Rhawn Gabriel Joseph, Ph.D.
BrainMind.com
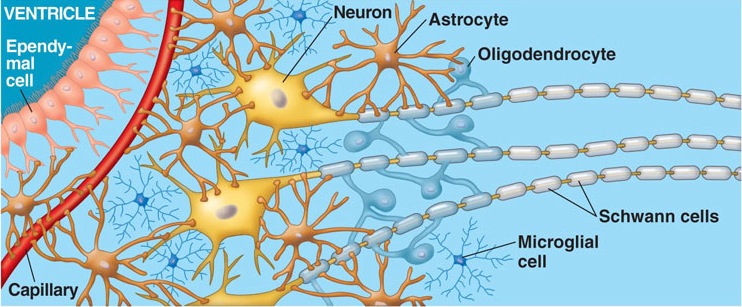
It is well established that the hippocampus and amygdala play important roles in memory functioning, and the storage and recall of visual, emotional, and linguistic experiences.
NEURAL NETWORKS
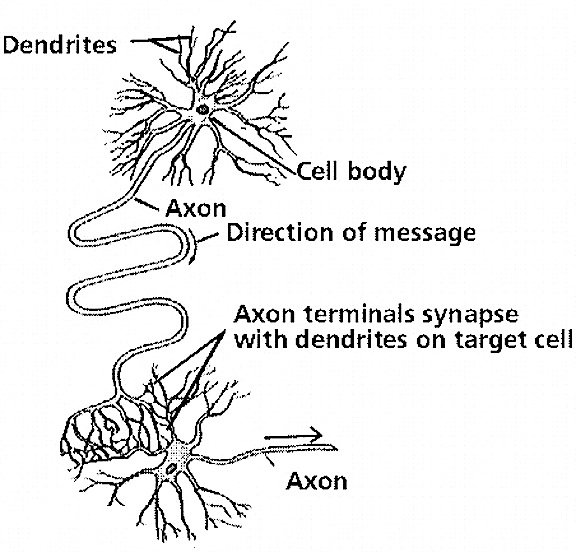
Almost a century ago it was proposed that learning was paralleled by physical changes in neuronal structure and function, as well as the establishment of new neuronal connections where before there had been none (Cajal, 1911; Tanzi, 1893, cited by Cajal 1954). Moreover, Cajal (1911) proposed that long term memories were stored via the establishment of these new synaptic links.
A quarter of a century later, Hilgard and Marquis (1940) and Hebb (1949), basing their hypothesis in part on the neuroanatomical work of Lorente de No (1938) and Cajal (1911), proposed that perceptual activity continues in neurons even after the cessation of stimulus input, thereby inducing structural alterations, which in turn makes learning and memory possible. Hebb (1949) also proposed that different neurons are linked together via this activity so as to form reverberating neural circuits that can fire with minimal stimulation.
Use it, or Lose it.
According to Hebb (1949, p. 62), "when an axon of cell A is near enough to excite cell B or repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells." Hebb (1940) also presumed that memories are represented via a specific pattern of activity across a network of neurons; a consequence of permanent reverberating structural changes. Behaviorally the establishment of these plastic, reverberating neural networks is represented by increases in learning efficiency and enlargement of the memory store.
In addition, like Cajal (1911), Hebb (1949) proposed that axonal-dendritic interconnections are likely to become more extensive in correspondence with the learning of events which are stored in long term memory. He also postulated that memory storage may well take place in the same cortical regions where the information is processed, such that those cells first involved continue to reverberate creating these structural changes, thus storing the memory as represented by these physical changes. Hence, different memories can be stored in different regions of the brain.
As will be detailed below, the theorizing of Hebb, Cajal, and others has since received considerable experimental support. In fact, because there are so many available synapses, cognitive complexity and memory capacity is potentially unlimited. Since the neocortex alone contains anywhere from 1014 to 1017 synapses, there are more than enough for all possible information storage requirements. Synapses make very low energy demands, so the system is energy efficient and requires little to maintain the memory stores. Therefore, as the number of neurons that come to be linked via perceptual and cognitive activity increases, so does complexity in mental functioning which in turn is maintained via a vast system of interconnected memories and semi-independent neural networks (Buchel et al., 2013; Miltner et al., 2013; Quartz & Sejnowski, 2011).
Neural networks also allow for the addition of new, albeit associated memories. Through correlated activity involving the same synapses, a new memory can be added to a neural network and/or the circuit can be modified so as to accommodate new learning. Moreover, a new set of activated neurons can be added to the network so long as their activity and interconnections become linked to the previously established pattern of excitation associated with a specific neural network (Buchel et al., 2013; Enbert & Bonhoeffer, 2013; Miltner et al., 2013; Quartz & Sejnowski, 2011; Xu et al., 2008). Behaviorally this is expressed as associative learning and even classical conditioning.
