Rhawn Gabriel Joseph, Ph.D.
BrainMind.com
The frontal lobes serve as the "Senior Executive" of the brain and personality, acting to process, integrate, inhibit, assimilate, and remember perceptions and impulses received from the limbic system, striatum, temporal lobes, and neocortical sensory receiving areas. The frontal lobes, therefore, subserve personality, attention, memory, speech, arousal, and motor behavior--the latter being the province of the medial and lateral frontal motor areas.
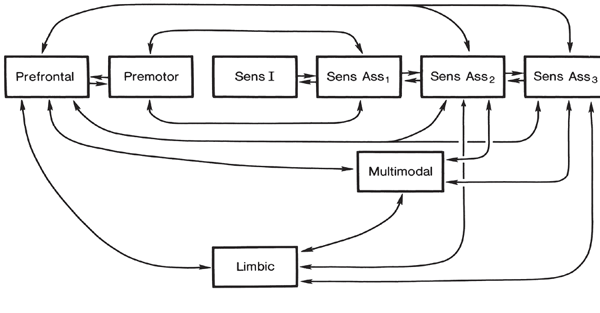
Movement and motor functioning are dependent on the functional integrity of the basal ganglia, brainstem, cerebellum, cranial nerve nuclei, the motor thalamus, spinal cord, as well as the primary, secondary and supplementary motor areas of the frontal lobes (Passingham, 2016; Schmahmann, 2016). Indeed, these areas are all interlinked and function as an integrated system in the production of movement (Mink, 2016; Mink & Thach, 2011; Parent & Hazrati 2015; Passingham, 2016).
However, as to fine motor movements including those involved in the articulation of speech, these are almost completely dependent upon the functional integrity of the primary motor areas located along the percental gyrus (area 4), and within which are represented the muscles controlling the hands, fingers, and oral laryngeal musculature.
And yet, the primary motor areas are in turn dependent upon motoric impulses which are organized in premotor and the supplementary motor cortex--the latter of which is located along the medial wall of the hemispheres. Considered from a very broad and simplistic perspective, it could be said that primary area is programmed and under the control of the secondary and supplementary motor areas as well as the "prefrontal" and other areas of the cerebrum, although neurons in the primary area also become active prior to and during movement (Passingham, 2016).
For example, Exner's writing area is in part, within areas 6 and becomes active prior to (as well as during) hand movements and appear to program hand movements, whereas the frontal eye fields (within areas 6,8,9) becomes active prior to (as well as during) eye movements and appears to program eye movements. As noted above, the primary area representing the oral-laryngeal musculature, is programmed by Broca's expressive speech area areas 45, 46 (Foerster 1936; Fox 2015; LeBlanc 1992; Petersen et al. 1988, 1989). Broca's area also becomes active prior to vocalization and during subvocalization as indicated by functional imaging.
Hence, like the sensory areas where information is generally received in the primary zones before transmission to the association areas as well as in the association areas independently of the primary areas (Zeki, 2016) motor impulses are processed and acted on in parallel (Passinghma, 2016), though in general, they generally begin their organizational journey in the supplementary motor areas (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010) and/or the cingulate and other forebrain structures (Passingham, 2016; Stephan, et al., 1999), which are then transmitted to the primary regions where they are acted upon.
Again, however, there are areas within the primary region which also become active prior to movement, though these same areas become increasing active during movement (Passingham, 2009).
Because, in general, the impulse to move originates outside the primary motor cortex, direct electrical stimulation of this region does not give rise to complex, coordinated, or purposeful movements (Penfield & Boldrey, 1937; Penfield & Jasper, 1954; Penfield & Rasmussen, 1950; Rothwell et al. 2013). Rather stimulation will only induce, for example, twitching of the lips, flexion or extension of a single finger joint, protrusion of the tongue or elevation of the palate. It is noteworthy that patients never claim to have willed these movements, which again suggests the will-to-move is initiated elsewhere.
However, if electrical stimulation is applied when the patient is attempting to move, the result is paralysis (Penfield & Jasper, 1954; Penfield & Rasmussen, 1950). Presumably the reception of impulses-to-move (which are initiated and organized elsewhere) are blocked by primary motor electrical stimulation.
Will, Speech & Mutism, Voluntary Movement, Obsessive-Compulsions, Gegenhalten, Waxy Flexibility, & the Alien Hand
The frontal neocortex is "interlocked" with the limbic system, striatum, and the primary and secondary receiving areas via converging and reciprocal connections, and receives verbal and ideational impulses transmitted from the multi-modality associational areas including the medial frontal and medial motor areas of the frontal lobes motor areas (areas 11,12,10,9,8, 6,32,33,25,24).
Medial frontal injuries produce a variety of profound symptoms including reduced speech output, mutism, severe apathy. With massive deep medial frontal dysfunction, the patient may become motionless, rigid, and completely catatonic (Hasslet 1980; Laplane et al. 1977; Luria 1980; Penfield & Jasper 1954; Penfield & Welch 1951). However, some patients may also develop "alien hand" symptomology, such that the right or left hand behaves in an independent and "alien" manner--sometimes completely opposite to what the patient intends.
Patients may seem apathetic, indifferent, and/or severely depressed. Left convexity lesions are also associated with reduced intellectual functioning, and possibly some forms of schizophrenic and obsessive-compulsive abnormalities.
