Rhawn Gabriel Joseph, Ph.D.
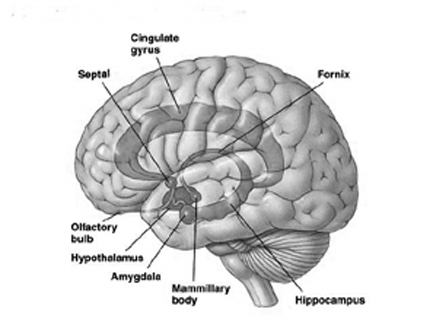

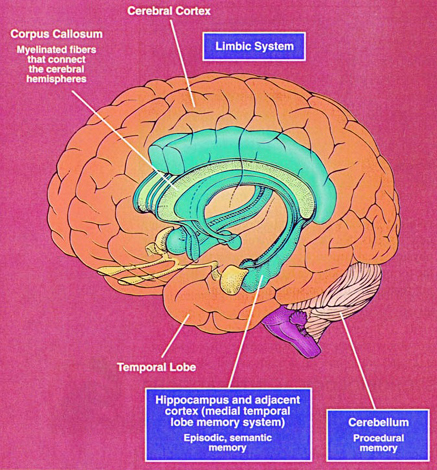
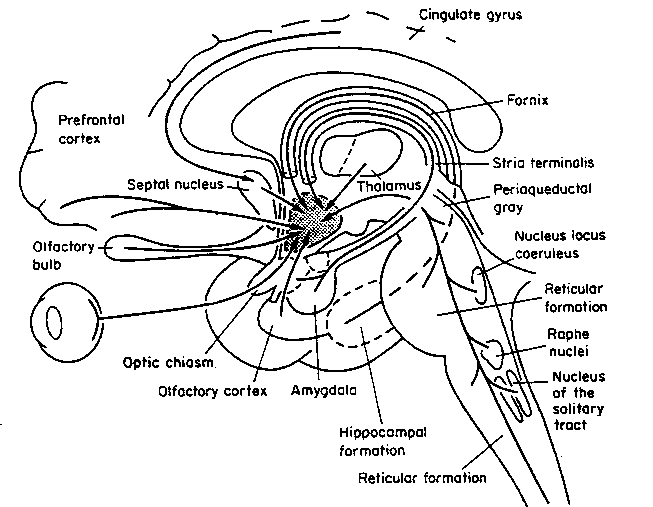
Buried within the depths of the cerebrum are several large aggregates of limbic structures and nuclei which are preeminent in the control and mediation of memory, emotion, learning, dreaming, attention, and arousal, and the perception and expression of emotional, motivational, sexual, and social behavior including the formation of loving attachments. Indeed, the limbic system not only controls the capacity to experience love and sorrow, but it governs and monitors internal homeostasis and basic needs such as hunger and thirst (Bernardis & Bellinger 2007; Gloor 1992, 2010; Joseph, 1990, 1992, 2000a; LeDoux 1992, 2012; MacLean, 1973, 1990; Rolls, 1984, 1992; Smith et al. 1990), including even the cravings for pleasure-inducing drugs (Childress, et al., 2009).
The structures and nuclei of the limbic system are exceedingly ancient, some of which began to evolve over 450 million years ago. Over the course of evolution, these emotional structures have expanded in size, some becoming increasingly cortical in response to increased environmental opportunities and demands. In fact, as the neocortical forebrain expanded and until as recently as 50 million years ago, the cerebrum of the ancestral line that would eventually give rise to humans, was dominated by the limbic system.
However, over the course of evolution a mantle of neocortex began to develop and enshroud the limbic system; evolving at first to serve limbic needs in a way that would maximize the survival of the organism, and to more efficiently, effectively, and safely satisfy limbic needs and impulses. In consequence, the frontal, temporal, parietal, and occipital lobes evolved covered with a neocortical mantle, that in humans would come to be associated with the conscious, rational mind. Sometimes, however, even in the most rational of humans, emotions can hijack the logical mind, and the neocortex, and even peaceful people might be impelled to murder even those they love.
Indeed, the old limbic brain has not been replaced and is not only predominant in regard to all aspects of motivational and emotional functioning, but is capable of completely overwhelming "the rational mind" due in part to the massive axonal projections of limbic system to the neocortex. Although over the course of evolution a new brain (neocortex) has developed, Homo sapiens sapiens ("the wise may who knows he is wise") remains a creature of emotion. Humans have not completely emerged from the phylogenetic swamps of their original psychic existence.

Hence, due to these limbic roots, humans not uncommonly behave "irrationally" or in the "heat of passion," and get into fights, have sex with or scream and yell at strangers thus act at the behest of their immediate desires; sometimes falling "madly in love" and at other times, acting in a blind rage such that even those who are 'loved" may be slaughtered and murdered.
Indeed, emotion is a potentially powerful overwhelming force that warrants and yet resists control-- as something irrational that can happen to a someone ("you make me so angry") and which can temporarily hijack, overwhelm, and snuff out the "rational mind."
The schism between the rational and the emotional is real, and is due to the raw energy of emotion having it's source in the nuclei of the ancient limbic lobe -- a series of structures which first make their phylogenetic appearance over a hundred million years before humans walked upon this earth and which continue to control and direct human behavior.
The hypothalamus could be considered the most "primitive" aspect of the limbic system, though in fact the functioning of this sexually dimorphic structure is exceedingly complex. The hypothalamus regulates internal homeostasis including the experience of hunger and thirst, can trigger rudimentary sexual behaviors or generate feelings of extreme rage or pleasure. In conjunction with the pituitary the hypothalamus is a major manufacturer/secretor of hormones and other bodily humors, including those involved in the stress response and feelings of depression.

