Kandel, 1986; Kraemer, 2007; Krystal, 1990).
Specifically under conditions of chronic stress, NE levels may be altered or even depleted (Bliss et. al., 1968; Rosenblum et al., 2009; Southwick et al., 2013; Spoont,2007; Witvliet 2007). Since NE also serves a neural protective function (Glavin, 1985; Ray et al., 1987b), if NE levels are reduced, neurons are exposed to the damaging effects of enkephalins and corticosteroids which at high levels attack and kill amygdala and hippocampal pyramidal neurons (Gahwiler 2003; Henriksen et al., 1978; Packan & Sapolsky, 1990)-cells which normally display synaptic growth and dendritic proliferation in response to new learning (Chapman et al., 1990; Clugnet & LeDoux 1990; Rolls, 1987, 2003).
Under high levels of stress, corticosteroids are secreted in massive amounts, but also directly attack and injure the hippocampus due to the abundance of Type II adrenal steroids receptors which abound within this structure (Lupien & McEwen, 2007; Pugh, Fleshner & Rudy, 2007). In addition, coupled with NE depletion, repeated or prolonged stress induced secretory episodes of corticosteroids and enkephalins can injure cells within the dentate gyrus and Ammon's horn (Lupien & McEwen, 2007) such that the hippocampus will atrophy (Lupien & McEwen, 2007; Sapolsky, 2005; Uno, Tarara, Else, & Sapolsky, 1989).
Even if the hippocampus and amygdala are not injured, memory and new learning may be negatively impacted through stress-induced elimination of long term synaptic potentiation (Diamond et al., 2007, 2009; Dubrovsky, et al 1987; Shors et al., 1989; Spoont, 2007). Long term potentiation (LTP) is believed to represent a prolonged form of synaptic activity which binds together those neurons responsible for transferring information from short-term, to long term memory (Lynch, Larson, Muller, & Granger, 1990) and both the amygdala and the hippocampus develop LTP in response to new learning (Barnes & McNaughton, 1985; Chapman et al., 1990; Clugnet & LeDoux 1990; Diamond et al., 2007, 2009; Enbert & Bonhoeffer, 1999; Lynch et al., 1990). In fact, hippocampal LTP tends to build up within the first 30 minutes after learning and during memory acquisition (Lynch et al., 1990), though once this information has been consolidated and placed in long-term storage, LTP apparently ceases to be a factor in memory maintenance. Moreover, the buildup of LTP is accompanied by morphological dendritic changes and the growth of new spines on postsynaptic dendrites (Engbert & Bonhoeffer, 1999).
During periods of extreme fear or arousal, or in the course of high impact emotional learning situations, hippocampal LTP and theta activity disapear (Redding 1967; Shors et al. 1989; Vernderwolf & Leung 2003) and are replaced by irregular electrophysiological activity (Vernderwolf & Leung 2003). In addition, prolonged stress inhibits the development of LTP in the hippocampus (Shors et al. 1989). For example, if a subject is placed in a situation where they are surrounded by painful physical threat, such as an electrified grid floor, theta disappears (Vanderwolf & Leung 2003). In addition under conditions of stress or when the body has been anesthetized (Green & Arduini 1954), hippocampal theta disapears, as does LTP (Shors et al. 1989). In other words, when repetitively stressed and highly emotionally aroused the hippocampus appears to become so aroused that it is essentially deactivated (following the well established inverse U curve of arousal) and ceases to participate in memory formation.
It has also been demonstrated that the stress induced secretion of corticosteroids interfere with the development of LTP (Diamond et al. 2007; Shores et al., 1989; Spoont, 2007) and can thus directly disrupt learning (Diamond et al., 2007, 2009; Kovacs et al., 1976; Lupien & McEwen, 2007; Warembourg, 1975), memory retrieval, including the ability to discriminate between relevant and irrelevant stimuli (Lupien & McEwen, 2007). Even brief episodes of stress-induced enkephalin and corticosteroid secretion can exert deleterious influences, and can eliminate LTP (Diamond et al. 2007; Shors et al., 1989; Spoont, 2007). In consequence, although immediate retention may remain intact for a few seconds or minutes following severe stress, the buildup of LTP and thus long term memory may be significantly disrupted. In fact, cortisol secretion can also disrupt memory retrieval (de Quevain, Roozendall, & McGaugh, 2011).
In part, the deleterious effects of high levels of corticosteroids on learning, memory, and LTP are due to their suppressive influences exerted on membrane receptor proteins which are detached from their cellular receptor (Beaulieu, 1987). These conditions reduce excitability and information transmission between neurons (Hua & Chen, 1989; Majewska, Harrison, Schwartz, Baker & Paul, 1986) and interfere with messenger RNA protein transcription. Hence, incoming messages cannot be acted on, learning cannot take place, and damaged cells cannot be repaired due to DNA/RNA interference.
Hence, hippocampal memory storage and memory retrieval are significantly disrupted by severe stress and high levels of arousal. However, because the amygdala also play a significant role in emotional memory (Gloor, 2007, 2007; Halgren, 2007; LeDoux, 2005), and as this structure may continue to process affective stimuli even in the absence of hippocampal input (Joseph, 2011b; Seldon et al., 1991), the emotional and traumatic aspects of the experience may thus be memorized. As will be detailed below, the preservation of emotional memory formation likely contributes to flashbacks and the involuntary experience of intrusive imagery.
It is important to emphasize that a single high impact emotional experience is not sufficient to induce complete hippocampal deactivation and the loss of theta and LTP; that is unless the victim is plagued by a predisposition to overreact and/or has been exposed to a previous trauma (see below). That is, victims would not be expected to suffer a complete but only partial memory loss in response to a single traumatic episode. If these stressful episodes are prolonged and repetitive in nature, however, hippocampal theta and LTP may be may be abolished such that hippocampal synaptic depression results (Lynch 1986); a condition that is also related to rapid calcium depletion, for LTP and synaptic plasticity are effected by calcium levels.
OPIATES, CORTISONE & HIPPOCAMPAL AMNESIA
The amygdala interacts with the hippocampus and can depress hippocampal activity during periods of excessive stress. As the amygdala also mediates the stress response, not only does this structure become highly active, but it will secrete corticotropin-releasing factor (CRF) and enkephalins. The amygdala is rich in cells containing enkephalins, and opiate receptors can be found throughout this nucleus (Atweh & Kuhar 1977; Uhl, Kuhar, & Snyder 1978). In response to pain, stress, shock and fear, the amygdala and other limbic nuclei secrete high levels of opiates which can induce a state of calmness as well as analgesia. Under non-painful conditions, the release of opiates can increase feelings of pleasure as well as act as a reinforcer and a reward in response to certain experiences including those which are purely sexual.