THE FUNCTIONAL NEUROANATOMY OF THE SUPPLEMENTARY MOTOR AREAS
The supplementary motor area (SMA) is located along the medial walls of the hemispheres and appears to be concerned with the general problem of guiding and coordinating the movements of the extremities through space (Andres, et al., 1999; Passingham, 2016; Stephan, et al., 1999). The SMA appears to be especially concerned with movement preparation, and in directing movements toward or away from specific stimuli, and it becomes bilaterally activated during movements of the right or left half of the body.
The SMA is tightly interlinked with the striatum, anterior cingulate, amygdala, as well as the motor thalamus, and contributes axons to the corticospinal tract and makes synaptic contact with the brainstem, cerebellum, and spinal cord (Brodal, 1981; Carpenter, 2011; Schmahmann, 2016). Moreover, the SMA receives input from and transmits to the premotor and primary frontal motor areas as well as from and to the parietal lobe. Hence, this structure is situated at the cross-roads where divergent impulses and sensory impression are received and apparently acts to integrate these signals. Presumably because of its integrating role, the SMA also becomes highly active during the modification, learning and establishment of new movement programs (Brinkman & Porter, 1979; Roland et al.1980; Tanji et al. 1980), and may well act to make decisions regarding possible movements based on these signals well before a movement is initiated.
In fact, single cell recordings (Brinkman & Porter, 1979; Tanji & Kurata, 1982) and studies of blood flow studies (Orgogozo & Larsen, 1979; Shibasaki et al. 2009) and movement related evoked potentials (Ikeda et al. 1992) indicate increased activity in this area when simply imagining as well as when performing complex movements of the fingers and hands.
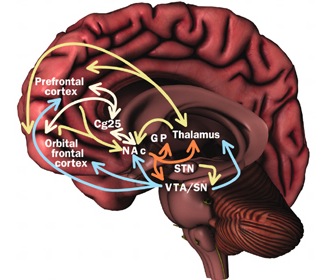
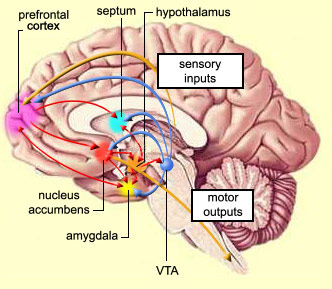
In general, however, and as based on single cell recording (vs functional imaging) activity begins in the SMA well before movements are initiated and prior to activation within the premotor and primary motor areas (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Mink, 2016; Mink & Thach, 2011). For example, when anticipating or preparing to make a movement, but prior to the actual movement, neuronal activity will first begin and then dramatically increase in the SMA, followed by activity in the premotor and then the primary motor area, and then the caudate and last of all the putamen and globus pallidus (Alexander & Crutcher, 2010; Mink, 2016; Mink & Thach, 2011).
Presumably the putamen, in conjunction with the caudate, transmits this information to the globus pallidus which in turn projects to the motor thalamus, brainstem reticular formation, as well as to the motor neocortex, thus creating a very elaborate feedback loop (Mink, 2016; Mink & Thach, 2011; Parent & Hazrati 2015) which is also influenced by the anterior cingulate and amygdala.
The SMA, however, is not concerned with fine motor functioning, as this is the province of the primary motor areas. Rather, it is concerned with coordinating gross and bilateral movements of the hands and extremities, including grasping functions (Andres et al., 1999; Passingham, 2016; Stephan et al., 1999). Electrical stimulation of the SMA has produced complex semipurposeful movements, vocalization (Penfield & Jasper, 1954), and postural synergies involving the trunk and extremities bilaterally (Van Buren & Fedio, 1976), but not fine motor movements.
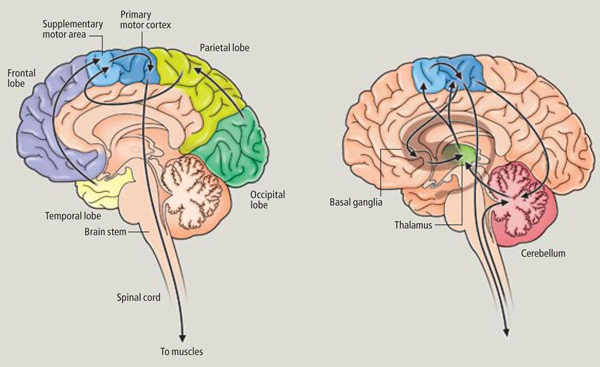
Because of its role in guiding and programming gross body movements, if this structure is damaged, the body may become stiff and movements tend to be slow and incoordinated--a condition also seen with mild lesions (Penfield & Jasper, 1954). Patients demonstrate clumsiness, severe agraphia, impairments of bimanual coordination and difficulty performing rapid or alternating movements (Brinkman, 1981; Gasquione 2009; Goldberg & Bloom 2010; Goldberg et al. 1981; McNabb et al. 1988; Penfield & Jasper, 1954; Stephan et al., 1999; Travis, 1954; Truell et al. 2015; Watson et al. 1986). Patients may walk with short steps and suffer disturbances involving posture, balance, and gait. As detailed above, with extensive and massive injuries patients may become mute (McNabb et al., 1988; Watson et al., 1986) and so stiff and unmoving that they appear to demonstrate all the classic signs of catatonia including gegenhalten and waxy flexibility.