In fact, almost every region of the cerebrum interacts with and communicates with the hypothlamus and is subject to its influences (Swanson, 2007). Moreover, the hypothalamus utilizes the blood supply to transmit hormonal and humoral messages to peripheral organs as well as other brain structures and utilizes the blood supply to receive information as well, thus bypassing the synaptic route utilized by almost all other regions of the neuroaxis (Markakis & Swanson, 2010). Through the blood supply (as well as via the cerebrospinal fluid), the hypothalamus not only regulates, but is subject to feedback regulation by the same structures that it controls.
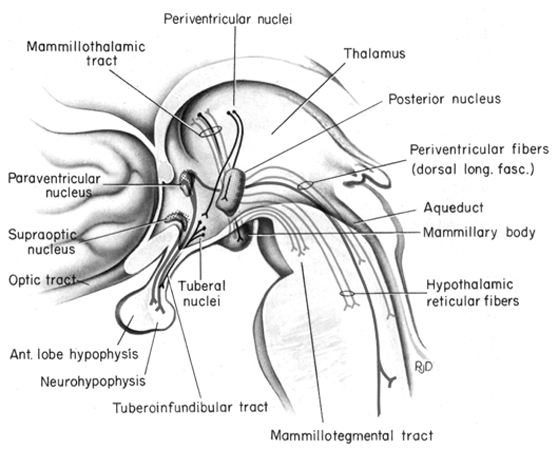
Certain areas of the diencephalon, midbrain, and brainstem, are exceedingly exceedingly sensitive to hormones, humors, and peptides circulating within the blood plasma, and the cerebrospinal fluids; chemosensory information which is used for maintaining homeostasis. Broadly considered, these chemosensory sensitive areas are generally located near or surrounding the cerebral ventricles (Johnson & Gross, 2013) and they tend not to be effected by the so called "blood brain barrier;" referred to as circumventricular organs (CVOs). There are perhaps dozens of CVO's at least 8 of which are located in or near the ventricular systems which feed the brainstem and diencephalon including the hypothalamus, pineal gland and pituitary (Johnson & Gross, 2013).
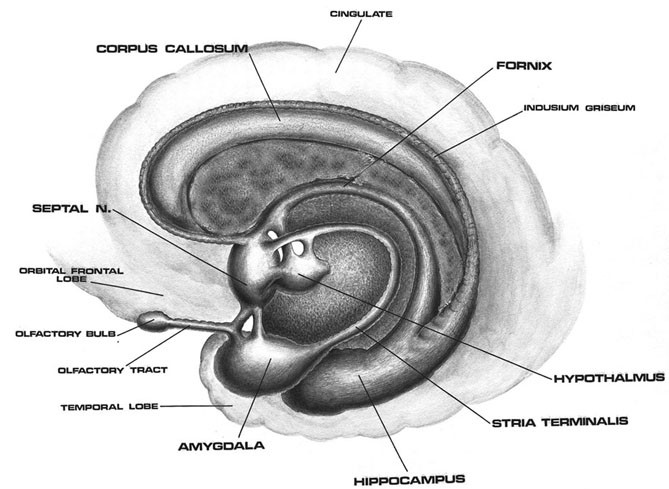
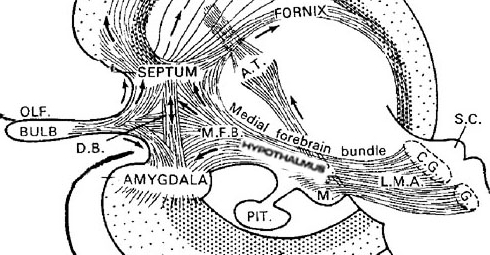
The hypothalamus, however, does not act solely through the blood supply or via cerebrospinal fluid, and its also receives sensory information synaptically, and often indirectly, as is the case with the majority of olfactory fibers. In general, sensory stimuli reach the hypothalamus from a variety of routes. These include the solitary tract of the brainstem, a structure which receives, processes, and transmits data received principally from the vagus and glosopharyngeal cranial nerves. Through this pathway the lateral hypothalamus is informed about cardivocascular activities, respiration, and taste. These pathways are also bidirectional (Swanson, 2007). Other major pathways include the medial forebrain bundle (which contains axons from a variety of different cellular groups) and the stria terminalis through which the amygdala and hypothalamus interact. The hypothalamus also maintains massive interactive pathways with the frontal lobes and septal nuclei (Risvold & Swanson, 2012).
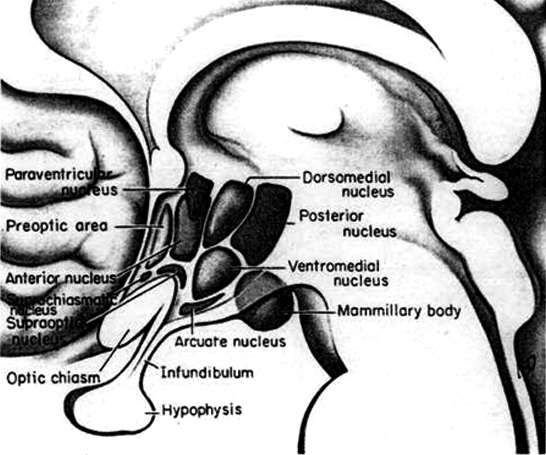
Broadly considered, the hypothalamus consists of three longitudinal subdivisions which extend along its anterior to posterior axis. These are the medial, lateral, and periventricular (Swanson, 2007). The periventricular zone is concerned with neuroendocrine regulation, whereas the lateral and medial zones are concerned with affective states, including hunger and thirst. These zones, in turn can be further subdivided into subnuclei.