However, if opiate activity increases beyond a certain level, they can greatly interfere with memory and learning (McGaugh 1989; Gold 2007), particularly in situations involving pain or terror. This is because opiates and opioid peptides result in hyperactivation of the hippocampus and hippocampal pyramidal cells (Gahwiler 2003; Henriksen, Bloom, McCoy et al. 1978). In consequence, hippocampal epileptiform and seizure activity can develop (albeit in the absence of convulsive seizures) which may be accompanied by abnormal high voltage EEG paroxysmal waves which can last from 15 to 30 minutes (Gahwiler 2003; Henriksen, et al. 1978). In addition, LTP and theta activity is completely abolished as is the ability of the hippocampus to learn and remember (McGaugh 1989; Gold 2007). Hippocampal memory functioning is severely disrupted. Presumably, in consequence, learning and memory is mediated by the amygdala and other nuclei, in the absence of hippocampal input.
In response to continued fear, emotional stress, and anxiety, the amygdala (and other limbic nuclei) also begins to secrete large amounts of the amino peptide, CRF which potentiates the behavioral and autonomic reaction to fear and stress (Davis 2007). Cortisol levels also increase dramatically when the subject is stressed by restraint, and restraint activates the amygdala (Henke, 2007). However, CRF and cortisol has an inhibitory effect on hippocampal functioning and can eliminate hippocampal theta. In consequence, hippocampal participation in memory functioning is again eliminated, altered, or reduced.
In addition to interfering with hippocampal memory formation, the massive secretion of cortisone and enkaphalins may create a "state dependent memory" (see Eich 1987 for related discussion). Under conditions of state dependent learning, the subject is best able to recall what is learned when they are in the same state of mind and under the same environmental and drug induced influences. In consequence, state dependent memories may be recalled and retrieved only under certain "painful" or neurochemical conditions that reproduce the original "state", and/or when an individual is presented with contextual cues or experiencing high levels of emotional stress -which in turn is associated with amygdala activation and opiate and cortisone release; i.e. state dependency. However, when stressed, not just the traumatic memory, but any personal reactions (i.e. fragmentary personality features) may also be recalled due to activation of the associated neural network (Charney et al. 2013; Krystal 1990).
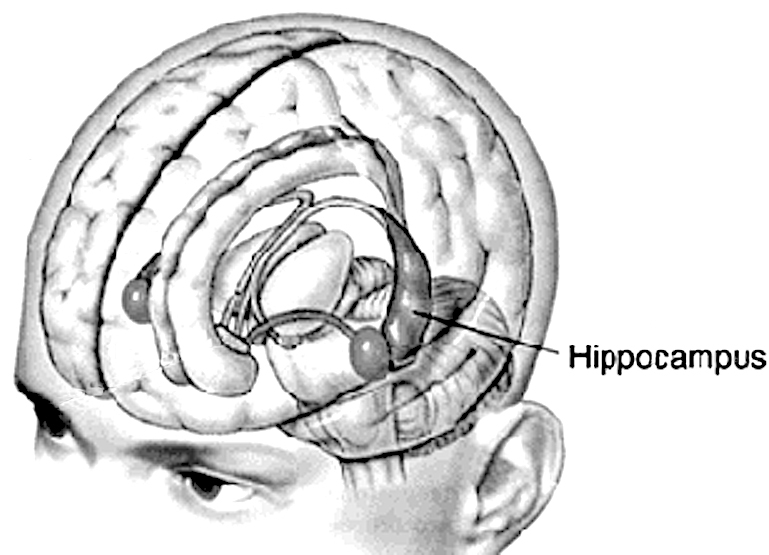
Indeed, it is probably exactly these stress-induced abnormalities which explains the dissociative disturbances of memory, hallucinatory-flashbacks, and post-traumatic stress disorders which afflicts some victims of sexual abuse, front line combat, physical assault and injury), and natural disasters and related trauma.
It is important to note that forgetting can be exceedingly adaptive. That is, the individual (or animal) need not be tormented by repeatedly recalling something that is terrifying. However, when these memories are triggered, many individuals feel they are vividly reexperiencing the original trauma--an experience which is not adaptive.
THE AMYGDALA: EMOTIONAL LEARNING AND HIPPOCAMPAL AMNESIA
It has been repeatedly demonstrated that the amygdala is primary in regard to all aspects of emotional functioning including the capacity to experience fear, terror, sexual arousal, or form complex multimodal emotional memories. The amygdala is responsible for the formation and establishment of sexual-social-emotional memories and in fact contributes to the emotional stream of cognitive and perceptual experience, by associating aversive or rewarding emotions to the flow of overall experience including that which is stored in memory (Gloor 2007, 2007; Halgren 2007; Rolls 2007). However, the amygdala appears to act in conjunction with the hippocampus (and temporal lobe) so that emotional memories are stored alongside associated cognitive attributes (Gloor 2007, 2007; Halgren, 2007; Seldon, et al. 1991). That is, in addition to the specific neural networks created by the hippocampus which insures the consolidation and storage of cognitive experience, the amygdala appears to create a semi-independent neural network which stores emotional information (detailed in chapter 14). Moreover, the amygdala appears to exert a steering influence on the hippocampus so as to reinforce and thus insure that certain memories are formed (see chapters 13, 14).


In fact, even when the hippocampus ceases to participate in memory functioning during periods of repetitive and chronic stress, the amygdala may become potentiated and continue to form emotional memories. However, if the amygdala is excessively activated it may develop kindling and abnormal activity, which in turn may contribute to the development of post-traumatic stress disorder (PTSD)--conditions referred to in the past as "soldier's heart," "shell shock," and so on. PTSD is often characterized by an extremely heightened startle response (Charny et al. 2013) which is mediated by the amygdala (Davis, 2007; Hitchock & Davis, 1991), and patients may experience flashbacks, hallucinations, and intrusive imagery.
Krystal (1990, p. 10) argues that "PTSD could be associated with fundamental and long-lasting neuronal modifications, including alterations in neuronal structure and gene expression. This suggests that many PTSD symptoms may have the indelible qualities of long-term memory," which in turn may result in "long-lasting sensitization of alarm systems with transient habituation, with a subsequent reemergence of symptoms when the habituation fades. From the perspective of evolutionary biology, the resistance of traumatic learning to modification probably reflects the fundamental importance of avoiding catastrophic situations at all costs."