THE SECONDARY/PREMOTOR MOTOR AREAS
The SMA is directly linked with the premotor areas (area 6), which in turn appears to be dependent on the SMA for functional programming. That is, the premotor area receives impulses to move that have been organized in the SMA, processes and integrate these signals, and then acts to program the adjacent primary motor areas with which it is intimately interconnected. Both the premotor and primary motor areas are located along the lateral and medial surface of the hemisphere. Although the premotor area does not contain giant Betz cells (which are found in area 4), it contributes almost 1/3 of the fibers of the corticospinal tract.
In addition to its interconnections with the SMA and primary motor areas (Jones et. al., 1978; Jones & Powell, 1970), the premotor area receives information directly from the primary and secondary somesthetic and visual (area 17, 18, 19) cortices (Jones & Powell, 1970; Pandya & Kuypers, 1969). It is precipally concerned with the guidance and refinement of movement via the assimilation of sensory information provided by the sensory areas (Godschalk et al. 1981; Porter 2010) and interacts with the basal ganglia, motor thalamus, and SMA so as to achieve these goals (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Mink & Thach, 2011; Parent & Hazrati 2015).
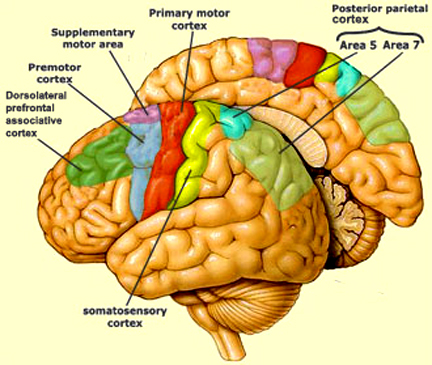
As noted, the SMA becomes activated prior to the premotor area, which becomes activate prior to the primary motor area. Hence, whereas neurons in the primary motor region become active during movement, excitation in the premotor cortex precedes cellular activation of the primary region (Weinrich et al. 1984). Moreover, cells in the premotor cortex become activated before movements are even initiated, whereas electrical stimulation of this area induces the illusion of a impending movement (Penfield, 1938). These and other findings suggest that the premotor area may be modulating and exerting controlling and integrative influences on impulses which are to be transmitted to the primary region for expression.
Indeed, the premotor area appears to be highly involved in the programming of various gross and fine motor activities, and becomes highly active during the learning of new motor programs (Passingham, 2016; Porter 2010; Roland et al. 1981). Moreover, electrical stimulation elicits complex patterned movement sequences as well as stereotyped and gross motor responses such as head turning or torsion of the body (Fulton, 1934; Passingham, 1981, 2009).
Unlike the primary area, damage limited to the pre-motor cortex does not result in paralysis but disrupts fine motor functioning and dexterity, including simple activities such as finger tapping (Luria, 1980). With extensive damage fine motor skills are completely lost and phenomena such as the grasp reflex are elicited (Brodal, 1981); i.e. if the patient's hand is stimulated it will invulantarily clasp shut.
THE SMA & MEDIAL FRONTAL LOBES
The supplementary motor area is located along the medial walls of the hemispheres but has no clear cut anatomical boundaries. It's anterior portion abuts the medial overlap of the primary area and it extends downward along the medial wall where it meets the anterior cingulate gyrus (area 24). The supplementary motor area (SMA) contains a crude neuronal representation of the body (Goldberg, 1985), and receives axonal projections from the primary and association somatosenory areas (Jones & Powell, 1969; Pandya & Vignolo, 1971) and shares rich interconnections with primary motor cortex, the anterior cingulate (Devinksy et al. 2015; Jones, et al. , 1978), and the basal ganglia (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Mink, 2016) as well as the right and left frontal speech areas. The SMA, in fact, becomes activated during inner speech and subvocal articulation (Paulesu, et al., 2009; Demonet, et al., 1994), and when engaged in speech processing (Peterson et al., 1988) and even when reading (Bookheimer, et al., 2015; Price, et al., 1996)--information which is transmitted to the lateral speech areas.
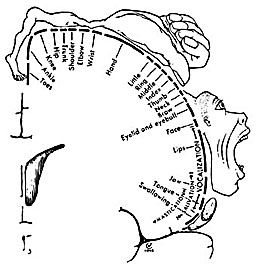
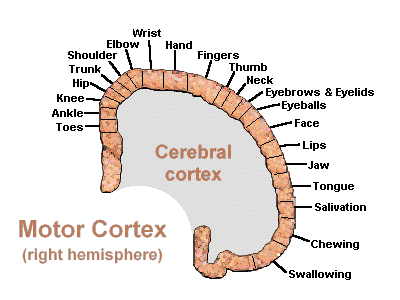
The SMA is also concerned with the general problem of guiding and moving the extremities through space including bimanual coordination (Andes et al., 1999; Passingham, 2016; Stephan et al., 1999) and both the right and left SMA become active during movements (Passingham, 2016). Moreover, the SMA appears to contain what Rizzolatti et al., 1996) refer to as "mirror" neurons. According to Rizzollati (1998, p. 528) "mirror neurons appear to form a system that allows individuals to recognize motor actions made by others by matching them with an internal motor copy," thus enabling them to perform and mimic the actions of others.