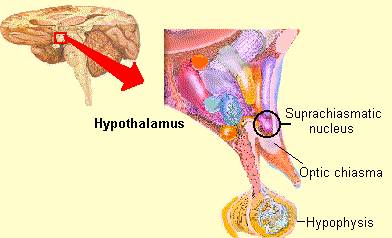
Phylogenetically, structurally, and embryologically the hypothalamus is traditionally considered part of the diencephalon. During embryological development it emerges from the diencephalic vessicle of the neural tube along with those anterior-lateral evaginations which become the optic nerves and retina of the eye, as well as the pituitary gland (ventrally) and the pineal gland and thalamus (dorsally). There is some dispute, however, over the developmental patterns of the hypothalamus, as some scientists believe that it develops from the outside in (the "hollow hypothalamus hypothesis").
On the other hand, the hypothalamus originates from the medially situated neuroepithelium, and thus begins its developmental journey in a medial (or rather paramedial) to lateral arc, such that it appears that the medial hypothalamus is fashioned (and matures) in advance of the lateral hypothalamus.

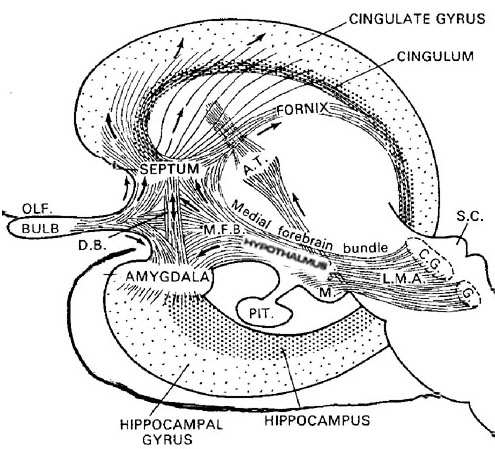
The hypothalamus is exceedingly responsive to olfactory (and pheromonal) input. Perhaps reflecting this partial and putative olfactory origin is the fact that this structure utilizes chemical (hormonal, humoral) molecules to communicate with other areas of the brain, and reacts to these same molecules as well as olfactory cues, including those directly related to sexual status.
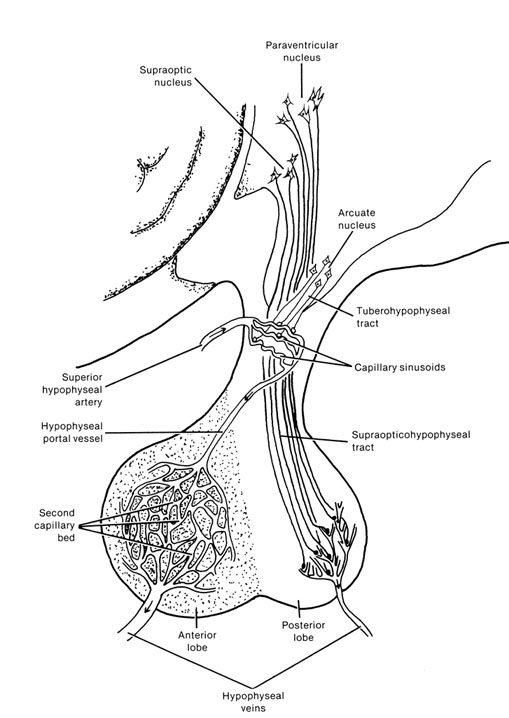
It is this olfactory-chemical origin and sensitivity which in turn may explain why portions of the hypothalamus (like the amygdala) are also sexually dimorphic and reacts to pheromonal sensory stimuli including those which signal sexual status. That is, structurally and functionally the hypothalamus of males and females are stucturally dissimilar (Bleier et al. 1982; Dorner, 1976; Gorski et al. 1978; Rainbow et al. 1982; Raisman & Field, 1971, 1973) and perform different functions depending on if one is a man or a woman, and if a woman is sexually receptive, pregnant, or lactating. For example, the sexually dimorphic supraoptic and paraventricular nuclei project (via the infundibular stalk) to the posterior lobe of the pituitary which may then secrete oxytocin--a chemical which can trigger uterine contractions as well as milk production in lactating females (and which can thus make nursing a pleasurable experience). The male hypothalamus/pituitary does not perform this function.
SEXUAL DIMORPHISM IN THE HYPOTHALAMUS
As is well known, sexual differentiation is strongly influenced by the presence or absence of gonadal steriod hormones during certain critical periods of prenatal development in many species including humans. Not only are the external genitalia and other physical features sexually differentiated but certain regions of the brain have also been found to be sexually dimorphic and differentially senstitive to steriods, particularly the preoptic area and ventromedial nucleus of the hypothalamus, as well as the amygdala (Bleier et al. 1982; Dorner, 1976; Gorski et al. 1978; Rainbow et al. 1982; Raisman & Field, 1971, 1973).