TRAUMA CONGRUENT PERCEPTUAL PROCESSING
Individuals who have been severely traumatized tend to function at a heightened level of limbic (amydaloid) arousal. They may also experience intrusive, recurrent, trauma congruent thoughts and memories, or may demonstrate all the signs of PTSD coupled with loss of memory (Grinker & Spiegel, 1945; Terr, 1990). Those who suffer loss of memory, and those who suffer flashbacks, are also more sensitive to any cues which are in anyway associated with the trauma memory. Hence, when exposed (e.g. sounds from the battlefield) emotional and cognitive processing tends to be severely effected, much more so than when exposed to neutral or even positive material. For example, a woman who suffered amnesia after being brutally raped, began crying when she was brought, by police, to an area with a pile of bricks. Later it was determined that she had been raped in a nearby field where there was also a pile of bricks.
These cues strike at the heart of their trauma complex and concurrently arouse a number of congruent associations that must be attended to, as well as defended against. This increased sensitivity is also a function of the amygdala system (and right hemisphere) being essentially primed to receive and selectively respond to this material, or to selectively inhibit and suppress all associated memories (chapter 29).
For example, Vietnam combat veterans with post traumatic stress disorder (PTSD) are slower to name and show more interference when presented with traumatic words (e.g. bodybags) vs negative ("Germs") vs neutral words than those without PTSD (McNally, Kaspi, Riemann & Zietlin, 1990).
Similarly, rape victims with PTSD are slower than those without to name high threat words (e.g. rape) than moderate threat (e.g. "crime") or postive or neutral words (Cassiday, McNally & Zietlin, 2007; Foa, Feske, Murdock, et al. 1991), indicating that these words evoked negative intrusions on cognitive processing, and/or that some effort was being exerted at preventing recall (see chapter 29). They also showed interference for positive emotional words as well as when viewing colors which suggests that overall cognitive efficiency also appeared to be slowed among those with PTSD (Cassiday, et al. 2007).
Given the right hemispheres role in emotional processing (chapter 9), the inhibitory actions of the right frontal lobe, coupled with the mood congruent data reviewed in chapter 29, showing that when in a negative mood and when the right hemisphere is activated, that positive material and memories are recalled and reported less slowly, this research is not only consistent with that reviewed above, but suggests a role of the right hemisphere, and probably the right amygdala and right hippocampus, in PTSD.
The Amygdala & Traumatic Learning
The amygdala (and right hemisphere) is directly implicated in traumatic learning,. Affective memories formed by the amygdala appear to be rather indelible (LeDoux, 2005). Thus even with hippocampal amnesia, the emotion associated with the trauma may be experienced, and all associated feelings recalled, whereas the place and the circumstances around which the trauma was experienced may be forgotten to varying degree (Seldon, et al. 1991). However, because the amygdala is also involved in learning, and when activated can produce vivid hallucinations and traumatic memories (Gloor, 2007), not only the emotion but aspects of the memory, including flashback might be triggered if presented with certain cues.
Specifically, amygdala synapses and pathways develop LTP and display synaptic plasticity in response to fear stimuli (Chapman, et al. 1990; Clugnet & LeDoux 1990; Goelet & Kandel, 1986; Krystal, 1990). The amygdala not only becomes active when learning emotional stimuli, but patients have been reported to experience a variety of exceedingly vivid memory flashbacks, including even memories of physical abuse and sexual intercourse following depth electrode activation (Gloor 1986, 2007, 2007; see also Penfield & Perot 1963).
As noted, LTP is associated with the development of a long lasting increase in synaptic strength and the growth of additional dendritic spines (Enbert & Bonhoeffer, 1999) such that various interacting neurons come to be interlocked, thus forming widespread neuronal networks which in turn presumably corresponds to the establishment of long (rather than short) term memories. Moreover, it is during the course of the first half hour or so after learning that LTP develops thus presumably interlocking the pre and postsynaptic membrane. Hence, LTP within hippocampal and amygdala neurons and neural pathways may correspond to the development of long-term cognitive and emotional memory and the linkage of neurons which subserve and maintain these memories (see chapter 14).
Hence, fear induced LTP within amygdala neurons is probably a reflection of the amygdala's involvement in most aspects of emotional experience, including the formation of cross-modal emotional associations and memories (Gloor 2007; Halgren 2007; LeDoux 2005; Rolls 2007). However, as noted above, under conditions of repetitive and prolonged trauma, although the hippocampus may cease to participate in memory formation, the amygdala continues to function and will produce LTP. In consequence, painful and distressful memories are formed by the amygdala in the absence of hippocampal participation (Seldon et al., 1991).
Even if the hippocampus destroyed, although new learning and associated cognitive attributes may be forgotten, this amnesia does not extend to fear or pain related stimuli (Seldon et al. 1991). In fact, aversive conditioning is completely unaffected (Seldon, et al. 1991), particularly when the conditions are highly negative, painful or emotional as this form of learning is mediated by the amygdala (Cahill & McGaugh 1990; Hitchcock & Davis 1987; Kesner & DiMattia 1987) as well as the hypothalamus (see chapter 13).
However, under conditions of excessive stress resulting in hippocampal amnesia, the victim may remain amnesic such that all associated memories are repressed unless activated by relevant cues. On the other hand, due to the involvement of these structures in dreaming, these troubling memories may be reproduced in the form of fragmented and terrifying dreams (Donaldson & Gardner 1985; Kramer, Schoen & Kinney 1987), that in turn may be forgotten (Kaminer & Lavie 1991; Kramer et al. 1987); that is, forgotten by the left cerebral hemisphere upon waking (Joseph 2003a).
On the other hand, trauma related memories may be recalled when confronted by related stressors, and contextual or emotional cues (Charney et al. 2013). Likewise, flashbacks, and exceedingly vivid affective memories may be reactivated and recalled if the amygdala is aroused by similar contextual cues or if it is directly activated (Gloor 2007, 1999, Halgren 2007; Seldon, et al. 1991), including even memories and fragmentary flashbacks of sexual intercourse or instances of physical abuse and terror (Gloor 2007; see also Penfield & Perot 1963).
Likewise, combat vets with PTSD may have flashbacks involving cries for help, battle scenes, dead and wounded bodies, and so on (Mueser & Butler, 1987; Wilcox, et al. 1991). Presumably those with PTSD continue to experience intrusive images and flashbacks because those amygdaloid neural-memory pathways associated with the original trauma remain primed or in a partially activated state, such that, if activated, aspects of the original trauma-memories are recalled.
