by Rhawn Joseph, Ph.D.
Rhawn Joseph, Ph.D.
BrainMind.com
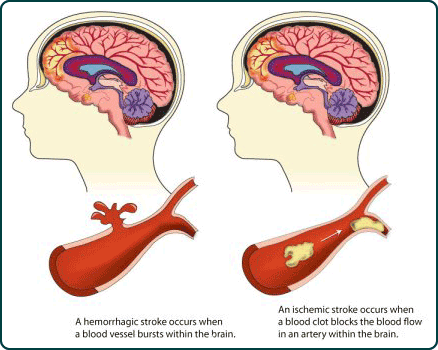
Cerebral infarction or stroke is often secondary to cerebrovascular disease and vascular abnormalities involving the heart and/or blood vessels which induce the formation of thrombi and emboli which in turn may occlude the blood vessels, which reduces the blood supply to the brain thereby inducing ischemia or or rupture of the vessel which causes hemorrhage.
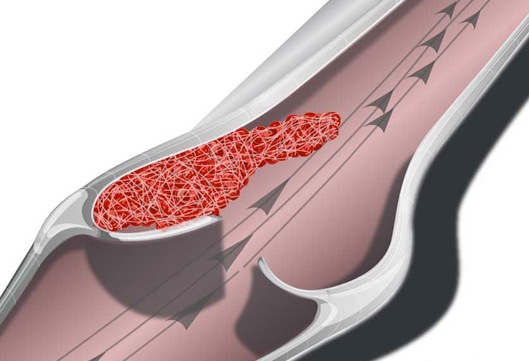
The development of thrombi is generally a consequence of (as well as a contributing factor to) atherosclerosis and injury to the endothelial cells lining the blood vessel. Blood platelets, lipids and cholesterol begin to be deposited in the injured arterial intima thus forming a mound of tissue (e.g. a patch-like accumulation of lipid or cholesterol).
When blood platelets begin to build up, they will rupture which in turn triggers the formation of thrombin and insoluable fibrin proteins, thromboplastin. These thrombin proteins act to create blood clots which further occlude the vessel.
Specifically, these fibrin proteins resemble web-like threads which are tangled together. These tangles act like nets which act to trap more blood cells and other debris which then clot together. Once started the clot tends to grow as more and more platelets are enmeshed and rupture thereby releasing more thromboplastin fibrin proteins. Furthermore, the plaques which narrow the lumen may serve as a nidus upon which even more thrombi come to adhere and from which emboli may be dislodged. Hence, these elevated fibrous plaques (thrombi) invariably grow larger and may even become vascularized. A complicated lesion is thus formed.
Obstructive Influences
As the clot grows blood flow is reduced which causes even more thromboplastin to accumulate; particularly in that these deposits create turbulence or eddy current which further damage the smooth endothelium of the artery as well as reduce resilience.
As the clot grows and thrombi increase in thickness, the lumen becomes progressively smaller which results in arterial dilation. By reducing the resilience and the diameter of the large arteries atherosclerosis can induce systolic hypertension. The driving force of the blood against vessel walls is therefore increased which causes further roughening and more plaque formation. Atherosclerosis is thus an insidious process.
When the lumen of the artery is narrowed by about 50%, pressure in the proximal segment increases whereas distal to the clot pressure begins to fall. With 70% reduction, flow is significantly decreased and neurological functioning may be altered. Typically, however, patients remain asymptomatic until there is a sudden reduction in blood flow, or thrombi or emboli are thrown off creating a complete obstruction. However, if the core of the plaque becomes necrotic and calcifies, the surface may disintegrate thus exposing the weakened underlying surface of the vessel. When this occurs the vessel may rupture and hemorrhage.
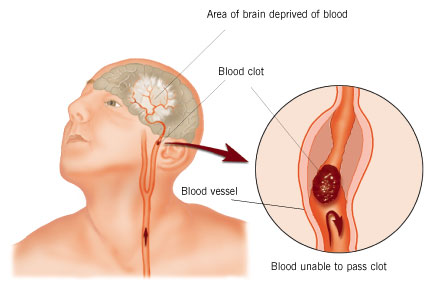
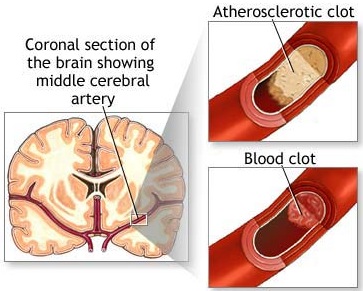
When a thrombotic clot enlarges sufficiently the patient suffers a cerebral infarct; i.e. thrombosis. A thrombosis is due to a thrombus--a clot produced by coagulated blood (thromboplastic: a protein that forms clots). Thrombotic strokes often occur during periods of prolonged inactivity or sleep such that the symptoms are present upon arising in the morning. As the patient attempts to swing out of bed or a chair, they find that they are weak on one side, that they cannot speak, or that some other deficit has developed.