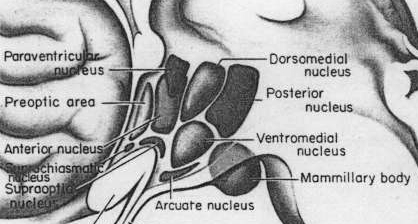
Indeed it has now been well established that the amygdala and the hypothalamus (specifically the anterior commissure, anterior-preoptic, ventromedial and suprachiasmatic nuclei) are sexually differentiated and have sex specific patterns of neuronal and dendritic development, (Allen et al. 1989; Blier et al. 1982; Gorski et al. 1978; Rainbow et al. 1982; Raisman & Field, 1971, 1973; Swaab & fliers, 2005).
This is a consequence of the presence or absence of testosterone during fetal development in humans, or soon after birth in some species such as rodents. Specifically, the presence or absence of the male hormone, testosterone during this critical neonatal period, directly effects and determines the growth and pattern of interconnections between the amygdala and hypothalamus, between axons and dendrites in these nuclei as well as the hippocampus, septal nuclei, olfactory system (ref), and thus the organization of specific neural circuits. In the absence of testosterone, the female pattern of neuronal development occurs. Indeed, it is the presence or absence of testosterone during these early critical periods that appear to be responsible for neurological alterations which greatly effect sex differences in thinking, sexual orientation, aggression, and cognitive functioning (Barnett & Meck, 1990; Beatty, 1992; Dawson et al. 1975; Harris, 1978; Joseph, et al. 1978; Stewart et al. 1975).
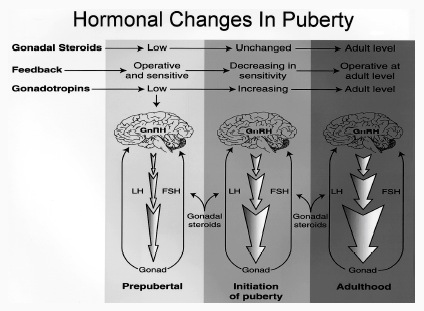
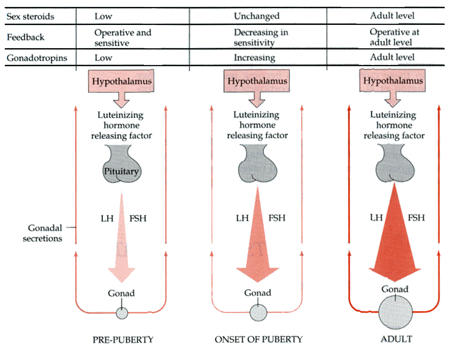
For example, if the testes are removed prior to differentiation, or if a chemical blocker of testosterone is administered thus preventing this hormone from reaching target cells in the limbic system, not only does the female pattern of neuronal development occur, but males so treated behave and process information in a manner similar to females (e.g., Joseph et al. 1978); i.e. they develop female brains and think and behave in a manner similar to females. Conversely, if females are administered testosterone during this critical period, the male pattern of differentiation and behavior results (see Gerall et al. 1992 for review).
That the preoptic and other hypothalamic regions are sexually dimorphic is not surprising in that it has long been known that this area is extremely important in controlling the basal output of gonadotrophins in females prior to ovulation and is heavily involved in mediating cyclic changes in hormone levels (e.g. FSH, LH, estrogen, progesterone). Chemical and electrical stimulation of the preoptic and ventromedial hypothalamic nuclei also triggers sexual behavior and even sexual posturing in females and males (Hart et al., 2005; Lisk, 1967, 1971) and, in female primates, even maternal behavior (Numan, 2005). In fact, dendritic spine density of ventromedial hypothalamic neurons varies across the estrus cycle (Frankfurt et al., 1990) and thus presumably during pregnancy and while nursing.



Specifically, the hypothalamic neurons secrete gonadotropin-releasing hormone, which acts on the anterior lobe of the pituitary which secretes gonadotropins. However, given that in females, this is a cyclic event, whereas in males sperms are constantly reproduced, is further evidence of the sexual dimorphism of the hypothalamus.
Although the etiology of homosexuality remains in question, it has been shown that the ventromedial and anterior nuclei of the hypothalamus of male homosexuals demonstrate the female pattern of development (Levay, 1991; Swaab, 1990). When coupled with the evidence of male vs female and homosexual differences in the anterior commissure which links the temporal lobe and sexually dimorphic amygdala (see below) as well as the similarity between male homosexuals and women in regard to certain cognitive attributes including spatial-perceptual capability (see below), this raises the possibility that male homosexuals are in possession of limbic system that is more "female" than "male" in functional as well as structural orientation.
It is also interesting to note that the sexually dimorphic preoptic region contains thermosensitive neurons, and controls the physiological and behavior responses to excessive external cold or heat. That is, it is responsible for internal thermoregulation and thus heat loss or retention (Alam et al., 2011). Although we can only speculate, it may well be sex differences in this structure which accounts (at least in part) for the stereotypical differences in male vs female perceptions of cold, and why, stereotypically, females (despite their extra-layers of heat-retaining fat) are more likely to insist on elevating room temperature.