Electrical stimulation of the SMA has produced complex semipurposeful movements, vocalization (Penfield & Jasper, 1954), and postural synergies involving the trunk and extremities bilaterally (Van Buren & Fedio, 1976). Moreover, single cell recordings (Brinkman & Porter, 1979; Tanji & Kurata, 1982) and studies of blood flow studies (Orgogozo & Larsen, 1979; Shibasaki et al. 2009), movement related evoked potentials (Ikeda et al. 1992) and functional imaging studies (Passingham, 2016) indicate increased activity in this area bilaterally while performing and even imagining complex movements of the fingers and hands. This region also becomes highly active during the modification, learning and establishment of new movement programs (Brinkman & Porter, 1979; Roland et al.1980; Rizzollati, 1998; Tanji et al. 1980).
The SMA appears to organize impulse to move, to the premotor and primary motor areas where these impulses are acted on. That is, although neurons in these different areas often become active simultaneously (Passingham, 2016), the SMA appears to organize the primary and premotor areas. Hence, activity begins in the SMA well before movements are initiated and prior to activation within the majority of cells in the premotor and primary motor areas (Alexander & Crutcher, 2010; Crutcher & Alexander, 2010; Mink & Thach, 2011). SMA activity is triggered just by thinking about moving (Passingham, 2016)--activity which spreads to the association and primary motor areas as well as the striatum.
The SMA, however, appears to be concerned with gross body movements, as well as vocalization, and is concerned with the general problem of guiding and coordinating the movement of the extremities through space (Andres, et al., 1999; Passingham, 2016; Rizzollati, 1998; Stephan, 1999). Electrical stimulation of the medial frontal lobes will produce complex semipurposeful movements, vocalizations (Penfield and Jasper 1954), and postural synergies involving the trunk and extremities bilaterally (Van Buren and Fedio 1976). Moreover, single cell recordings (Brinkman and Porter 1979; Tanji and Kurata 1982) blood flow studies (Orgogozo and Larsen 1979; Shibasaki et al. 2009), movement related evoked potentials (Ikeda et al. 1992) and functional imaging studies (Passingham, 2016) indicate increased activity in the SMA and medial tissues while performing and even imagining complex movements of the fingers and hands.
Unlike injuries to the primary and association motor tissues (see below), paralysis or paresis does not result with medial frontal damage, although the body may become slow, stiff, and uncoordinated (Luria, 1980; Stephan et al., 1999). Gegehalten and waxy flexibility may also be demonstrated. Indeed, with massive lesions patients may become so stiff and unmoving that they appear catatonic and lose even the will to speak (Hasslet 1980; Joseph, 1999a; Laplane et al. 1977; Luria 1980; McNabb et al. 1988; Penfield and Jasper 1954; Penfield and Welch 1951; Watson et al. 1986).
With slowly growing medial frontal tumors, patients become increasingly apathetic, unresponsive, uncommunicative, and disinterested, and may be misdiagnosed as depressed.
DISTURBANCES OF WILL
The medial frontal lobes are directly associated with the limbic system and integrate, assimilate, and shunt emotional and motor impulses to the neocortex where they may be incorporated into speech and ongoing cognitive and motoric activity. Indeed, the limbic system transmits streams of affective stimuli to the frontal lobe via the medial frontal areas.
Hence, lesions in this vicinity may severe and disconnect the fiber pathways which enable emotional and motivational impulses arising in the limbic system to become integrated and expressed by the frontal neocortex. Due to the loss of motivational input, ideas and thoughts are no longer generated, and the "Will" to speak or to initiate or complete a voluntary movement may become completely attenuated and temporarily abolished (Hasslet 1980; Laplane et al. 1977; Luria 1980; Penfield and Jasper 1954; Penfield and Welch 1951).
The anterior cingulate is contiguous with the medial frontal lobe and often becomes active prior to and while engaging in various movements (Passingham, 2016; Stephan et al., 1999) and while speaking (Passingham, 2016) and reading (Peterson et al., 1988). Likewise, injuries affecting the anterior cingulate can also induce a paralysis of will and mutism and reduced movement (Barris and Schuman 1953; Devinksy et al. 2015; Kennard 1955; Laplane et al. 1981; Smith 1944).
Damage or surgical cingulate removal has resulted in generalized emotional dampening and unresponsiveness, hypokinesis, severe social indifference and apathy, a decreased awareness of the environment, and a failure to make avoidance responses (Barris and Schuman 1953; Kennard 1955; Laplane et al. 1981; Pechtel et al. 1958; Smith 1944; Tow and Whitty 1953)--a function, in part, of the role of the anterior cingulate in the perception of painful and noxious stimuli (Casey et al., 1994; Coghill et al., 1994).
Barris and Schuman (1953) describe one patient who rapidly developed akinetic mutism and indifference following bilateral strokes to the anterior cingulate and portions of the SMA. After returning home from work he sat down on the sofa, held up a newspaper as if reading, i.e. compulsive utilization behavior (see below), became incontinent of urine (about which he was totally unconcerned), and completely unresponsive and unable to reply to questions.