Thrombotic strokes developed gradually. Symptoms and signs usually progress in a stepwise fashion, sometimes taking 2-4 days to fully develop. Patients deteriorate, improve slightly, then deteriorate further; a process referred to as stroke in evolution. This stepwise evolution is due to edema and alterations in blood flow and metabolism as thrombotic particles continually form and break off. Stabilization with improvement begins after 7-14 days after stroke onset.
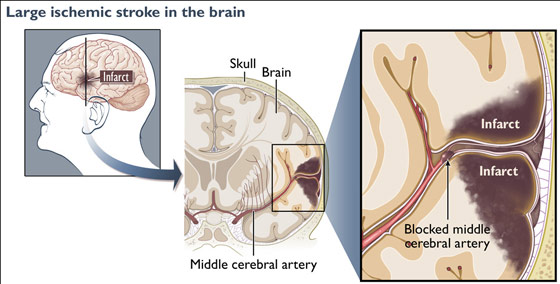
The size of the infarct in part is dependent on the presence and integrity of collateral vessels. If collateral circulatory channels are not in existence, occlusion exerts drastic effects and the infarct is quite large.
Warning signs include headache and/or previous transient neurological symptoms or a transient ischemic attacks (TIA). When transient ischemic attacks precede a stroke, they almost always stamp the process as thrombotic (Toole, 2002; Adams & Victor, 2013). However, the long the duration of the TIA, the grater is the likelihood that the infarct is due to embolism (Kimura et al., 2009).
The most common cause of thrombotic stroke is arteriosclerosis. Thrombosis follows clotting of the blood at a site where its flow is impeded by a sclerotic plaque usually in the bifurcations of the carotids. Complete occlusion of the carotids is always the consequence of thrombosis.
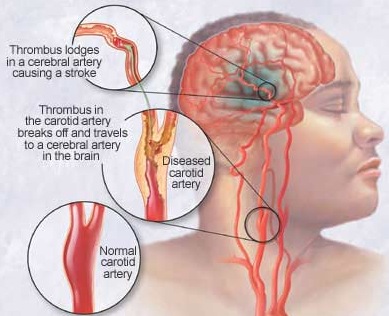
In contrast to a thrombosis which is due to a localized buildup, emboli are free particles which become lodged within a vessel causing occlusion. Like thrombosis embolic strokes create an ischemic cerebral infarct. However, unlike thrombosis, embolitic strokes are often followed later by hemorrhage (Chamorro et al. 1995). Both thrombotic and cerebral embolic strokes occur most commonly among older individuals. Among younger age groups, however, embolism is the most frequent culprit.
An embolism may be composed of thrombotic particles which have broken off from a clot, air bubbles, masses of bacteria, or blood clots. They may also consist of cholesterol crystals which are thrown off from atheromatous plaques situated in the vertebral-basilar and carotid arteries, pieces of tumor cells from the lung, stomach, or kidney which subsequently take root in the brain after drifting in the bloodstream, and fat globules released into the bloodstream after trauma to the marrow of the long bones (Toole 2002). Oral contraceptives among women can give rise to the development of embolism, and an association among those with mitral valve prolapse has been noted (Jackson, 2005; Toole. 2002).
CARDIAC EMBOLIC ORIGINS
Cerebral emboli may develop when there is plaque buildup on the cardiac or carotid valves and may also result from mitral stenosis, arterial or auricle fibrillation, aortic valve calcification, fragments of left atria or ventricular thrombus, mitral valve pollips, myocardial infarction, or vegetations of infective endocarditis. Heart disease, mitral stenosis and atrial fibrillation are thus major risk factors for cerebral embolism. Chronic atrial fibrillation in particular carries a high risk of stroke (Bornstein & Kelly 2001; Petersen & Godtfredsen, 2005; Wolf et al, 2003), the clot coming from a paralyzed atrium. When the embolus is infected, meningitis or abscess may develop. Indeed, it is estimated that approximately 90% of emboli from the heart end up in the brain. Thus embolic stroke is often a sign of systemic heart disease.
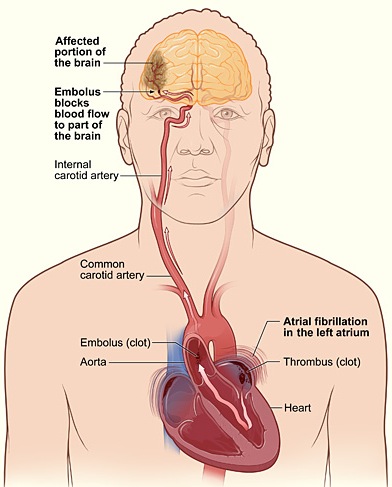
EMBOLI & THE CAROTID & LEFT MIDDLE CEREBRAL ARTERY.