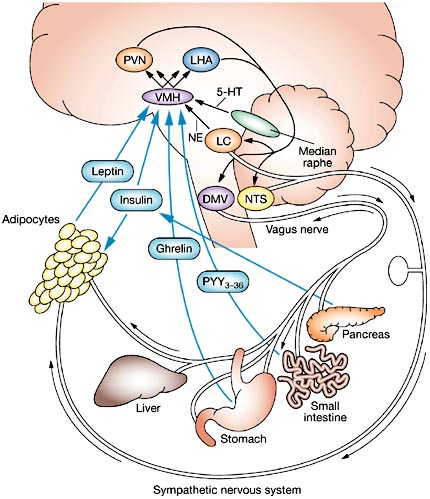
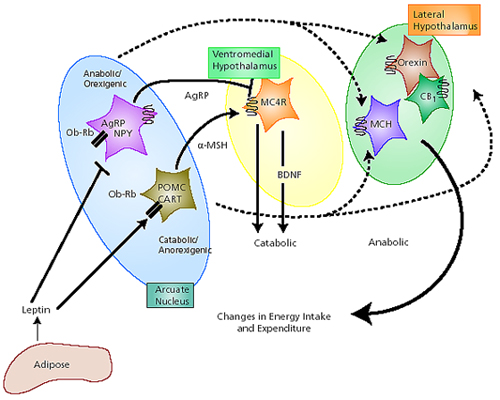

The lateral and medial region are highly involved in monitoring internal homeostasis and motivating the organism to respond to internal needs such as hunger and thirst (Anand & Brobeck, 1951; Bernardis & Bellinger 2007; Hetherington & Ranson, 1940). For example, both nuclei appear to contain receptors which are sensitive to the body's fat content (lipostatic/caloric receptors) and to circulating metabolites (e.g. glucose) which together indicate the need for food and nourishment. For example, when food is digested, the viscera secretes various hormones which act on the alimentary tract, which in turn stimulates the solitary tract (ST) which projects directly to the hypothalamus. However, in the absence of food, the viscera also begins to secrete various hormones which when coupled changes in caloric blood levels, signals to the hypothalamus the need for food. The lateral hypothalamus also appears to contain osmoreceptors (Joynt, 1966) which determine if water intake should be altered.
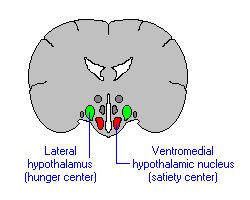
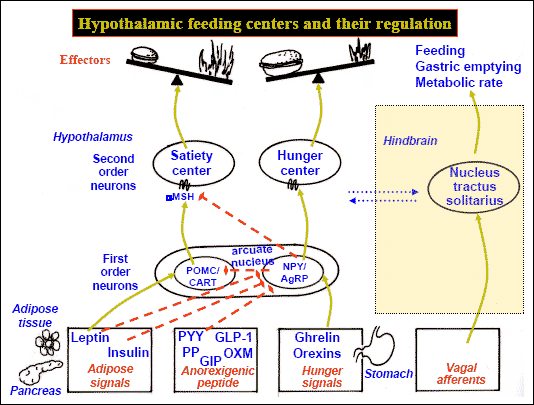
If the medial hypothalamus is surgically destroyed, inhibitory influences on the lateral region appear to be abolished such that hypothalamic hyperphagia and severe obesity result (Anand & Brobeck, 1951; Hoebel & Tetelbaum, 1966; Teitelbaum, 1961). Hence, the medial area seems to act as a satiaty center; but, a center that can be overridden.
Specifically, with ventromedial lesions, animals not only eat more, but the intervals between meals becomes shorter such that they eat more meals. Thus they begin to gain weight. In part this is also due to changes in the sympathetic nervous system which increases vagal activity, thus signaling the need for more food. As noted, the ST is bidirectional.
Normally the hypothalamus can act via the ST and thus the vagal complex and can signal satiation. However, with medial destruction, the ST becomes hyperactive thus inducing parasympathetic overactivity which induces more rapid gastric emptying and the rapid storage of ingested calories. Because these calories are rapidly stored, caloric blood levels are reduced and the lateral hypothalamus is stimulated to begin eating again--which explains the increased frequency of meals. In fact, if animals that have become obese following ventromedial lesions are starved back to their normal weight, once they are allowed free access to food, they again become obese (Hoebel & Tetelbaum, 1966).



In part, these nuclei exert these differential influences on eating and drinking via motivational/emotional influences they exert on other brain nuclei (e.g. via reward or punishment). However, it should be stressed that there are a number of other structures and hormones and peptides involved, including the pancreatic islets, and insulin secretion.
PLEASURE & REWARD
In 1952, Heath (cited by Maclean, 1969) reported what was then considered remarkable. Electrical stimulation near the septal nuclei elicited feelings of pleasure in human subjects: "I have a glowing feeling. I feel good!" Subsequently, Olds and Milner (1954) reported that rats would tirelessly perform operants to receive electrical stimulation in this same region and concluded that stimulation "has an effect which is apparently equivalent to that of a conventional primary reward." Even hungry animals would demonstrate a preference for self-stimulation over food.
Feelings of pleasure (as demonstrated via self-stimulation) have been obtained following excitation to a number of diverse limbic areas including the olfactory bulbs, amygdala, hippocampus, cingulate, substantia nigra (a major source of dopamine), locus coeruleus (a major source of norepinephrine), raphe nucleus (serotonin), caudate, putamen, thalamus, reticular formation, medial forebrain bundle, and orbital frontal lobes (Brady, 1960; Lilly, 1960; Olds & Forbes, 1981; Stein & Ray, 1959; Waraczynski et al. 2007).