DENDRITIC LEARNING AND SYNAPTIC NEURAL NETWORKS
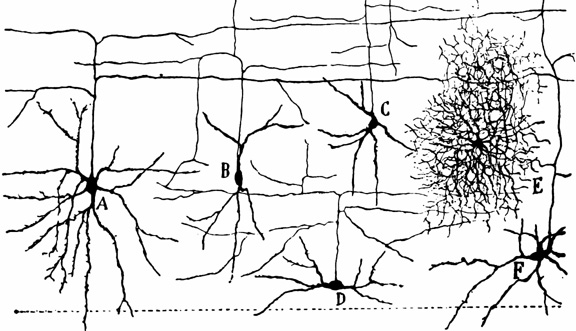
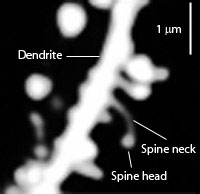
A single dendrite may receive input from hundreds of axons, each of which may be concerned with different perceptual, emotional, behavioral, or cognitive functions. For example, dendrite D (via its dendritic spines) might receive input from axons 1 through 99. However, dendrite D and axons 1-50 may belong to one neural network, whereas dendrite D and axons 51- 99 might belong to another. This is made possible as each dendrite consists of numerous dendritic spines, each with their own synaptic surface, and which grow or bloom in response to new experiences. Moreover, depending on the variables involved, axon 50 could become part of both dendritic circuits.
Learning, however, is also paralleled by synaptic growth and the development of new dendritic spines (Engert & Bonhoeffer, 2013)--alterations which likely result in the establishment of not only new synaptic connections but the creation of additional neural circuits and associated networks of neurons. In this regard, we can also divide dendrite D, into D-1 through D-100, with D-1-25 and D-50-75 maintaining synaptic connections with axons 1-5, axons 11-14 and axons 37-44, and with D-12-24, and D-72-74, and D-92-99, sharing connections with Axon 76, and D-44 maintaining a connections only with axon 49, and so on.
Presumably, due to the creation of these neural circuits widepread and complex associated memories can be formed, and due to the development of synaptic strength (which links these synapses), complex actions can be initiated in an effortless and routine fashion due to the strength of the connections which maintain the pattern (Hebb, 1949). Hence, these neural circuits can be associative, such that a variety of memories are linked as a whole and in parallel and they may also be temporal-sequential, such that one neuron of synapse can predict or trigger the next step in the behavioral sequence by activating the next neuron and so on. Based on Hebb's theory it could be predicted that memories can be activated by a variety of simple and even fragmentary cues, each of which can trigger activation of a portion or the entire circuit.
NEUROPLASTICITY AND LEARNING
Over the course of the last twenty five years these theories have found considerable empirical support (Barnes, 1979; Buchel et al., 2013; Davies, et al. 1989; Diamond, 1985; Engert & Bonhoefferm 2013; Gustafsson & Wigstrom, 2012; Lynch, 1986; Miltner et al., 2013; Quartz & Sejnowski, 2011; Rosenzweig, 1971; Singer, 2010; Stevens, 1989; Xu et al., 2008).
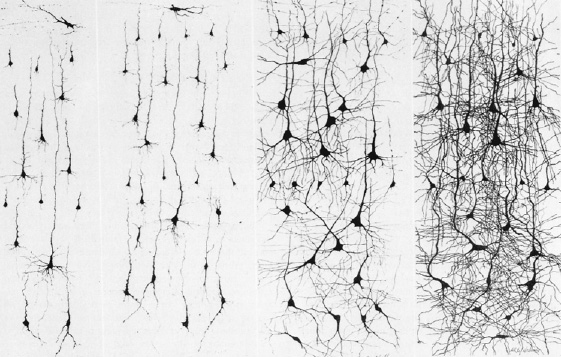
For example, when animals are reared in a complex versus simplified environment, not only are they found to have superior learning and memory capabilities (Joseph, 1979; Joseph & Gallagher 1980), but the cortex becomes more complex, with increases in synaptic density and post-synaptic thickness (Diamond, 1985, 2012; Greenough & Chang 1985; Greenough & Black, 1992; Rosenzweig,1971). As such, even neurological and perceptual functioning is altered (Casagrande & Joseph, 1978, 1980; Joseph & Casagrande, 1980), as is learning and memory (Joseph, 1979; Joseph & Gallagher, 1980).
In fact, as demonstrated in this laboratory, if the dorsal hippocampus is removed on one side and an animal is reared in an enriched environment, the neocortex on the side of the lesion fails to demonstrate any of the positive effects of enrichments, whereas the neocortex on the opposite side of the removal displays increased dentritic growth as represented by increased neocortical thickness.
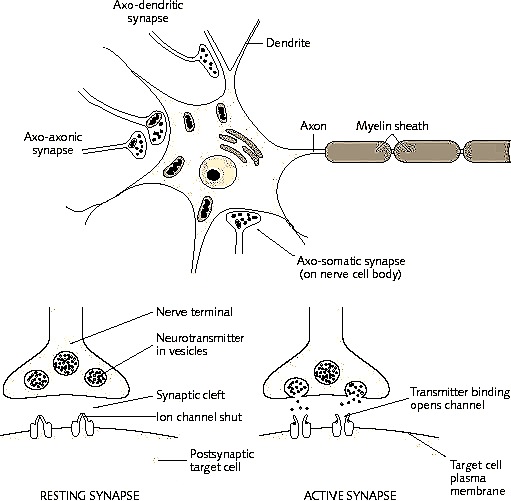
Moreover, these synaptic changes have been found to occur in conjunction with the development of long-term synaptic potentiation in dendrites and axons, especially in the hippocampus (Enbert & Bonhoeffer, 2013; Gustafsson & Wigstrom, 2012; Lynch et al. 2010). Long-term synaptic potentiation has been correlated with learning and increased environmental input and is associated with an increase in synaptic contacts and changes in the morphology and number of dendritic spines (Enbert & Bonhoeffer, 2013; Lynch et al. 2010). As will be detailed below, this long lasting synaptic activity in turn appears to bind the pre and post-synaptic surfaces involved so that future neuronal activity is correlated as well. The proliferation of dendritic spines and associated synapses would allow for the creation of wide spread neuronal networks.