Upon recovery many patients may later remark they had completely lost the will to speak, that thoughts did not enter their head, and that they were unable to think or generate ideas, and instead experienced a motivational-ideational void and complete emptiness (Brutkowski 1965; Hasslet 1980; Laplane et al. 1977; Luria 1980; Mishkin 1964). In part, this appears to be due to the destruction of those neurons which normally receive emotional and motivational input from the limbic system; impulses which may then be shunted to the orbital, lateral, or motor areas of the frontal lobes. In this regard, the symptoms produced also tend to vary depending on if orbital vs lateral/motor areas are affected or both.
ORBITAL-MEDIAL EMOTIONAL UNRESPONSIVENESS
The medial frontal lobes arch dorsally to become the superior frontal lobe, and ventrally to become the orbital frontal lobes. With massive orbital-medial injuries or lobotomy, patients may initially display severe reductions in activity, arousal, and emotional, motivational functioning, and appear apathetic and indifferent to loud noises or threats (Butter et al. 1970; Freeman and Watts 1942 1943; Stuss and Benson 1986).
With massive orbital-medial destruction reduced responsiveness persists and patients may sit nearly motionless, making little or no attempt to communicate as if mute. Social-emotional responsiveness seems abolished (Kling and Steklis 1976) and patients may appear profoundly apathetic, indifferent, and depressed (Grafman et al. 1986; Stuss and Benson 1986). In fact, as the medial walls and SMA programs the body and extremities for gross movements, if the lesion encroaches on the SMA, patients become catatonic, mute, and remain in odd, uncomfortable postures for long time periods.
GEGENHALTEN AND WAXY FLEXIBILITY
Gegenhalthen (counterpull), i.e. involuntary resistance to movement of the extemities, appears to be exclusively associated with medial frontal lobe/SMA abnormalities (Feeman and Watts 1942; Joseph 1996; Rose 1950; Travis 1955). If a physician lifts an affected arm, it will stiffen and become increasingly rigid as pressure to move it increases. Although aware, the patient cannot decrease the resistance, and the arm not only becomes stiff, but frozen in place. In some cases, patients who are locked into one position, when forced into another, may spring back to the former frozen posture.
However, it is also not uncommon for patients to move exceedingly slowly, such that although they maintain a particular posture for long time periods, the affected limb may slowly return to a resting posture--a condition referred to as "waxy flexibility." Patient's can be molded into a variety of positions and postures which they briefly maintain. However, within minutes or hours, the arm or body slowly "melts" and returns to a normal resting position.
CATATONIA
Since abnormalities of the medial frontal lobes are typically associated not only with Gegenhalten, and waxy flexibility but mutism, apathy, and loss of "Will," affected individual, therefore, stereotypically display the classic signs of catatonia, or, in the less extreme, the rigidity associated with Parkinson's diease.
For example, Freeman and Watts (1942) described one individual who developed gegenhalten, waxy flexibility, mutism, and catatonia after a gunshot wound that passed completely through the frontal lobes. "The patient layed in a catatonic-like stupor for two months, always upon one side with slightly flexed arms and legs, never changing his uncomfortable position; if he were rolled into some other position, he would quickly get back into his former one. He did not obey commands, but if food and drink were given to him, he swallowed them naturally. He was incontinent, made no complaints, gazed steadily forward and showed no interest in anything. He could not be persuaded to talk, and then suddenly he would answer quite correctly about his personal affairs and go back to mutism. From time to time he showed a peculiar explosive laugh, especially when his untidiness was mentioned." Incredibly, the patient "was eventually returned to active duty" (Freeman and Watts 1942, pp 46-47).
In another case, a 42 year old male with no previous psychiatric history, developed gegenhalten, mutism, and catatonia after a beating and suffering frontal and midline subdural hematomas (which later required the drilling of burr holes for evacuation). He resisted the efforts of others to move him, and would sit motionless and unresponsive for hours in odd and uncomfortable positions. Interestingly, the patient's symptoms seemed to wax and wane such that he demonstrated some periods of seeming normality.
As discussed in chapter 16, Parkinson's disease, although associated with reduced dopamine in the putamen and other striatal abnormalities, is also linked to SMA abnormalities--as first proposed in the second edition of this text--such that the lateral convexity can no longer receive internally generated signals and can no longer respond to internally generated cues. Indeed, as based on functional imaging, it has been recently found that patients with Parkinson's disease display reduced activity in the SMA, whereas the lateral motor area appears functionally normal (Passingham, 2016).
COMPULSIONS AND THE MEDIAL FRONTAL LOBE
FORCED GRASPING AND COMPULSIVE UTILIZATION
As noted, the medial lobe receives and projects to wide areas of the neocortex and is involved in movement initiation and programming. Hence, with extensive injuries the patient may cease to move or speak. However, with less extensive injuries the ability to voluntarily control one's movements may be compromised, such that the arms and hands become freed of internal control. That is, they may respond involuntarily to external stimuli by reaching out, touching, grasping, or even utilizing whatever they touch (Denny-Brown, 1958; Gasquoine 2009; Goldberg & Bloom 2010; Lhermitte, 1983; Travis, 1955). Presumably this is due to a loss of internal motivational controls (i.e. disconnection), and is partially a release phenomenon such that patients appear reflexively or magnetically directed solely by external stimuli which trigger involuntary motor reactions.