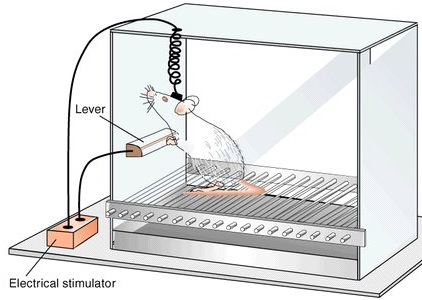

Electrophysiological studies of single lateral hypothalamic neurons indicate that these cells become highly active in response to rewarding food items (Nakamura & Ono, 1986). In fact, many of these cells will become aroused by neutral stimuli repeatedly associated with reward such as a cue-tone --even in the absence of the actual reward (Nakamura & Ono, 1986; Ono et al. 1980). However, this ability to form associations appears to be secondary to amygdaloid activation (Fukuda et al. 2007) which in turn influences hypothalamic functioning.
Nevertheless, if the lateral region is destroyed the experience of pleasure and emotional responsiveness is almost completely attenuated. For example, in primates, faces become blank and expressionless, whereas if the lesion is unilateral, a marked neglect and indifference regarding all sensory events occurring on the contralateral side occurs (Marshall & Teitelbaum, 1974). Animals will in fact cease to eat and will die.
AVERSION
In contrast to the lateral hypothalamus and it's involvement in pleasurable self-stimulation, activation of the medial hypothalamus is apparently so aversive that subjects will work to reduce it (Olds & Forbes, 1981). Hence, electrical stimulation of the medial region leads to behavior which terminates the stimulation--apparently so as to obtain relief (e.g. active avoidance). In this regard, when considering behavior such as eating, it might be postulated that when upper weight limits (or nutritional requirements) are met, the medial region becomes activated which in turn leads to behavior (e.g. cessation of eating) which terminates its activation.
It is possible, however, that medial hypothalamic activity may also lead to a state of quiescence such that the organism is motivated to simply cease to respond or to behave. In some instances this quiescent state may be physiologically neutral, whereas in other situations medial hypothalamic activity may be highly aversive. Quiescence is also associated with parasympathetic activity which is mediated by the medial area.
The hypothalamus, via it's rich interconnections with other limbic regions including the neocortex and frontal lobes, it able to mobilize and motivate the organism to either cease or continue to behave. Nevertheless, at the level of the hypothalamus, the emotional states elicited are very primitive, diffuse, undirected and unrefined.
The organism feels pleasure in general, or aversion/unpleasure in general. Higher order emotional reactions (e.g. desire, love, hate, etc.) require the involvement of other limbic regions as well as neocortical participation.

Nevertheless, in humans, disturbances of hypothalamic functioning (e.g. due to an irritating lesion such as tumor) can give rise to seemingly complex, higher order behavioral-emotional reactions, such as pathological laughter and crying which occurs uncontrollably. However, in some cases when patients are questioned, they may deny having any feelings which correspond to the emotion displayed (Davison & Kelman, 1939; Ironside, 1956; Martin, 1950). In part, these reactions are sometimes due to disinhibitory release of brainstem structures involved in respiration, whereas in other instances the resulting behavior is caused by hypothalamic triggering of other limbic nuclei.
UNCONTROLLED LAUGHTER
Pathological laughter has frequently been reported to occur with hypophyseal and midline tumors involving the hypothalamus, aneurysm in this vicinity, hemorrhage, astrocytoma or pappiloma of the 3rd ventricle (resulting in hypothalamic compression), as well as surgical manipulation of this nucleus (Davison & Kelman, 1939; Dott, 1938; Foerster & Gabel, 1933; Martin, 1950; Money & Hosta, 1967; Ironside, 1956; List, Dowman, & Bagheiv, 1958).
For example, Martin (1950, p.455) describes a man who while "attending his mother's funeral was seized at the graveside with an attack of uncontrollable laughter which embarrassed and distressed him considerably." Although this particular attack dissipated, it was soon accompanied by several further fits of laughter and he died soon thereafter. Post-mortem a large ruptured aneurysm was found, compressing the mammillary bodies and hypothalamus.
In a similar case (Anderson, 1936; Cited by Martin, 1950), a patient literally died laughing following the eruption of the posterior communicating artery which resulted in compression (via hemorrhage) of the hypothalamus. "She was shaken by laughter and could not stop: short expirations followed each other in spasms, without the patient being able to make an adequate inspiration of air, she became cyanosed and nothing could stop the spasm of laughter which eventually became noiseless and little more than a grimace. After 24 hours of profound coma she died."
Because laughter in these instances has not been accompanied by corresponding feeling states, this pseudo-emotional condition has been referred to as "sham mirth" (Martin, 1950). However, in some cases, abnormal stimulation in this region (such as due to compression effects from neoplasm) has triggered corresponding emotions and behaviors -- presumably due to activation of other limbic nuclei.
For example, laughter has been noted to occur with hilarious or obscene speech--usually as a prelude to stupor or death--in cases where tumor has infiltrated the hypothlamus (Ironside, 1956). In several instances it has been reported by one group of neurosurgeons (Foerster & Gagel, 1933) that while swabbing the blood from the floor of the 3rd ventricle, patients "became lively, talkative, joking, and whistling each time the infundibular region of the hypothalamus was manipulated." In one case, the patient became excited and began to sing.
HYPOTHALAMIC RAGE
Stimulation of the lateral hypothalamus can induce extremes in emotionality, including intense attacks of rage accompanied by biting and attack upon any moving object (Flynn et al. 1971; Gunne & Lewander, 1966; Wasman & Flynn, 1962). If this nucleus is destroyed, aggressive and attack behavior is abolished (Karli & Vergness, 1969). Hence, the lateral hypothalamus is responsible for rage and aggressive behavior.