NEURAL CIRCUITS & LONG TERM POTENTIATION
The networking principles first proposed by Cajal, Hebb, Hilgard, Marquis, and Tanzi, have been explored in a number of laboratories through the analysis and experimental manipulation of long term synaptic potentiation (Barnes, 1979; Bliss & Lomo, 1973; Enbert & Bonhoeffer, 2013; Lynch, 1986; Xu et al., 2008). Long term potentiation, first discovered by Lomo (1966) in the dentate gyrus and which lasted for several days, is a form of "reverberating" neural activity that has been noted to occur in both pre but predominantly in postsynaptic neurons, and has since been directly associated with new learning. Long term potentiation (LTP) is thus associated with what Hebb (1949) described as reverberation. LTP is associated with the creation of long lasting memories as represented by neuronal circuits that are highly active.
LTP is best described as a long lasting post synaptic dopolarization which is induced through the repetitive stimulation and summation of excitatory post-synaptic potentials (EPSP). Presumably the prolonged and repetitive nature of these EPSPs counter inhibitory influences of GABA (Davies et al., 2012). However, LTP can also be triggered by very brief periods of repetitive excitation, and may persist from hours to days to weeks to months (Barnes, 1979; Bliss & Lomo, 1973; Davies et al., 2012; Lomo, 1966; Lynch, 1986). There are in fact different types of LTP depending on their length. The short version of LTP is referred to as short-term potentiation (STP) and lasts for about 30 minutes, whereas the longer version of LTP is divided into LTP1, LTP2, LTP3, depending on how long it lasts.
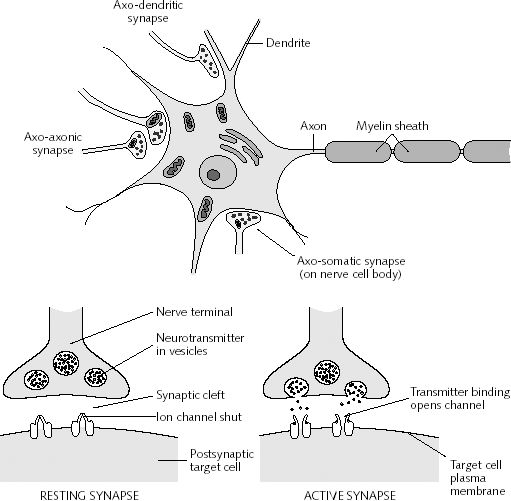
In addition to countering GABA, or activating MNDA receptors, LTP may also be produced due to increases in neurotransmitters. As is well known, those axons which are repeatedly utilized for information transmission increase their supply and release of neurotransmitters. Specifically, it has been shown that the induction of LTP is directly associated with increases in glutamate secretion with each subsequent stimulation (Bliss & Collingridge, 1993). As gutmate is excitatory, this would account for increased excitation in the post synaptic dendrite.
In addition, a dendrite which is repeatedly stimulated becomes more complex and grows new dendritic spines (Enbert & Bonhoeffer, 2013), thus increasing the number of synaptic contacts, including the surface area of each individual receptor surface (at the synaptic junction) --all of which becomes more extensive so as to take advantage of the increased amount of neurotransmitter available. Hence, LTP appears to be both a pre and post synaptic phenomenon (e.g. Bashir et al., 2012; Gloor, 2011) and appears to interlock the pre and post synaptic junction (Davies, et al. 1989; Kauer, et al. 2012) of the various interacting neurons such as through synaptic growth and the addition of dendritic spines (Enbert & Bonhoeffer, 2013). Synaptic growth and an increase in dendritic spines would obviously improve the quality and amount of information being received and processed, thus enhancing memory.
Because LTP develops in the presynaptic terminal during the course of the first half hour or so after learning it is believed that this activity corresponds to the development of short-term and then long-term memory. As noted, the short version of LTP is referred to as STP. However, LTP1 lasts for about 6 hours and appears to be maintained not only by countering the influences of GABA but through the buidup of protein kinases (in the post synaptic dendrite). the increase in kinases appears essential for transforming STP to LTP1 and thus prolonging this activity (Bliss & Collingridge, 1993).
As per LTP2, which last for about 10 hours, this appears to require a buildup in dopamine and activation of (excitatory) glutamate receptors.
LTP3 lasts for weeks and is associated with dendritic growth, and thus reflects genetic activity and protein synthesis (Bliss & Collingridge, 1993) --possibly related to plasmid exchange beween neurons and increased electrical activity within the proneural-genes (Joseph, 2008c,d,e). LTP3 is also associated with glutamate activity (which is excitatory, and activation of an assemble of neural-genes, including c-fos, jun-B, and zif/268 (Richardson, et al., 1992) which are associated with neural growth and differentiation. Genetic activity would thus account not only for LTP, but synaptic growth, all of which acts to bind together innumerable synapses thus creating enduring memories.
Hence, it appears that long-term memories are created through the establishment of numerous new synaptic connections formed through LTP-induced synaptic growth.