For example, with extensive destruction and/or as the lesion extends mesially, the mere visual presence of an object near the hand triggers groping movements as well as grasping. Similarly, if an object touches the palm of the hand, the patient grips it involuntarily and cannot let go. Denny-Brown (1958) has referred to this as "magnetic apraxia" and "complusive exploration". He notes that touching the hand will elicit orienting movements to bring the object into the palm. Once it is grasped the patient cannot release his grip.
This aberration is often accompanied by gegenhalten. Immediately prior to actually grasping the object the entire arm will stiffen and one is met with resistance if an attempt is made to move the extremity. If a patient were to attempt to write, the hand stiffens and becomes seemingly "stuck" to the paper. If they try to walk, their feet seem to stick to the floor as if glued, and steps are made with great difficulty (Denny-Brown, 1958).
Denny-Brown (1958) believed that magnetic groping was due in part to a perseveration of contractual reactions (as well as parietal lobe disinhibitory release), for so long as stimulation is applied to the skin, the deficit, including gegenhalten, not only persists but becomes more intense.
This abnormality is also triggered visually. As if stimulus bound, the patients eyes and head may compulsively following moving objects. If an object is brought near the face they may compulsively reach out and take it, and/or if near the lips may attempt to mouth or suck it. Such behavior is completely involuntary and will occur even if the patient "Willfully" attempts to oppose it.
Presumably this is due to a disconnection involving the medial and the lateral frontal lobes. That is, since the medial area does not normally respond to visual input whereas the lateral area does (Passingham, 2016), lesions to the medial area may result in the disinhibition of the lateral motor areas which compulsively respond to visual stimuli.
Patients may involuntarily pick up and compulsively utilize various tools, utensils and other objects, and may compulsively reach out and take the examiner's pen or swipe their glasses from their face and put them on. One patient put three pair of spectacles on and wore them simultaneously (Lhermitte 1983).
If a hammer or glass of water is placed on the testing table the patient may involuntarily begin to hammer, or they may drink from the glass although they are not thirsty and when told to refrain from these behaviors (Denny-Brown 1958; Gasquoine 2009; Goldberg and Bloom 2010; Lhermitte 1983; McNabb et al. 1988; Travis 1955). Hence, damage is this area can cause patients to respond and use whatever objects or tools may be close even when they understand that they are not to do so (Lhermitte, 1983).In fact, these involuntary movements may become so purposeful and complex and uncontrolled that this condition has also been referred to as the "alien hand" (Feeman and Watts 1942; Gasquoine 2009; Goldberg and Bloom 2010; Joseph 1988ab; Travis 1955).
THE ALIEN HAND
In rare instances, this compulsive groping and utilization behavior can become confined to one limb and involve complex and seemingly purposeful actions (Goldberg 2013; Goldberg & Bloom 2010). This may occur in cases where the lesion has predominantly destroyed either the right or left SMA and medial portion of the hemisphere as well as the anterior corpus callosum (McNabb et al., 1988; Gasquoine 2009; Goldberg & Bloom 2010; Goldberg et al., 1981), such that the right and left frontal lobe became partially or fully disconnected. As noted, the medial frontal lobes and SMA serve to coordinate bimanual activity , and both the right and left SMA become active during a unilateral movement (Andes et al., 1999; Passingham, 2016; Stephan, et al., 1999). Hence, when injured, there is a disconnection.
McNabb et al. (1988, pp. 219, 221) describe one woman with extensive damage involving the medial left frontal lobe and anterior corpus callosum, whose right "hand showed an uncontrollable tendency to reach out and take hold of objects and then be unable to relese them. At times the right hand interfered with tasks being performed by the left hand, and she attempted to restrain it by wedging it between her legs or by holding or slapping it with her left hand. The patient would repeatedly express astonishment at these actions." A second patient frequently experienced similar difficulties. When "attempting to write with her left hand the right hand would reach over and attempt to take the pencil. The left hand would respond by grasping the right hand to restrain it" (p. 221).
Similar problems, however, have also plagued some patients following complete (surgical) destruction of the corpus callosum (Joseph, 1988ab). In many of these instances, however, the independent "alien" behaviors demonstrated were often purposeful, intentional, complex and obviously directed by an awareness maintained by the disconnected right hemisphere. For example, one patient's left hand would not allow him to smoke and would pluck lit cigarettes from his mouth, whereas another patients left hand (right brain) preferred different foods and even television shows and would interfere with the choices made by the right hand (left hemisphere). In one instance the patient was pulled to the television and the left hand changed the channel.


However, in some instances, patients may display alien movements in both hands (Gasquoine 2009; Goldberg & Bloom 2010) as well as alien vocalization of thoughts. For example, a patient described by Gasquoine (2009) had a propensity to reach out and touch female breasts, as well as novel objects and persons with in reach. He reported this caused him great embarrassment and that he would typically attempt to take hold of his right with his left hand or voluntarily grasping objects, such as his lap tray, so that he would not spontaneously reach out and grab someone. Moreover, although he had difficulty initiating speech, early in his illness he would spontaneously vocalize his thoughts.
Internal Utilization.