In man, inflammation, neoplasm, and compression of the hypothalamus have also been noted to give rise to rage attacks (Pilleri & Poeck, 1965), and surgical manipulations or tumors within the hypothalamus have been observed to elicit manic and rage-like outbursts (Alpers, 1940). These appear to be release phenomenon, however. That is, rage, attack, aggressive, and related behaviors associated with the hypothalamus appears to be under the inhibitory influence of higher order limbic nuclei such as the amygdala and septum (Siegel & Skog, 1970). When the controlling pathways between these areas are damaged (i.e. disconnection) sometimes these behaviors are elicited.
For example, Pilleri and Poeck (1965) described a man with severe damage throughout the cerebrum including the amygdala, hippocampus, cingulate, but with complete sparing of the hypothalamus who continually reacted with howling, growling, and baring of teeth in response to noise, a slight touch, or if approached. Hence, the hypothalamus being released responds reflexively in an aggressive-like non-specific manner to any stimulus. Lesions of the frontal-hypothalamic pathways have been noted to result in severe rage reactions as well (Fulton & Ingraham, 1929; Kennard, 1945).
Nevertheless, like "sham mirth", rage reactions elicited in response to direct electrical activation of the hypothalamus immediately and completely dissipate when the stimulation is removed. As such, these outbursts have been referred to as "sham rage".
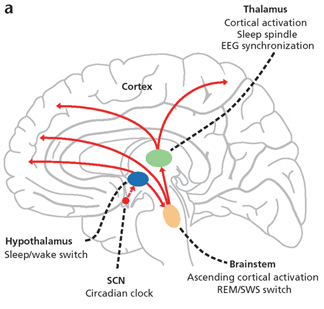
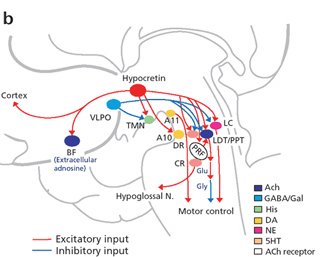
For example, the hypothalamic-pituitary axis secretes melatonin in phase with the circadian rhythm. Phase-delayed rhythms in plasma melatonin secretion have been repeatedly noted in most (but not all) studies of individuals with SADs (see Wirz-Justice et al. 2013, for review). However, with light therapy, not only is the depression relieved but the melatonin secretions return to normal. This is significant for melatonin is derived from tryptophan via serotonin and low serotonin levels have been directly linked to depression (e.g. Van Pragg 1982).
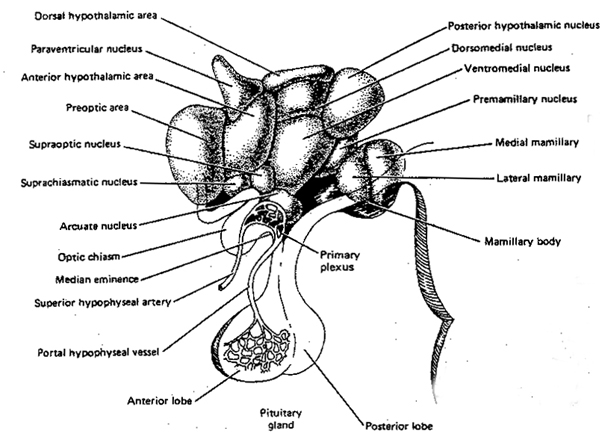

It is also possible, however, that although light therapy can assist in alleviating depressive symptoms associated with SADs, that the deregulation of the SCN (and melatonin/serotonin) might be unrelated to light, temperature, or aging, but may be a consequence of stress on the hypothalamus (Chauloff 2013). For example, the hypothalamic-pituitary axis is tightly linked with and in fact mediates stress induced alterations in serotonin (see Chauloff 2013, for review); as well as norepinephrine (Swann et al. 2014) which has also been repeatedly implicated in the genesis of depression.
These events in turn appear to be under the modulating influences of norepinephrine. That is, as stress increases, NE levels decrease, which triggers the activation of the HPA axis. As is well known, low levels of NE are associated with depression.
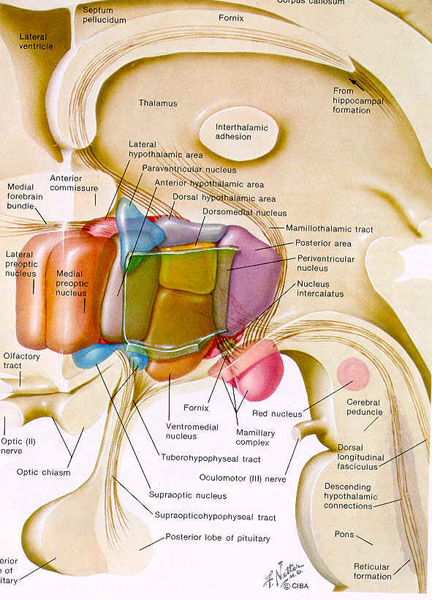
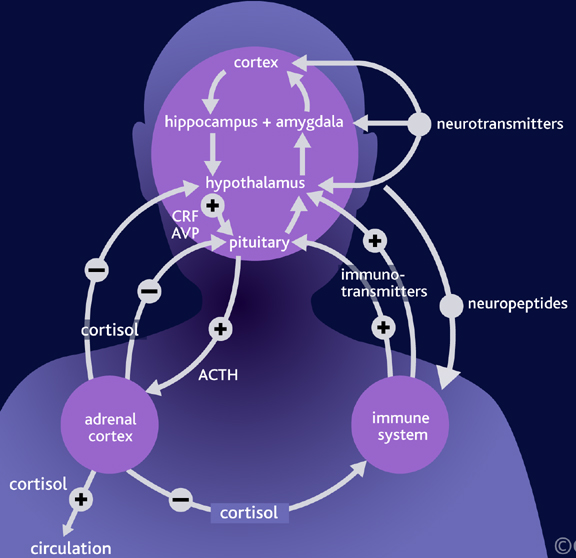
Among certain subgroups suffering from depression, it appears that this entire feedback regulatory system and thus the HPA axis is disrupted (Carrol et al. 1976; Sachar et al. 1973). This results in the hypersecretion of ACTH and cortisol with a corresponding decrease in NE; which results in NE induced depression. It was these findings which led to the development of the Dexamethasone suppression test over 25 years ago.