LTP is believed to be a major factor in the establishment of wide spread neural networks. These putative neural networks thus maintain complex and associated memories related to those significant events which were learned and which induced the LTP. That these LTP-induced networks may be widespread, is also suggested by findings demonstrating the development of LTP not only in the hippocampus, but the amygdala, entorhinal cortex, the frontal lobes, visual cortex, and the motor areas of the frontal lobes (Artola & Singer, 2010; Chapman et al., 2010; Sutor & Hablitz, 2018). Possibly, the development of LTP in different cortical areas may represent the binding together of these areas when engage in certain types of learning.
Temporal and spatial contiguity in axonal-dendritic synaptic activity has been postulated to be very important in establishing neural networks including those specific to particular memories (Gustafsson & Wigstrom, 2012; Lynch, 1986; Singer, 2010). Presumably different axons and dendrites must be highly active whereas yet others must be silence and at the same time for these exclusionary networks to form (Xu et al., 2008), thus creating networks for specific memories. That is, since a dendrite may receive input from hundreds of axons all of which may fire at different times, in order for a neural circuit to be formed so as to maintain a particular memory, it has been hypothesized that the activity between a number of specific presynaptic and post synaptic surfaces be correlated and thus linked together, whereas others remain at a low level of excitation so that they do not become linked with the emerging network. A large number of specific axons and dendrites must be actived simultaneously which in turn allows these pathways to consolidate (Singer, 2018) whereas yet others are silenced (Xu et al., 2008).
Moreover, this specificity must occur not only throughout the brain as related to the same experience, but within specific dendritic spines. Since a single dendrite can be likened to a tree but with innumerable branches and twigs, i.e. spines, a specific spine must also be activated whereas other "twigs" must remain silent.
For example, axon 1 and dendritic spine-1 (DS-1), and others like them in other parts of the brain and linked to the same experience, must be activated at the same time, so as to form one circuit that is associated with a specific memory. As to a second memory, activity occurring between axon 99 and dendritic spine DS-2 (and others like them) and even between axon-1 and DS-2 must occur at a different point in time (as compared to axon-1 and DS-1) so as to become incorporated into a second circuit (DS-2/axon-99). In this manner, large number of neurons and axons and specific dendritic spines become linked together by specific experiences, whereas others become linked by different experiences.
Moreover, only those synapses which show the highest level of activity simultaneously can be linked. For example, Lynch and colleagues (2010), sequentially stimulated three different afferents which terminated at the same dendrite, such that each burst of activity overlapped somewhat with the next. They found that the degree of LTP induced was greatest at the synapse which was first stimulated, intermediate for the second, and least of all for the third. Hence, LTP appears to bind together those neurons which initially share parallel activity--a process that must occur in numerous synapses which are activated simultaneously to process the same event (or different aspects of the same event).
As described by Singer (2010, p. 225), "the integration interval during which presynaptic and postsynaptic activation must coincide in order to lead to stabilization of a pathway...(and)...excitatory and inhibitory inputs to the same dendrite must be activated and silenced respectively. The efficiency of stimuli to induce modifications of cortical circuitry will increase to the extent that the stimuli not only match the response properties, but also conform with the resonance properties of more distributed neuronal assemblies."
However, it is also necessary for yet other neural networks to become silenced so that specific circuits can be formed. As summed up by Xu et al. (2008, p. 891), "extensive long-lasting decreases in synaptic efficacy may act in tandem with enhancements at selected synapses to allow the detection and storage of new information by the hippocampus."
Hence, it appears that various neocortical and limbic regions may come to be linked via spatially and temporally correlated synaptic activity coupled with decreases in the activity of yet other circuits so that specific stimuli can be attended to, learned, and stored in memory. Similarly, structural alterations and synaptic links may be induced by processing similar or associated perceptual experiences. Due to LTP, not only might different neurons come to be activationally linked (whereas yet others are excluded), but the neuron may come to have a lower firing threshold due to an increase in transmitter levels and activity as well as other factors. For example, LTP has been demonstrated in those synapses with glutamate receptors (see Stevens, 1989). These receptors open up which enables ions to flow through into the postsynaptic neuron. According to Stevens (1989, p. 461), "the glutamate that is released from the axon terminal... acts on the postsynaptic membraine to change the postsynaptic voltage and --under the right circumstances-- permits an influx of calcium ions" which allows for the develop of postsynaptic LTP.
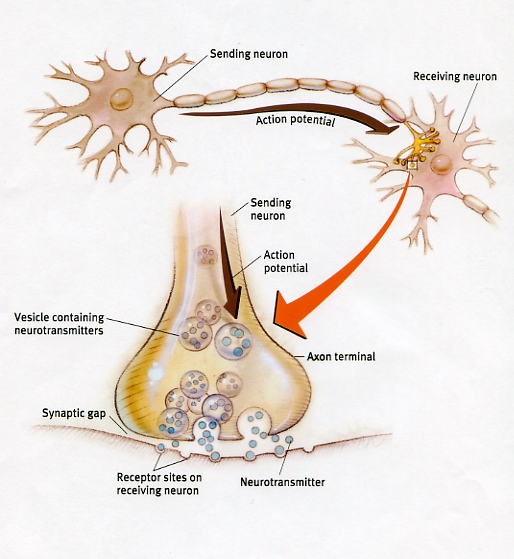
In this regard, LTP appears to be, at least in part, a function of increased transmitter levels (due to the release of more vesicles) as well as the changes in the post synaptic receptors which appear to increase in size which allows them to absorb more of the excess transmitter. In this manner the post synaptic membrane becomes increasingly sensitive which reduces the threshold for activation thus allowing LTP to develop (Davies, et al. 2009).