As the locus of the lesion becomes even more inferior and posterior, encroaching on the orbital-temporal (amygdaloid) area, external utilization is replaced by attempts at internal (oral) utilization. Instead of utilizing an object by motorically interacting with it, the individual orally interacts and attempts to consume the object. Everything that is seen and touched is placed indiscriminantly in the mouth and orally "explored."
This latter disturbance, extreme orality, has been referred to as "psychic blindness" and the Kluver-Bucy syndrome, and is a consequence of amygdaloid destruction--a nucleus with massive interconnections with the orbital/inferior medial area.
OBSESSIVE COMPULSIONS & THE MEDIAL-ORBITAL FRONTAL LOBES
Compulsive utilization, and forced groping and grasping, are obviously compulsive in nature, and the patient is unable to control these motor behaviors and independent actions of the extremities. In some respects, these disorders are reminiscent of obsessive-compulsive disorders, which are also associated with frontal lobe injury, especially of the orbital-medial frontal lobes and the striatum. The medial frontal lobes, in fact, are a major motor center involved in programming the hands and body. The medial frontal lobe, as the name indicates, is located within the medial portions of the frontal lobes, and extends dorsally to become the superior frontal lobe, and ventrally becoming the orbital frontal lobes--tissues which are intimately interconnected. Likewise, as the orbital tissues extend laterally, they become the inferior lateral frontal lobes.
In consequence, damage to the ventral regions often simultaneously disrupt the orbital, medial, as well as the inferior lateral frontal lobes, and can produce a unique constellation of compulsive, perseverative symptomology, including "forced thinking" (Iversen and Mishkin 1970; Milner 1963, 1964, 1971; Penfield and Jasper 1954; Stuss and Benson, 1986; Ward 1988) and uncontrolled obsessive compulsions (Baxter et al. 2013; Rappoport 2011; Rauch et al. 1994). That is, a patient may suffer from a difficulty suppressing or inhibiting previous thoughts or behaviors, which then occur again and again (Malloy et al. 2009; Passingham 2009; Stuss and Benson 1986).
PERSEVERATIONS
Orbital injuries and orbital lobotomies are associated with high rates of behavioral perseveration and failures to maintain correct responses due to perseverative intrusion (Malloy et al. 2009; Mishkin 1964; Passingham 2009; Stuss and Benson 1986) and a loss of inhibitory control (Fuster 2016; Goldman-Rakic 2013; Mishkin 1964). They repeat previous responses or provide the same answers even when the context is no longer appropriate or when the response is punished and even if the individual realizes his or her responses are incorrect (Butter 1969; Iverson and Mishkin 1970; Jones and Mishkin 1972; Kolb et al. 1974; Luria 1980; Passingham 2009; Stuss and Benson 1986).
If a patient with extensive and severe orbital frontal injuries is asked to "draw three circles, a square, then a cross", they may instead draw five circles, thus repeating their first three responses. A patient described by Luria (1980) was asked to draw a pair of spectacles, and did so correctly. When asked to draw a watch, he again drew the spectacles. When the mistake was pointed out, he again drew a pair of spectacles but this time drew a watch in the center of one of the lens.
Perseverative tendencies can influence all aspects of behavior including speech, depending on the extent and laterality of the lesion. For example, perseveration of speech (of which there are several subtypes), are often associated with inferior lateral injuries that disrupt Broca's area (Peitro and Rigdrodsky 1986; Sandson and Albert 2013) or the right frontal emotional-melodic speech area (Joseph, 1986a, 1988a). With left frontal injuries patients may be capable of only repeating simple words over and over. Left frontal injuries are also associated with perseverative errors on the Wisconsin Card Sort even when the orbital area is seemingly spared (Drewe 1974; Milner 1964, 1971). By contrast, those with right frontal injuries may perseveratively repeat complex statements or phrases.
Although perseverations are associated with difficulties inhibiting previous responses, perseveration secondary to injuries localized to the orbital frontal lobes are not necessarily associated with impulsivity. A patient who is impulsive may do or say whatever occurs to him, whereas a patient who perseverates has difficulty inhibiting a previous response and engaging in new behaviors. Thus, across tasks requiring delayed responding, orbital lesions have little effect on performance (Brutkowski et al. 1963; Rosenkilde 1979). Rather, with orbital (inferior lateral/medial) lesions, the problem is shifting sets and inhibiting the recurrence of a previous response when a new action is called for. Once a behavior occurs it persists and contaminates the performance of unrelated actions. Thus rather than disinhibited, they suffer from a compulsion to repeat.
OBSESSIVE-COMPULSIONS, ORBITAL-MEDIAL FRONTAL LOBES & STRIATUM
Perseverations have an obvious compulsive quality, and there is now considerable evidence that frontal lobe dysfunction may give rise to obsessive-compulsive (Ob-C) disorders (Baxter et al. 1988; Flor-Henry 1983; Flor-Henry et al. 1979; Joseph, 1999a; Malloy 2013; Miller 2010; Rappoport 2011; Rauch et al. 1994). It is noteworthy that Ob-C disturbances are also frequently accompanied by strong feelings of depression and anxiety (Goodwin & Guze, 1979) --affective states linked to alterations in the functional integrity of the anterior regions of the brain.