In fact, it has been demonstrated that adults with PTSD when asked to recall trauma-memories or when presented with combat-related photographs, display increased blood flow activity in the amygdala (Rauch et al., 2005; Shin et al., 2005). And the amygdala also becomes activated by bizarre stimuli (Halgren, 2007). Hence, the amygdala is directly implicated in the storage and recall of trauma-imagery.
SEROTONIN, FLASHBACKS & INTRUSIVE IMAGERY
The amygdala also becomes particularly active when recalling emotional memories (Halgren 2007; Heath 1964; Penfield & Perot 1963), and in response to cognitive and context determined stimuli regardless of their specific emotional qualities (Halgren 2007). Electrical stimulation of the amygdala (and/or the overlying temporal neocortex) can trigger the recollection of recent and forgotten memories, including incidents involving sexual intercourse, fear, terror, or conversely, joy and happiness (Halgren 2007; Gloor 2007; Penfield & Perot 1963). In addition, associated emotional, visual, or contextual cues can trigger amygdala activation and the recall of these memories (Rolls 2007; Halgren 2007). However, because associated stimuli can trigger their recollection, in some instances, the recollection of these memories are not only involuntary, but intrusive and may take the form of flashbacks.
These flashbacks may also have a dream-like hallucinatory quality, and only fragments of the original memory may be produced--as is common during dreaming. In fact, the amygdala and hippocampus, and overlying temporal lobe play a significant role in the production of REM sleep and dream activity (Hodoba, 1986; Meyer, Ishikawa, Hata, & Karacan, 1987). These structures also become disinhibited in reaction to hallucinogens such as LSD, and are directly implicated in the production of LSD-induced hallucinations (Baldwin et al., 1955; Chapman et al., 1963; Serafetinides, 1965). Moreover, LSD-hallucinations, may also be later reexperienced as flashbacks. In fact, be they trauma induced or a product of LSD synaptic changes, hallucinations, flashbacks and intrusive imagery, appear to be secondary to similar structural and neurochemical changes in the amygdala and hippocampus--structures directly implicated in the production of complex and exceedingly vivid and emotionally disturbing hallucinations (Gloor, 1990, 2007; Horowitz et al., 1968; Halgren, 2007; Halgren et al., 1978).
Specifically, LSD-induced hallucinations are produced secondary to alterations in serotonin (as well as NE) secretion and uptake within the amygdala, hippocampus and overlying temporal lobe. Likewise, prolonged, or repeated episodes of emotional distress or severe emotional trauma can alter the secretion of serotonin (5HT) and NE (Bliss et al., 1968; Rosenblum et al., 2009; Southwick, Krystal et al., 2013; Spoont,2007; Witvliet 2007). However, 5HT and NE fluctuations not only negatively impact amygdala and hippocampal neurons, axons, dendrites, and their pre and post synaptic substrates (Cain, 2007; Goelet & Kandel, 1986; Kraemer, 2007; Krystal, 1990), but can produce profoundly disturbing, exceedingly vivid hallucinations.
For example, as 5HT exerts an inhibitory influence and induces perceptual filtering so as to narrow the range of sensory input received, the stress-induced depletion of 5HT can induce sensory distortions and dissociative phenomenon including hallucinations similar to those produced by the administration of LSD (Spoont, 2007). LSD acts to inhibit 5-HT production and blocks synaptic uptake within the amygdala, hippocampus, and temporal lobe (chapter 13) which induces hallucinations.
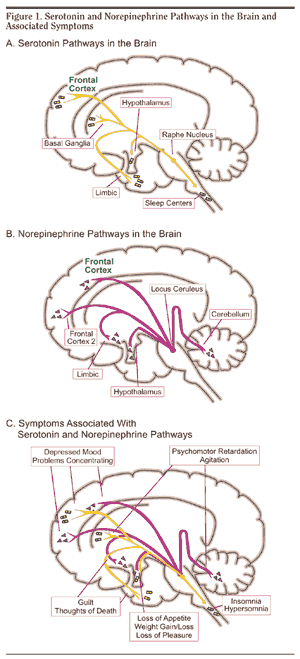
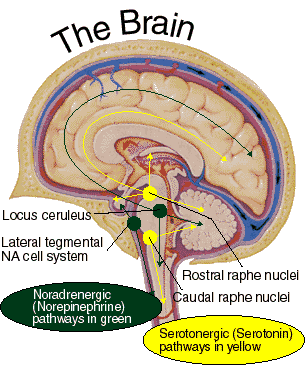
Hence, with 5HT reductions, the traumatized victim may be so overwhelmed by sensory overload that they may hallucinate--sensory distortions which may be forgotten or committed to memory. However, because of 5HT-induced neuroplastic changes and the development of stress-induced kindling and excessive activity in these structures, associated neural networks and the memories they maintain, may be easily activated and the memories recalled, albeit in a dream-like hallucinatory fashion.
Specifically, the main source for limbic and neocortical 5-HT is the dorsal (DR) and median raphe (MR) nuclei (Azamita, 1978; Azamitia & Gannon, 1986; Wilson & Milliver, 1991ab). The amygdala, hypothalamus, basal ganglia, primary and association receiving areas, and the frontal lobe are innervated by the DR, whereas the hippocampus, cingulate gyrus, and septum receive their 5-HT from the MR nuclei (Azamita, 1978; Azamitia & Gannon, 1986; Wilson & Milliver, 1991ab). The MR, however is diffusely organized and appears to exert a non-specific and global influence on arousal and excitabilty (Wilson & Molliver, 1991ab). The DR is much more discretely organized and can exert highly selective inhibitory or excitatory influences and plays a role in the coordination of excitation in multiple functionally related areas including the frontal lobes and amygdala (Wilson & Molliver, 1991ab). Because of the manner in which they are organized, the DR and MR can exert select inhibitory influences so as to promote perceptual filertering in one or a variety of neocortical and subcortical areas while simultaneously exciting yet other multiple regions which in turn aid selective attention and in the creation of neuronal networks.
Thus, one of the major roles of serotonin (5-HT) is inhibition of neural activity (Applegate, 1980; Soubrie, 1986; Spoont, 2007). It restricts perceptual and information processing and in fact increases the threshold for neural responses to occur at both the neocortical and limbic level. For example, in response to arousing stimuli, 5-HT is released (Auerbach, Fornal & Jacobs, 1985; Roberts, 2004; Spoont, 2007) which acts to aid attentional and perceptual functioning so that stimuli which are the most salient are attended to. That is, 5HT appears to be involved in learning not to respond to stimuli that are irrelevant and are not rewarded (See Beninger, 1989) which are in turn filtered out and inhibited.