Hence, corresponding with the development of LTP it is presumed that a neuron can fire more easily, particularly in response to input from the same exact source. In addition, axons and dendritric spines become linked due to LTP and associated neuroplasticity. Hence, LTP is strongly related to the coupling of different neurons which repeatedly interact, thus forming a neural network that can be easily activated and which represents the totality of an experience. By contrast, neuronal sources that provide a different or later form of stimulation, come to be excluded from this particular circuit and may be stored in a different neural network representing separate and distinct memories and thus different experiences.
However, because these memories are widely distributed through a network of LTP-interlocked neurons, then just a fragment of the original experience may be able to trigger the entire network, thus generating the remembrance of the entire experience.
LONG-TERM DEPRESSION
Whereas the synaptic connections between certain axons and dendritic spines may be strengthened, yet others may be inhibited from forming (due to lateral inhibition via GABA). Moreover, some already established synaptic links may come to be inhibited through the development of long-term depression, which may also be a function of lateral inhibition. For example, if two neurons form synapses with the same dendrite but with different dendritic spines, the more active of the two may inhibit the activity of the other (Artola & Singer, 1993). Even synaptic links that have developed LTP may be inhibited and develop long-term depression (Artola & Singer, 1993) --which may be associated with forgetting.
PARALLEL, SEQUENTIAL & ISOLATED NEURAL NETWORKS
Different memories are presumably represented by specific neuronal circuits, which when later activated, may recreate the original cognitive, emotional, and perceptual experience (Halgren, 1992; Gloor, 2011; Joseph 1982). Correspondingly, a complex memory may be triggered by a variety of stimuli as well as a single cue which initially activates only part of the neural network. When one region of the neural network is activated, associated neurons within the circuit are likely to become aroused in parallel or sequentially.
Not all neural networks, however, may be characterized by parallel processing. For example, neural networks may be sequentially linked, such that activation of one neuron within the network will result in a step-wise pattern of sequential activation of various parts of the circuit. This would allow for one neuron to predict or determine what stimulus pattern or sequence will come next (Gloor, 2011; Sejnowski & Tesauro, 2010). For instance, in getting dressed a whole sequence of associated actions take place which are so well learned that one need not even think about the different steps involved; e.g. putting on shirt, fastening clothes, etc. The entire circuit of experience is sequentially activated and occurs almost in reflex fashion. However, some neural circuits may be characterized by both parallel and sequential activation.
Isolated Memories.
Certain memories may be shared between circuits, or confined to a specific neural network. If there are few other memories associated with that circuit, then this memory can come to be isolated and may not easily retrieved. That is, only a selective and quite narrow assortment of thoughts or associations or future experiences may trigger memory recall, in the absence of which the memory may appear to be forgotten (see chapter 29, 30). This is particularly likely during early infancy and childhood due to the immaturity of the neocortex which results in the establishment of isolated memories (Joseph, 1982, 2000). Indeed, these memories can become progressively more isolated due to programmed cell death.
Moreover, over the course of development an earlier learned response may come to be superseded by a more complex learning experience. Or one neural circuit may act to suppress and inhibit the activation of a second network which is associated with certain behavioral acts which are no longer appropriate; the adult ceases to act like a child, the old man no longer behaves like a teenager. With decreased activation, the original circuit may decay as neurons and dendrites drop out, and as LTP diminishes. The result is memory loss or amnesia for specific events and experiences.
The Permanence & Instability of Memory.
Because of this neuronal complexity and the large number of synapses involved, it is presumed that individual neurons can die, or be eliminated from the circuit without significantly disrupting the overall integrity of the pattern and associated memory (Gloor, 2010, 2011). As such, memories can be resistant to degradation. Moreover, this allows individual neurons to be involved in a variety of circuits, or to drop out of one and become an integral part of another without significantly affecting memory.
Presumably it is because memories are stored in different neurons and in different regions of the brain, that different components may be retained or lost at different rates or as a consequence of different forms of interference, including brain damage and degeneration. It is also because there are different neural circuits that certain memories can come to be isolated, selectively forgotten, or repressed, or conversely, recalled forever with ease.
As noted above, however, memories formed during infancy and early childhood may be stored in isolation due to the immaturity of the neocortex and the fact that as the child ages neurons continue to grow and new synaptic connections are being massively formed. However, as the brain matures, neurons not only continue to grow (and possibly divide) and establish new synaptic connections, but excess neurons and synaptic connections are also eliminated as initially brain cells and synapses are produced in excess and innumerable random interconnections are formed (Rakic, Bourgeois, Echenhoff, Zaecevic & Goldman-Rakic, 1986). In fact, neuronal density declines by up to 50% over the first decade (Blinkov & Glezer, 1968; Huttenlocher, 2010). Programmed cell death is an integral part of the maturational process and "fine tunes" the nervous system (Finlay & Slattery, 1983; Joseph, 1982; Oppenheim, 1981) as well as learning and memory.
However, programmed cell death may also encompass the elimination of "excess" memory-laden neurons and synapses which may be shed by the hundreds of millions, thus erasing from the mind and brain associated early memories; a process which may contribute, over time, to an amnesia for early childhood experiences.