In general, emboli expelled from the heart and the left ventricle are carried into the brachiocephalic artery and then the left common carotid. From the left carotid, emboli are transmitted and occlude the arteries supplying the left cerebral hemisphere. Hence, the left side of the brain is a common destination of emboli originating in the heart (Absher & Toole 1996).
When emboli are repeatedly freed into the blood system they tend to lodge in the same artery, usually the middle cerebral. This is because the middle cerebral artery is a direct extension of the internal cartoid and receives almost 80% of the blood. Hence, most emboli end up within the middle cerebral system.
ONSET
After the lodgment of an embolus the vessel usually goes into spasm and thrombosis may occur. Embolic strokes characteristically begin suddenly, often while the patient is awake and active. There is usually no warning, and the effects of the infarction may reach its peak almost immediately (Adams & Victor 2013). However, it is not uncommon for the initial manifestations of these strokes to have a duration of 2 to 15 minutes (Kimura, et al., 2009).
Cerebral embolization will be major if the embolus is large or temporary is the fragment is small. Smaller fragments have a greater likelihood of disintegrating. Not infrequently an extensive disturbance from embolism reverses itself dramatically within a few hours or a day or two (Absher & Toole 1996; Meier & Strauman 2001). It is not uncommon for many to suffer a recurrence, frequently with severe damage (Kimura et al., 2009). Early anticoagulant therapy should be emphasized as it is important in the prevention of embolic stroke recurrence (Chomorro et al. 1995; Lodder & van der Lugt, 2003; Koller, 1982).
EMBOLIC HEMORRHAGE
Approximately 65% of those with a cerebral embolic stroke develop hemorrhagic infarction. Conversely, less than 20% of those with thrombosis will develop hemorrhage (Ott et al. 2005). Similarly, approximately 50% of all hemorrhagic infarcts are associated with embolic strokes (Fisher & Adams, 2001; Hart & Easton, 2005). Hence embolic strokes have a special propensity for hemorrhagic transformation and hemorrhagic infarction nearly always indicates embolism.
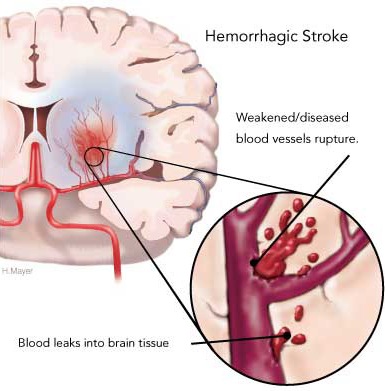
Blood vessels effected by embolism characteristically hemorrhage within 12-48 hours (Cerebral Embolism Study Group, 2014; Hart & Easton, 2005). However, the hemorrhage may take several days to fully develop (Laureno et al. 2007). Hemorrhagic transformation occurs when the emboli disintegrates and/or migrates distally thus allowing reperfusion of the damaged vessel (Fisher & Adams, 2001: Jorgensen & Torvic, 2005). That is, when a vessel is occluded, it is damaged and weakened, and both the brain and the involved portion of the vessel may become necrotic. When the weakened portion of a previously occluded vessel is subsequently reexposed to the full force of arterial pressure it ruptures and hemorrhages. Fortunately, blood vessels have he capacity to regenerate.
Frequently embolic hemorrhages are asymptomatic (Hakim et al. 2003; Ott, et al., 2005) and supposedly benign since the tissue involved has already been damaged. However, if the hemorrhage is secondary to anticoagulant therapy the bleeding may be more profuse and cause significant neurological deterioration and even death.
Transient ischemic attacks (TIAs)are due to a brief and temporary reduction in blood flow to a focal region within the brain, though the TIA itself may last from 2 to 15 minutes (Kimura et al., 2009). Short duration TIA's are accompanied by transitory and focal neurological disturbances which seem to immediately and completely resolve with no apparent residuals. However, longer duration TIAs are indicative of serious arterial disease (Kimura et al., 2009) which increase the risk of stroke.
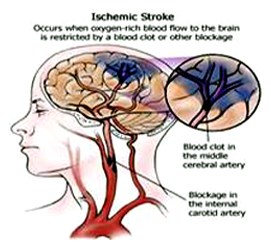
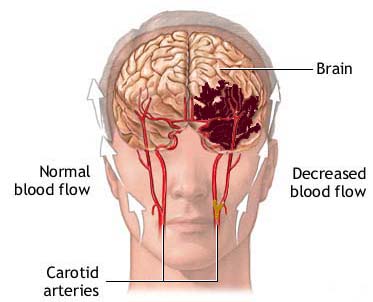
The most frequent cause of TIAs is atherosclerosis and microembolization of platelet aggregates from ulcerated atherosclerotic plagues within the carotids or from the cardiac valves (Adams & Victor 2013). These conditions act to reduce blood flow. Other causes include vascular spasm, aggregations of cholersterol crystals, fibrin and/or blood platelets which temporarily occlude a vessel, or a combination of these factors.