In response to highly arousing and stressful, fearful, or painful stimuli, 5-HT (as well as opiate) levels, at least initially are increased which results in numbing, analgesia, and a loss of pain perception (Auerbach et al. 1985; Roberts, 2004; Spoont, 2007). However, under conditions of prolonged stress and fear, 5-HT is rapidly depleted. In consequence, the individual may begin to feel overwhelmed and unable to appropriately process incoming sensory stimuli. Soon thereafter they may also demonstrate a heightened fear and startle response (Davis, 2004) as well as social withdrawal (Raleigh et al. 2003); disturbances directly associated with the amygdala (see chapter 13). Moreover, because sensory filtering is reduced, neurons in the amygdala, hippocampus, and inferior temporal lobe cease to be inhibited, which in turn can result in depersonalization (Sallanon et al. 2003) and increased social fear, as well as dream-like states and hallucinations (Sallanon, et al, 2003; Spoont, 2007), including flashbacks. Trauma-memories and associated emotional reactions are reexperienced.
KINDLING, THE AMYGDALA, PTSD, & FLACHBACKS
Opiate receptors appear to be located presynaptically on norepinephrine (NE) cells (Llorens, et al1978), and opiate peptides appear to exert an inhibitory effects on the release of NE (Izquierdo & Graundenz 1980). Stress increases NE turnover in the amygdala (Tanaka, et al. 2002) and increases the production of opiates (Krystal 1990). As noted, the repeated release of these substances, including cortisol and 5HT, coupled with fear induced LTP and synaptic plasticity can in turn significantly and permanently alter the functioning of the amgydala.
When the amygdala is repeatedly and continually stimulated by excitatory neurotransmitters, or via pharmacological agents including opiates, or through direct electrical activation, an abnormal form of neuronal plasticity and a lowered threshold of responding results. These changes are associated with increases in the size of the evoked potential amplitudes and can give rise to epilptiform after discharges, seizures and convulsions, as well as induce kindling (subseizure activity) within the amygdala (Cain 2007; Racine 1978).
Normally, the release of NE retards kindling in the amygdala. However, with repetitive stress and high levels of arousal, opiates will inhibit NE release. This can lead to permanent structural and functional alterations within amygdala neurons, effecting their postsynaptic densities and in the size of the presynaptic terminals as well as their capacity to process and transmit information (Cain 2007; Racine 1978). The amygdala may in fact develop abnormal levels of activity which may take the form of kindling. In consequence, the threshold for amygdala activation and information transmission and recollection may be lowered.
Moreover, even when other brain areas are repeatedly and highly activated, kindling begins to occur within the amygdala, and in fact builds up more rapidly than in any other brain region (Cain, 2007; Racine, 1978). In contrast, the hippocampus is one of the slowest regions to develop kindling, with the anterior hippocampus being more likely to do so than the posterior regions (Cain, 2007; Racine, 1978).
Kindling in the amygdala can potentiate neuronal transmission and can induce neural plasticity, which in turn significantly effects learning and memory and the manner in which kindled circuits transmit non-seizure relative activity and information (Cain, 2007; Racine, 1978). That is, high activity induced kindling can effect the actual creation and functioning of neural circuits as well as their manner of transmitting and processing information, particularly in that it also enhances inhibitory neurotransmission (Cain, 2007). For example, it has been found that kindling induces increases in postsynaptic densities and in the size of the presynaptic terminals (Cain, 2007).
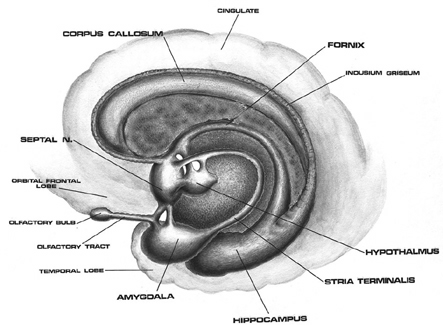
Because of the development of amydaloid kindling and excessive activity, this structure and associated memories and emotional reaction might be easily activated, thus resulting in a flashback. For example, in hearing a car backfire in the middle of the night, a traumatized former vet might experience a flashback, spring from their bed, search for their gun, and/or crawl about the room in search of, or in fear of enemies (Charney et al. 2013; Krystal 1990). In addition to the flashbacks and the original feelings of fear, enkephalins and related substances might also be released in response to the hallucinations and reexperience of fear. That is, once the trauma is experienced and an associated neural network is created, associated feelings and/or the experience of the trauma itself may be later reactivated by associated cues, and not only might the entire circuit of experience be recalled but the body and brain will respond the same as when originally traumatized.
Unfortunately, even benign cues may trigger hyperarousal, traumatic feelings, and even defensive postures in situations unrelated to the original stressful event -even in the absence of obvious stressors or provoking situations (Charney et al. 2013; Krystal, 1990). For example, because an excessively activated amygdala is easily activated (such as due to kindling) the invidual may display a heightened startle reaction, and it may periodically trigger the recollection of associated and trauma-related memories due simply to the waxing and waning of this activity or due to benign stressors including even the abnormal secretion of corticosteroids (see below). For example, an overly stressed and hyper-trophied HPA axis may forever hypersecrete corticosteroids which in turn can activate and induce kindling in the amygdala.
Specifically, reductions in 5HT can increase amygdala activation and is associated with the increased secretion of opiates, cortisone, and dopamine. The possibility of amygdala kindling would be enhanced, as well as the development of stress induced LTP and synaptic plasticity. Amygdaloid kindling and reduced 5-HT levels would in turn increase the likelihood of an enhanced startle reaction, social withdrawal, feelings of depersonalization, hallucinatory states, sleep disturbances, increased REM latency, and other associated symptoms associated with a chronic post traumatic stress disorder.