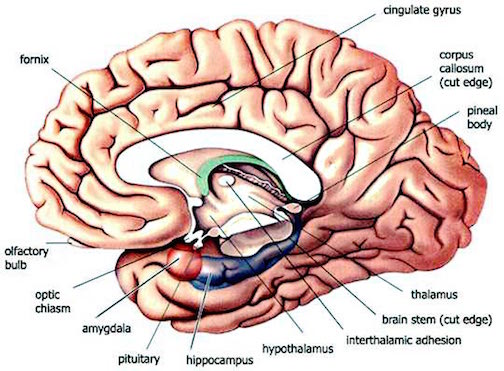
SHORT & LONG TERM MEMORY: THE ANTERIOR & POSTERIOR HIPPOCAMPUS
In humans, the hippocampus, the anterior and ventral hippocampus in particular, is usually associated with learning and memory of cognitively relevant information e.g. the long term storage and retrieval of newly learned information (Fedio & Van Buren, 1974; Milner, 1966; 1970; Penfield & Milner, 1958; Gloor, 2011; Rawlins, 1985; Scoville & Milner, 1957; Squire, 1992; see also commentary in Eichenbaum et al. 1994).
During learning activities LTP has been repeatedly found to occur within the hippocampus (Barnes & McNaughton, 1985; Enbert & Bonhoeffer, 2013; Lynch, 1986; Xu et al., 2008). Dendritic proliferation and the creation of specific neural circuits, as well as LTP, also occurs in the hippocampus during learning (Barnes, 1979; Enbert & Bonhoeffer, 2013; Lynch, 1986). It has also been demonstrated that hippocampal pyramidal cells undergo synaptic modification when flexible stimulus response associations are being formed (Enbert & Bonhoeffer, 2013; Rolls, 1987, 2012). Similar correlations between hippocampal LTP and learning have been found on tasks involving memory for visual-spatial relations (Barnes, 1979). Moreover, during acquisition, not only does LTP increase but so to does EEG evoked responses within the hippocampus (see Barnes & McNaughton, 1985).
Barnes and McNaughton (1985) found, however, that long-term hippocampus synaptic potentiation was more long lasting and more quickly reached by young than old animals. Older animals also demonstrated slower learning and faster rates of forgetting of spatial information.
Many of these synaptic and activational changes, in turn, are most apparent within the anterior regions of the hippocampus (Lynch, 1986) --which maintains rich interconnections with the amygdala (Amaral et al. 1992). Moreover, this same region of the hippocampus will become electrophysiologically potentiated during learning tasks. In fact, long term potentiation lasting up to several days have been noted in the hippocampus following successful learning trials (Lynch, 1986), which in turn may reflect the transition of information from short-term, to long term memory, at which point LTP ceases to be a factor in further memory maintenance.
These findings are consistent with the notion that the longer the potentiation, either at the cellular or hippocampal level, the stronger might be the memory, and the more likely it will persist over time. Hence, the formation of short-term memories appears to dependent on the anterior portion of the human hippocampus and the binding action of LTP which creates links between different synapses thus allowing for the transition from short to long term memory. It is noteworthy, however, that the amygdala appears to act on the anterior hippocampus in order to emotionally reinforce as well as modulate its functional activity (Gloor, 1992, 2011; Halgren, 1992), and LTP occurs in both nuclei.
If the anterior hippocampus is injured or functionally suppressed, very little new cognitive learning occurs and potentiation does not appear. However, once these memories have been established in long term memory, the role of the anterior regions of the human hippocampus appears to diminish. This would explain why long term memory for long ago events is spared with hippocampal destruction.
Squire argues (1992, p. 222) that "the hippocampal formation is essential for memory storage for only a limited period of time. A temporary memory is established in the hippocampal formation at the time of learning in the form of a simple memory, a conjunction, or an index. The role of the hippocampus then gradually diminishes, and a more permanent memory is established elsewhere that is independent of the hippocampus...and... the neocortex alone gradually becomes capable of supporting usable, permanent memory. This reorganization could depend on the development of cortico-cortical connections between separate sites in neocortex, which together constitute the whole memory."
On the other hand, there is some human evidence which indicates that the more posterior human hippocampus may be responsible for long term memory access which is why retrograde amnesia has been reported with damage to or electrical stimulation of this area (Fedio & Van Buren, 1974; Penfield & Mathieson, 1974). Indeed, Penfield and Mathieson (1974) suggested that memories might actually migrate over time along the length of the hippocampus in a posterior direction, and that the ability to retrieve these memories follows this posterior movement.