Via the administration of Dexamethasone (a synthetic corticosteroid) it was determined that many depressed individuals have excess cortisol, and an increased frequency of cortisol secretory episodes (Carrol et al. 1976; Sachar et al. 1973; Swann et al. 2014). Moreover, those who demonstrate excess cortisol were found to respond to NE potentiating agents, whereas those who were depressed but with normal cortisol, responded best to serotonin potentiating compounds (Van Pragg 1982).
It is also noteworthy that dexamethason nonsuppression rates are increased in mania; specifically "mixed manic" states which consist of lability, grandiosity, and lability superimposed over depression (see Swann et al. 2014). These "mixed manic" individuals also display elevated NE levels but respond poorly to lithium and show higher levels of cortisol during the depressed phase of their illness (Swann et al. 2014).
As noted, the hypothalamus may greatly influence circadian activities within the midbrain and pons, and thus the rhythmical secretion of various neurotransmitters. For example, corticotropin-releasing factor acts directly on the locus coeruleus (Valentino et al. 1983) which manufactures NE, and on the raphe thereby influencing serotonin release. These findings suggest a disturbance in circadian or rhythmical control of hypothalamic and midbrain-pontine activity can give rise to depression, or mixed mania in some individuals; women in particular.
Lateralization.
Although scant, there is some evidence which suggests that the right hypothalamus may be more heavily involved in the control of neuroendocrine functioning, particularly in females. Females are also far more likelyt to suffer from depression and from SADs. Moreover, right cerebral dysfunction can reduce NE levels in both the right and left hemisphere (Robinson 1979). Greater right hypothalamic concentration of substances such as LHRH (luteinizing hormone) has also been reported (Gerendai, 1984), which in turn is a "female" hormone involved in lactation and pregnancy.
Biologically the hypothalamus serves the body tissues by attempting to maintain internal homeostasis and by providing for the immediate discharge of tenions in an almost reflexive manner. Hence, as based on studies of lateral and medial hypothalamic functioning, it appears to act reflexively, in an almost on/off manner so as to seek or maintain the experience of pleasure and escape or avoid unpleasant, noxious conditions.
Emotions elicited by the hypothalamus are largely undirected, short-lived, being triggered reflexively and without concern or understanding regarding consequences; that is, unless chronically stressed or aroused. Nevertheless, direct contact with the real world is quite limited and almost entirely indirect as the hypothalamus is largely concerned with the internal environment of the organism. Although it receives and responds to light, it cannot "see." It has no sense of morals, danger, values, logic, etc., and cannot feel or express love or hate. Although quite powerful, hypothalamic emotions are largely undifferentiated, consisting of feelings such as pleasure, unpleasure, aversion, rage, hunger, thirst, etc.
As the hypothalamus is concerned with the internal enviornment much of it's activity occurs outside conscious-awareness. Moreover, being involved in maintaining internal homeostasis, via, for example, it's ability to reward or punish the organism with feelings of pleasure or aversion, it tends to serve what Freud (1911) has described as the pleasure principle.
THE PLEASURE PRINCIPLE
The lateral and medial nuclei exert counterbalancing influences which serve to modulate activity occurring in the other. As described by Freud (1911), the pleasure principale not only serves to maximize pleasant experiences, but acts to keep the psyche as a whole free from high levels of excitation (be they pleasurable or unpleasant).
Like the hypothalamus, the pleasure principle is present from birth and for some time thereafter the search for pleasure is manifested in an unrestricted manner and with a great deal of intensity as there are no oppositional forces (except those between the lateral and medial regions) to counter it's strivings. Indeed, higher order limbic nuclei have yet to mature.
Functionally isolated, the hypothalamus at birth has no way of reducing tension or mobilizing the organism for any form of effective action. It is helpless. When tensions associated with immediate needs (e.g. hunger or thirst) become unpleasant the only response available to the hypothalamus is to cry and make rage-like vocalizations. When satiated, the hypothalamus can only respond with a feeling state suggesting pleasure or at least quiescence. Indeed, as is well known, for the first few months of life the infants awareness largely consists of a very restricted matrix involving tactile, visceral (hunger) and kinesthetic sensations, where emotionally the infant is capable of screaming, crying, or demonstrating very rudimentary features of pleasure, i.e. an attitude of acceptance of quiescence (McGraw, 1969; Milner, 1967; Piaget, 1952; Spitz & Wolf, 1946).
It is only with the further differentiation and maturation of higher order limbic nuclei (e.g. amygdala, septal nucleus, hippocampus) that the infant begins to achieve some awareness of external reality and begins to form memories as well as differentiate and associate externally occurring events and individuals.