Moreover, just as perseverations of thought, speech, and behavior are associated with frontal lobe abnormalities, in some cases of frontal injuries, these perseverations may assume obsessive and compulsive dimensions and patients may experience unwanted, recurrent, perseverative ideas, or compulsions to repetitively perform certain acts, e.g. hand washing. Indeed, obsessions are perseverative in nature, often involving intrusive recurring thoughts, feelings, or impulses to perform certain actions. Motorically obsessive compulsions may involve repetitive, stereotyped acts including the perseverative manipulation and touching of objects.
As noted, orbital/medial damage, particularly, if the SMA is negatively impacted, can give rise to involuntary and compulsive motor behaviors involving the hands; and just as perseverations are associated with inferior/orbital frontal lobe damage, obsessive-compulsive abnormalities are associated with abnormalities of the left frontal (Flor-Henry 1983; Flor-Henry et al. 1979; Malloy 2013; Miller 2010), right frontal, and orbital/medial frontal areas (Baxter et al. 2013; Malloy et al. 2009; Rappoport 2011; Rauch et al. 1994).
Moreover, abnormal or electrical activation of the medial-orbital frontal lobes has been reported to trigger recurrent and intrusive ideational activity ("forced thinking"), as well as compulsive urges to perform aberrant actions, e.g. shouting (Penfield and Jasper 1954; Ward 1988), e.g. Tourretts syndrome. Likewise, perseverations of speech, thought, and behavior have been repeatedly associated with inferior medial-orbital injuries (Passingham 2009; Penfield and Jasper 1954; Ward 1988), including those which extend to and negatively impact the anterior cingulate (Flor-Henry 1983; Pardo et al. 2011), and the caudate nucleus (Baxter et al. 1992; Rauch et al. 1994).
Neuroimaging studies have consistently implicated the orbital frontal lobes, and orbital frontal-striatal pathways including the anterior cingulate, in the genesis of obsessive compulsions. As based on MRI, structural abnormalities have been noted in the cingulate (Garber et al. 1989), whereas PET studies show increased orbital (Baxter et al. 2013; Nordahl et al. 1989) and medial-orbital (Sawle et al. 2011) activity or glucose uptake. Conversely following pharmacological treatment, orbital (Benkelfat et al. 2010; Saxena et al. 1998; Swedo et al. 1992) and anterior cingulate activity (Baxter et al. 1992; Perani et al. 2015) decrease. By contrast, increased activity in the orbital and cingulate cortex have been documented among patients whose symptoms were actively provoked (Breiter et al. 1996; McGuire et al. 1994; Rauch et al. 1994).
As noted, the oribital ventral medial frontal lobes are intimately associated with the striatum and interact in regard to movement, emotional, and ideational activity. Hence, not only are the orbital frontal lobes and striatum implicated in the genesis of obsessive compulsions, but as based on CT-scan (Luxenberg et al. 1988) and MRI, the caudate (Jenike et al. 1996; Robinson et al. 2015) and putamen (Rosenburg et al. 2016) has been found to be smaller as compared to control subjects.
Likewise, studies employing SPECT demonstrate deceased activity within the caudate, the right caudate in particular (Lucey et al. 2016; Rubin et al. 1992). However, yet other MRI studies show that the right caudate to be greater in volume (Scarone et al. 1992), or as based on PET scan, to demonstrate greater activity and glucose uptake (Baxter et al. 2013).
Presumably, what appears to be conflicting findings as to the nature of the pathology may be a function of age, sex, symptom severity, depression, or medication. For example, following treatment with clomipramine (Benkelfat et al. 2010), fluoxene (Baxter et al. 1992) or paroxetine (Saxena et al. 1998) a decrease in putamen or caudate glucose metabolism have been reported. Conversely, increases in striatal activity have been reported among patients who are actively symptomatic (McGuire et al. 1994; Rauch et al. 1994).
As detailed above, medial frontal injuries are associated with forced groping, grasping, magntic apraxia and compulsive utilization behavior. Orbital injuries produce perseverative motor disturbances. Likewise, the striatum is concerned with the modulation of body movement. Hence, in contrast to lateral frontal-striatal disturbances, it appears that abnormalities involving the orbital/medial frontal lobes and the striatum, may well explain why obsessive compulsive disorders often involve the hands, including perseverative manipulation and touching of objects or the repetition of well learned motor tasks, such as hand washing. As these frontal cortices are interlinked and exchange information through varying pathways, if abnormally activated, or if specific pathways are destroyed, well learned motor activities (such as hand washing), and powerful thoughts and feelings, may come to be disinhibited, cannot be extinguished, and may repeatedly intrude upon and disrupt ongoing behavior, with each repetition commanding a repeat performance.
Hence, given that it is via the frontal lobes that thoughts and emotions come to be integrated and infused with emotion, and/or inhibited and suppressed, and as the SMA contains neurons which can "mirror" mimic and reproduce observed behavior (Rizzollati, 2014) it thus appears that with caudate/orbital/medial dysfunction this results in an inability to extinguish certain thoughts and actions, even in the absence of an underlying emotional problem, and that a behavior once performed is "mirrored" by being performed again and again in a perseverative fashion. Hence, the individual may perseverate on a single thought or action, which in turn may interfere with their capabiliy to engage in alternative modes of response such that they instead continually perform, mirror, and thus perseverate in thought, speech, and behavior previously occurring behaviors, particularly those involving the hands.