TIAS are related to the microcirculation where small irritants such as an embolus can lead to blockage. However they can involve any cerebral or cerebellar artery. TIA's may last a few seconds or up to 12-24 hours. As noted, most last 2 to 15 minutes (Kimura et al., 2009; Toole, 2002). The symptoms are transient because the occluding emboli almost immediately disintegrates which allows for the immediate reestablishment of blood flow and functional recovery.
TIAS which last less than 60 minutes are different from those of longer duration. The shorter ones are due to emboli which travel from one artery to another, whereas the longer ones are from emboli passed by the heart.
Hence, artery to artery emboli tend to be smaller and more quickly disintegrate whereas those from the heart tend to be larger and/or greater in number. Some patients may experience a single episode of TIA in a life time whereas others may have as many as 20 attacks in a single day. Usually they are fewer than one or two a week.
) Weakness or numbness of a single finger may be the only manifestation of a TIA. However, a person may suffer a TIA and not be conscious of it, or the manifestation may be a disturbance in memory, speech and word finding, brief confusion, etc. Usually with these kinds of disturbances the patients and their families ignore, fail to notice, or attribute little significance to these attacks as they are transient.
In some instances, the TIA may take the form of temporary blindness, like a shade being pulled over the eye. It is when the patient becomes aware that there is a loss of vision, or a loss of strength in the hand or foot which rapidly progresses to involve the entire extremity that they become alarmed.
TIAs can occur within any region of the cerebral vasculature. Some investigators, however, have classified them as either opthalamic, hemispheric, or vertebral-basilar.
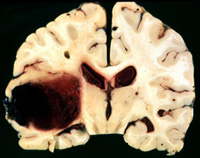
HEMISPHERIC TIAs
In hemispheric attacks, ischemia occurs foremost in the distal territory of the middle cerebral artery, producing weakness or numbness in the opposite hand and arm--however, different combinations may occur: face and lips, hand and foot, fingers alone, etc. In ocular attacks, transient monocular blindness occurs, and is described as a shade falling smoothly over the visual field until the eye is blind. The attack clears smoothly and uniformly.
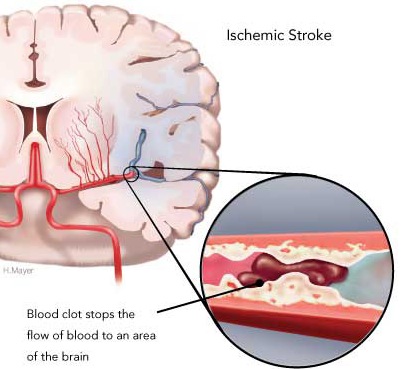
VERTEBRAL-BASILAR TIAs
If it occurs in the vertebral-basilar sytem, there may be dizziness, diplopia, dysarthria, bifacial numbness, and weakness or numbness involving part or parts of the body, or even both body halves (due to the involvement of diverse regions of the brainstem and thalamus supplied by the vertebral-basilar system). Other symptoms may include in order of frequency: headache, staggering, veering to one side, feeling of crosseyedness, dark or blurred or tunnel vision, partial or complete blindness, pupillary changes, and paralysis of gaze (Toole, 2002). In basilar artery disease each side of the body may be affected alternately. There is good evidence that TIAs may be abolished by anticoagulant drugs.
TIA & STROKE
Forty percent of individuals with TIAs subsequently suffer brain infarction, the majority of which occur within the first six months (Wolf et al., 2003). Stroke may occur after the first or second TIA or only after 100s have occurred over a periods of weeks or months. However, the greatest risk of stroke following TIA is within the first year and in particular the first 30 days; 20% occuring in the first month, and 50% within a year (Bornstein & Kelly 2001; Wolf et al., 2003).
When TIAs precede a stroke, however, they almost always stamp the process as thrombotic (atherosclerotic thrombosis). Nevertheless, it is noteworthy that although thrombotic strokes are said to occur predominantly at night the majority of infarctions in general occur between 6 am and 6 pm (Van Der Windt & Gijn, 2008).
Risk Factors
Risk factors include cigarette smoking, previous history of stroke or TIA, ischemic heart disease, and diabetes (Bornstein & Kelly 2001; Howard et al., 2007). As the number of risk factors increase the prognosis for recovery following stroke declines.
REFERENCES
