Increased startle, disturbances in sleep onset, increased REM latency and decreased REM, hypersensitivity to noises, hypnogogic and auditory hallucinations, intrusive images and emotions, depersonalization and dissociation, nightmares, as well as difficulty sleeping, are stereotypical features associated with PTSD. These symptoms have been repeatedly noted among combat veterans, concentration camp and holocaust survivors, victims of sea disasters and natural catastrophes, among individual who are physically assaulted, raped or sexually abused as children, including adults who witness the murder of children, relatives, friends, parents, husbands or wives (Charney et al. 2013; Grinker & Spiegel, 1945; Kaminer & Lavie, 1991; Mueser & Butler, 1987; Parson, 19; van Kammen et al. 1990; Van Putten & Emory, 1973; Wilcox et al. 1991). Indeed, the development of these disturbances appears to be a universal reaction to traumatic experiences. They also appear to be directly related to permanent neurochemical (i.e. 5-HT, NE, DA, and opiates) and neurological alterations involving the amygdala and hippocampus.
In fact, even a single trauma may result in alterations in NE and 5HT neurtransmitter turnover and release, which in turn can influence and increase the size of both the pre and postsynpatic substrates (Goelet & Kandel, 1986; Krystal, 1990) and even gene expression. This may induce alterations in learning and memory, and increase emotional reactivity and alarm responses which may persist indefinitely (Charney et al. 2013; Goelet & Kandel, 1986).
SEROTONIN, CORTISOL, DEPRESSION, APATHY AND STRESS
When 5-HT levels are reduced individuals may become depressed. There is also an increased tendency to respond to non-rewarding situations (Sourbrie, 1986) and to continue to respond regardless of punishment (Spoont, 2007). Effected individuals may fail to avoid or may even be drawn to abusive or potentially frightening or traumatic situations, and may seem helpless to alter their behavior, e.g. learned helplessness. Reduced 5-HT has in fact been repeatedly noted in the brains of those who have committed suicide (Brown et al. 2002; Cronwell & Henderson 1995) and among primates who have been repeatedly stressed and who are of very low status. In fact, young primates subjective to severe social stress may display reductions in 5HT levels for up to five years. Conversely, the anti-depressant drug Prozac, acts to increase 5HT levels.
Likewise, individuals with high cortisol levels tend to be passive, become easily depressed, exhibit an "uptight" rigidity in temparment (Johnson et al., 2005; Kagan, Reznick, & Snidman, 2003), and are easily overwhelmed by even minimal stress. Primates and human children with high basal levels of cortisol are less likely to react in an adaptive manner when stressed and are the most likely to freeze-up and passively surrender to their fate (Johnson et al., 2005; Kagan et al., 2003). If sufficiently stressed, they may also die (Johnson et al., 2005; Uno et al. 1989). As noted, cortisol can decrease the production of lymphocytes by the lymph nodes and thymus gland. Since a primary role of lymphocytes is immunological (e.g. to destroy invading bacteria), excessive cortical secretion can disrupt the immune response thus increasing the likelihood that one may get sick and die.
Because the amygdala and HPA axis are abnormally activated, coupled with kindling and the hypersecretion of cortisol and altered levels of 5HT, the individual may continually respond as if stressed even when in a benign environment, and may appear to be chronically depressed, sickly, and easily overwhelmed. It is thus noteworthy that blockage of 5-HT reuptake (which increases the amount of 5-HT in the synapse) acts to reduce the symptoms of post-traumatic stress disorder (Hollander et al. 1990) as well as depression (e.g. Prozac). However, in the absence of pharmacological intervention, the reductions in 5HT and increases in cortisol, coupled with the potentiation of associated trauma-related neural networks may continue to exert significant disruptive influences as if the individual were constantly subject to trauma; and this may include the recollection of trauma-related memories and flashbacks. Even when no longer stressed, the brain continues to respond as if exceedingly emotionally aroused, although they are otherwise severely depressed.
Moreover, because the brain may also hypersecrete enkephalins when in stressful situations, some victims may become addicted to trauma (see Charney et al. 2013; Krystal 1990). They may seek traumatic or highly arousing or dangerous experiences so as to induce the release of naturally occurring opiates. They become addicted to trauma, addicted to drugs, or addicted to unpleasant interpersonal relationships (Charney et al. 2013; Krystal 1990). Iin fact, individuals who have been repeatedly traumatized and those with PTSD, not uncommonly abuse opiates and other substances.
KINDLING & PSYCHOSIS
Amygdala kindling can also have a direct effect on personality including increasing social withdrawl and defensiveness (Cain, 2007) as well as increasing sensitivity to dopamine. It is noteworthy that the amygdala receives extensive dopamine projections and has a higher concentration of dopamine than the caudate nucleus (Stevens, 2007). These amygdala dopamine neurons project to wide areas of the neocortex, including the frontal lobes and are believed to play an inhibitory role on neocortical information processing, which in turn is important in selective attention.
In this regard, under conditions of continual high level emotional stress and arousal, opiates may be released, amygdala neural circuitry can be altered, the hippocampus may be damaged, NE production can be suppressed, and dopamine sensitivity and dopamine levels can be increased, particularly within the amygdala (see Krystal, 1990). Hence, not only might emotional responsivity be altered but wide areas of the neocortex can be abnormally effected due to alterations in amygdaloid neurochemical and dopamine functioning.
Altered dopamine sensitivity and transmission has long been associated with the development of schizophrenia and psychosis. Specifically, individuals with schizophrenia contain a higher density of dopamine receptors within the left half of their brain (Stevens, 2007), as does the normal left amygdala (Bradbury, et al. 1985). In fact, the left amygdala and left temporal lobe (Flor-Henry, 1969; Perez, et al. 1985; Stevens, 2007) have long been thought to be a major component in the pathophysiology of psychosis and schizophrenia (Heath, 1954; Stevens, 1973; Torey & Peterson, 1974). For example, abnormal activity as well as size decrements have been noted in the left amygdala (Flor-Henry, 1969; Perez, et al. 1985) as well as the left inferior temporal lobe in schizophrenic patients (see Stevens, 2007). Spiking has also been observed in the amygdala of psychotic individuals who are experiencing emotional and psychological stress (see Halgren, 2007). Thus there appears to be an abnormal concentration of dopamine in the left hemisphere and temporal lobe among psychotic individuals, which, in some cases may be secondary to repeated traumas experienced during childhood.
Given that stress increases amygdaloid activity, induces kindling, and increases the production of opiates and dopamine, this may explain why psychotic disturbances can be exacerbated when patients are subject to emotionally arousing conditions. Moreover, given that stress and emotional arousal also alters serotonin activity, and given the role of this transmitter in sensory filtering, emotional inhibition, dream production and hallucinatory states, the likelihood of severe psychological abnormalities is tremendously increased, particularly in those who may be predisposed to develop these disturbances due to adverse early environmental and childhood experiences.