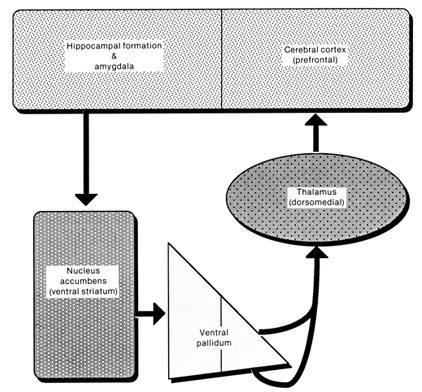
THE HIPPOCAMPUS AND ASSOCIATED MEMORY STRUCTURES
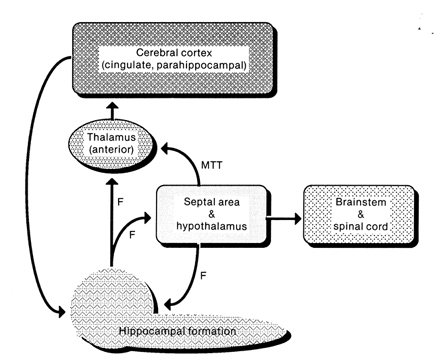
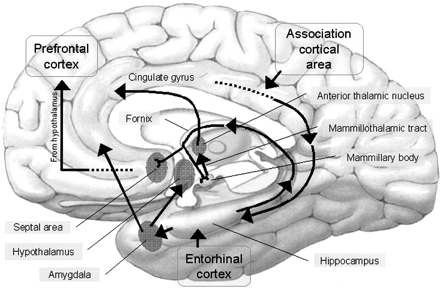
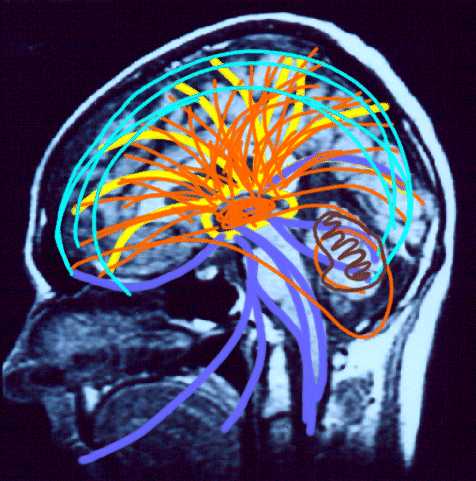
Reverberating neurons are presumably located in various regions of the neocortex, and are apparently bound together via the simultaneous activity and steering influences involving the frontal lobes (Joseph, 1986a, 2013a), dorsal medial thalamus, and in particular the amygdala and hippocampus (Gloor, 2011; Graff-Radford, et al. 2010; Lynch, 1986; Rolls, 1992; Squire, 1992). These structures are all interlinked and highly involved in attention, arousal, and memory functioning, and probably act together so as to establish and maintain specific neural circuits and networks associated with specific memories (e.g., Brewer et al., 2008; Squire, et al,. 1992; Tulving et al., 1994; Wagner et al., 2008). For example, these different networks and neurons may be linked via the steering influences exerted by the frontal lobes etc., which can selectively activate or inhibit these memories and associated tissues in a coordinated fashion, and which can tie together certain perceptual experiences so as to form a complex multi-modal memory (e.g., Dolan et al., 2011; Joseph, 1982, 1986a, 2012a, 2013a; Kapur et al., 2015; Squire, et al,. 1992; Tulving et al., 1994; Dolan et al., 2011; Brewer et al., 2008; Wagner et al., 2008).
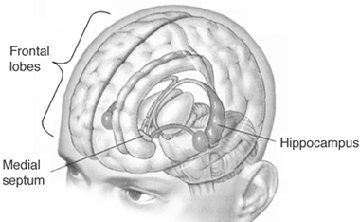
Likewise, since the hippocampus is a prime location for the development of LTP and is significantly involved in many aspects of memory functioning, it is presumably able to exert steering influences on different neocortical sites with which it is also richly, albeit indirectly interconnected via the entorhinal cortex. Via LTP (and the entorhinal cortex and dorsal medial nucleus) the hippocampus presumably acts to bind these divergent neocortical sites together so as to form a circuit of experience (Enbert & Bonhoeffer 2013; Lynch, 1986; Squire, 1992; Xu et al., 2008).
However, the human hippocampus (and overlying temporal lobe) also interacts with the amygdala and frontal lobes (Brewer et al., 2008; Squire, et al,. 1992; Tulving et al., 1994; Wagner et al., 2008), each of which also plays unique and overlapping roles in memory. Moreover, the hippocampus is important only in regard to certain aspects of memory, such as spatial, verbal, auditory, cognitive and recognition memory (Gloor, 2011; Nishitani, et al., 2013; Nunn et al., 2013; Xu et al., 2008), whereas the amygdala is concerned with emotional memory (Gloor, 2011; Halgren, 1992; LeDoux, 2014) and becomes activated by bizarre or traumatic stimuli, and when recalling trauma-memories (Rauch et al., 2014; Shin et al., 2011). By contrast, the frontal-thalamic system is more involved in retrieval and so called "working memory," as well as keeping something in mind so that it can be remembered and performed later (Brewer et al., 2008; Squire, et al,. 1992; Tulving et al., 1994; Wagner et al., 2008).
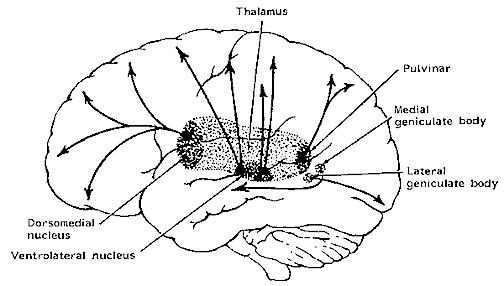
Again, LTP is not an exclusive property of the hippocampus but also appears in entorhinal cortex, the frontal lobes, visual cortex, the motor areas of the frontal lobes (Artola & Singer, 2010; Chapman et al., 2010; Sutor & Hablitz, 1989) and the adjacent amygdaloid nucleus (Chapman et al., 2010) with which it is richly interconnected. Indeed, amygdaloid neurons show plasticity in response to learning (Lynch, 1986), and LTP has been induced in amygdala neurons (Chapman, et al. 2010). Fear induced neural plasticity in the form of LTP has been noted in amygdala neural pathways as well (Clugnet & LeDoux, 2010). This is presumably a consequence of the amygdala's involvement in most aspects of emotional experience, including the formation of cross-modal emotional associations and memories (Gloor, 1992, 2011; Halgren, 1992; Kesner, 1992; LeDoux, 1992, 2014; Rolls, 1992; however, see Murray & Gaffan 1994).