STRESS-INDUCED PSYCHOSIS & PERSONALITY MULTIPLICITY
Dramatic alterations in personality following injury to the amygdala (and overlying temporal lobe) are not uncommon (Lilly et al., 2003; Marlowe et al., 1975; Shenk & Bear, 1981; Terzian & Ore, 1955). Indeed, damage to these structures, the amygdala in particular, can in fact abolish all aspects of a social personal identity (Lilly, Cummings, Benson, & Frankel, 2003; Marlowe, Mancall, Thomas, 1975; Terzian & Ore, 1955), and erase from the mind memories of the personal, social, and cognitive self. In cases of amygdala injury or excessive or epileptic activity, the patient may become aggressive and violent, or they may become withdrawn, lose all interest in social activities, and experience significant alterations in sexual drive. Some become impotent whereas others experience an increase in sexual feelings, and then act on it in an inappropriate and indiscriminate manner. They may masturbate and expose themselves in public, seek sex with family members, or experience alterations in sexual orientation and engage in intercourse with members of their own sex or even animals (chapter 28; Mesulam, 1981; Shenk & Bear, 1981).
In some cases of temporal lobe, amygdala, hippocampal abnormality, the alterations in personality are so dramatic patients may appear to be suffering from a multiple personality disorder (Fichtner, Kuhlman, & Hughes, 1990; Mesulam, 1981; Shenk & Bear, 1981). In fact, in several cases of multiple personality dissociative disorder, EEG or blood flow abnormalities involving the temporal lobe have been demonstrated (Drake 1986; Fichtner et al., 1990; Mathew, Jack & West, 1985; Mesulam 1981; Schenk & Bear 1981). Moreover, some patients may shift from one personality to another following a seizure or with increases in temporal lobe activity (Mesulam, 1981; Shenk & Bear, 1981). Similarly, it has been reported that heightened emotional distress may precede the appearance of alternate personalities (Greaves 1980; Putnam 1985); and as noted, stress can also abnormally active the amygdala and increase blood flow to the temporal lobe.
As per the development of additional personalities, as detailed in chapter 2, almost all humans are capable of displaying different personalities depending on context and situational restraints, e.g. first date, at work, etc., And almost all humans are capable of shifting between and extinguishing these alternative personalities at will. However, children are also capable of generating make-believe personalities, such as imaginary friends which they might confide in or blame for their own misdeed. In this regard, children may be more susceptible to developing psychotic disturbances involving the creation of multiple personalities. In fact alternative personalities are generally formed during childhood and during periods of extreme stress involving sexual abuse and torture (Bliss 1980; Coons, Bowman & Milstein 2003; Coons & Milstein 1986; Devinksy, Putnam, Grafman, Bromfield & Theodore 1989; Kluft 2004; Mathew, Jack, & West 1985; Putnam 1985; Putnam, Guroff Silberman, et al. 1986; Rosenbaum & Weaver 1980; Schreiber 1974; Wilbur 2004).
Specifically, it appears that these alternate personalities may be formed during a dissociative state during a period of severe and repetitive trauma. That is, under certain traumatic (or neurological) conditions, an aspect of the consciousness may fragment, break off, and thereafter variable act in a semi-independent or completely independent manner. This form of dissociation may be likened to a disconnection syndrome and would be due to abnormal activation of the hippocampus, temporal lobe, amygdala, which induces an out-of-body hallucination as well as a loss of will (due to medial frontal involvement).
As detailed above, it has been repeatedly demonstrated with neurosurgical and epilepsy patients that abnormal temporal lobe/hippocampal activation can cause an individual to experience themselves as separating and floating above their body, such that they may feel as if they are two different people, one watching, the other being observed (Daly 1958; Jackson & Stewart, 1899; Penfield 1952; Penfield & Perot 1963; Williams 1956). As described by Penfield (1952) during electrical stimulation of this area, "it was as though the patient were attending a familiar play and was both the actor and audience." Likewise, children, as well as adults, who are terribly abused or traumatically stressed and frightened often report dissociative experiences including even a splitting of consciousness such that one aspect of their mind will seem to be floating above or beside their body (Courtois, 2003; Grinker & Spiegel, 1945; Noyes & Kletti, 1977; Parson, 2003; Southard, 1919; Terr, 1990). Hence, the famous aside: "He was literally beside himself with fear."
Most abusers do not wish to molest an unresponsive body, but a personality that is either excited or tormented and terrified by what is occurring. According to Courtois (1995), it is the abusers desire "to achieve total control over the victim and her responses and for the victim to become a willing participant and to enjoy the abuse. To achieve this end, many incest offenders take great pains to sexually stimulate their victims to arousal." They may pinch, cut, and hurt them to obtain the desired reaction. Thus, although these particular children may have "split off" from their body, they are nevertheless forced to respond, as a personality, to the abuse in which case the "personality" may act in accordance with the manner in which it was created. Thus, the abuser slowly shapes a personality which is formed during period of abuse. Hence, some alternative personalities are highly sexual, or homosexual, or angry and self-destructive, mirroring either the role they were forced to play or the personality and behavior of the perpetrator. The dissociated aspects of conscious-awareness, therefore, may slowly establish its own independent identity; that is, an alternate personality--a personality which may be repeatedly triggered or purposefully shaped during subsequent traumas, and which may grow only to split apart again and yet again, forming multiple fragmentary personas each supported by a dissociated and abnormal neural network.
However, these "broken off" or alternate personalities, in turn, may be "state dependent" and supported by isolated neural networks maintained by the amygdala in the absence of hippocampal participation. Because the alternate "personality" essentially splits off from the main personality under certain highly stressful, in general, it can only be reactivated and function independently when the individual is stressed or in a similar state of mind. Moreover, because of the state dependent nature of this partial "personality," the main personality essentially becomes amnesic regarding it. In part this is a consequence of hippocampal deactivation. When the hippocampus returns to normal, it can no longer gain access to these personal memories (which may be maintained by the amygdala), such that dissociated personal memory and the associated, albeit disconnected personality, remains isolated and inaccessible.
INDIVIDUAL DIFFERENCES AND PREDISPOSING FACTORS
The manner in which one reacts to extreme stress or prolonged emotional trauma varies depending on age, gender, situational, individual, and neurochemical differences (Bremner et al., 2013; Johnson, Kamilaris, Carter, Calogero, Gold & Chrousos, 2005; Kalin et al., 2011). Not all individuals respond the same when stressed. When traumatized some individuals are unable to forget and continually think about the trauma and experience flashbacks and intrusive imagery. Some animals and humans respond with only minimal degrees of arousal or fluctuations in learning and memory, whereas others become easily overwhelmed and/or display profound memory loss or reductions in learning with even minimal stress (Como, Joseph, Fiducia, & Siegel, 1979; Johnson et al., 2005; Joseph, 2011b, 1999d; Joseph et al., 1981; Joseph, Hess, & Birecree, 2003; Joseph & Gallagher, 1980; Kalin et al., 2011).
For example, females may be more at risk than males for becoming highly stressed (Compas et al., 1989; Coons 2003; Cleary, 1987; Larson, & Ham,2013). Females are more easily frightened (Campbell, 1999; Cantor & Reilly, 2002; Sparks, 1986; Tamborini & Stiff, 1987), and cry easier than males, (Lombardo, Crester, Lombardo & Mathis, 2003; Oliver, 2013; Shields, 1987; Thomas, 2013). Females will begin crying and becoming upset even under conditions that seem affectively neutral (Oliver, Sargent & Weaver, 2011) and those which should engender joy, such as weddings. They are also two to three times more likely than men to develop mood disorders such as depression (Nolen-Hoeksema, 1990) and stress related abnormalities such as anxiety and phobias (DSM-IV), and appear to become more depressed than males (Cleary, 1987; Wichstrom, 1999). According to Cleary (1987, pp. 41, 49), "virtually all surveys find that women report more symptoms of distress than men," and that "women are more likely than men to experience symptoms of depression and anxiety."
Moreover, the younger the victim and the more profound or repetitive the trauma, the more likely may be the subsequent loss of memory (Joseph, 2011b, 1999d; Terr, 2009) and the more likely is it that they may develop post traumatic emotional and personality disturbances (DSM IV, 2009). The most severe forms of dissociative disorder are often directly linked to childhood trauma (DSM, 2009).
Children may be more at risk, simply because they are children and more vulnerable to repetitive instances of abuse and torture. For example, it has been estimated that of those who were sexually molested, approximately 29-31% were physically or aggressively forced and were pinned or held down, whereas from 2% to 5% were subject to substantial violence such as hitting or slapping or beating (e.g. Courtois, 2003; Russell, 1986). The amygdala becomes especially active when the body is restrained, as well as during sex and in reaction to pain. Penile erections, ovulation, uterine contraction and lactogenetic responses, and vaginal-vulva secretions in conjunction with orgasm and sexual feelings in the genital areas are all associated with heightened activity in the amygdala. (Halgren, 2007; Jacome, et al. 1980; Gloor, 1986; Remillard, et al. 2003; Robinson & Mishkin, 1968; Shealy & Peele, 1957). However, because of the susceptibility of the amygdala to develop abnormal kindling activity in reaction to severe stress, early abuse may more severely injure the amygdala due to the repetitive and vicious nature of the abuse.
In addition, children often respond to sexual abuse by pretending that it is not taking place (Courtois, 2003; Summit, 2003), such as by "playing possum." Many victims will lie in bed in a state of frozen fear and hypervigilance (Courtois, 2003)--which is associated with excessive amygdala activity. It is also not unusual for children to initially hyperventilate and then to hold their breath and pretend they are sleeping while the abuse occurs. Not only might they may be denied oxygen due to purposeful breath holding, but some may be denied sufficient oxygen by having an adult lie across them. In some cases, sexually abused children suffer compression injuries to the chest and will die from lack of oxygen. Moreover, some sadistically abusive adults will hold their hands over the child's mouth as a form of torture or so as to prevent them from crying out while they are being assaulted.
The amygdala, hippocampus, temporal lobe neural complex are highly susceptible to hypoxia, which in turn can result in loss of memory, or conversely create hallucinatory and dissociative states as well as injury. This is particularly true during childhood as the brain is still immature and plastic and consumes almost 50% of total body oxygen and glucose, compared with 25% in adults. Hence, whereas the amygdala may be highly activated due to sexual activity coupled with feelings of fear and terror, in some cases it (and the hippocampus) may be simultaneously denied sufficient oxygen which it requires even more than is usual (because it is highly active). In consequence, with repeated instances of abuse, coupled with oxygen depletion, the still immature temporal lobes and amygdala/hippocampal complex might be injured by these experiences and forever function abnormally.
In some cases, memory loss and/or the reaction to stress and the development of PTSD may be influenced by previous traumatic experiences. Bremner and colleagues (2013), for example, have shown that at least 26% of Viet Nam veterans with PTSD admit to a history of childhood abuse whereas only 7% of those without PTSD had a similar history. In another case described above, a 23-year old female who had been sexually abused s a child, lost all memory of her identity, her past life, or that of her relatives, boyfriend, or place of work after she had been accosted while jogging and brutally raped (Christianson & Nilsson, 2004).
In addition to previous emotional trauma, some individuals are predisposed due to underlying neurological disorders. In a case of profound dissociative amnesia described above, a 21-year-old man who had forgotten his name and all other personal information following the shocking death of his beloved grandfather, was subsequently discovered to have a right temporal lobe injury suggestive of gliosis (Schacter et al., 2002). Gliosis, particularly those involving the temporal lobe are highly likely to give rise to abnormal activity as well as seizure disorders which in turn will hyper-activate the amygdala (Bear, 1979; Cain, 2007; Gloor, 2007).
Moreover, in addition to previous traumas or neurological predispositions, there are individual differences in regard to basal levels of DA, NE, corticostereoids, etc., and the manner in which these neurotransmitters fluctuate when stressed. For example, individual (non-stressed) primates who have naturally elevated cortisol levels are observed to be passive, low status, submissive, and to exhibit an "uptight" rigidity in temperment (Johnson et al., 2005). Primates with elevated cortisol have difficulty coping with even mild stressors, and tend to "waste away and die with no apparent pathological cause" (Johnson et al., 2005, p. 333). Likewise, inhibited, passive, fearful, and submissive children tend to have higher levels of cortisol even under non-stressed conditions (Kagan, Reznick, & Snidman, 2003). As these individual primates and children are already functioning as if highly stressed (as suggested by their elevated cortisol levels) they also have the most difficulty functioning in an adaptive manner when stressed, and are the most likely to freeze-up and passively surrender to their fate (Johnson et al., 2005; Kagan et al., 2003). By contrast, those with low baseline levels of cortisol are far more likely to react in an adaptive fashion and are less likely to become depressed or develop immunological disorders leading to illness or death.