The amygdala is also intimately interlinked with the anterior hippocampus and appears to exert reinforcing and modulating influences on this nuclei (Gloor, 2011; Halgren, 1992). Moreover, they both project to adjacent thalamic relay neurons which raises the possibility they act conjointly to form separate but closely aligned neural networks concerned with different aspects of memory.
THE HIPPOCAMPUS AND ENTORHINAL CORTEX
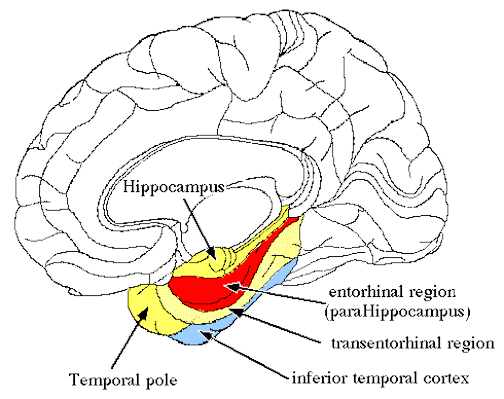
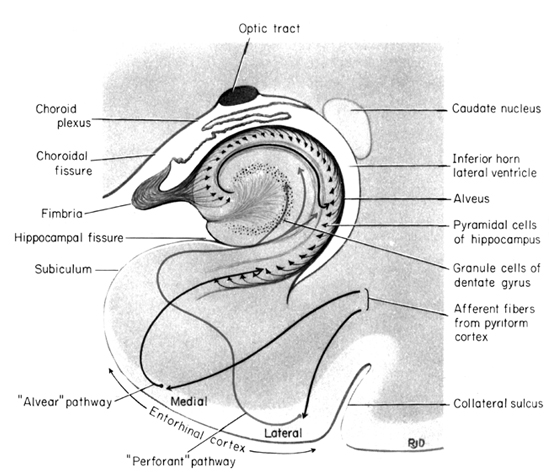
The hippocampus does not receive direct neocortical input. Moreover, the data it does received, at least from the neocortex, originates in the association areas and is first transmitted to the entorhinal cortex or amygdala, and is then relayed to the hippocampus (see Horel et al. 1987; Issausti et al. 1987; Squire, 1992); the only apparent exception being auditory input which is transfered directly from the primary auditory areas to the entorhinal cortex. It is also via the overlying entorhinal area that the hippocampus receives amygdaloid projections (Carlsen et al., 1982; Gloor, 1955, 2011; Krettek & Price, 1976; Steward, 1977) and fibers from the orbital frontal and temporal lobes (Van Hoesen, et al., 1972). Thus, the hippocampus only receives neocortical input indirectly, and for the most part this is relayed by the entorhinal cortex--the "gateway to the hippocampus."
The entorhinal cortex is truly unique, not only because it serves as an interface between the hippocampus and the neocortex, but because this medial located structure consists of between 7 and 8 layers (Braak & Braak, 1992; Ramon y Cajal, 1902/1955; Rose, 1926). The entorhinal cortex also maintains massive interconnections with all multi-modal neocortical association areas (as well as with the amygdala, hippocampus, septal nuclei, olfactory bulb, etc.) but apparently none of the primary sensory areas (Leichnetz & Astruc, 1976; van Hoesen, et al., 1975). Hence, the entorhinal cortex must play a supramodal role that is exceedingly unique and profoundly important in memory and cognitive processing, and may play different roles, in for example, recognition vs recall vs short term and long-term memory.
[-INSERT FIGURE 10 ABOUT HERE-]
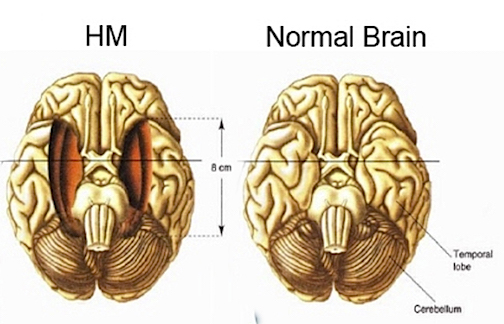
For example, some believe that the neocortex of the temporal lobe is the repository of those long term memories initially processed by the entorhinal cortex and hippocampus and that these latter structures are more important in recognition memory and act to store this material in the neocortex. Consider, for example, patients who undergo "hippocampal removals" whereas the overlying neocortex was spared (Milner, 2010) and patients such as the famous H.M., who underwent bilateral mesial temporal removals: amygdala, hippocampus, entorhinal cortex (Milner, 1968). These patients (particularly those with right sided destruction) perform exceedingly poorly on visual recognition memory tests, including those involving recurring nonsense figures (Kimura, 1963) and human faces (Milner, 2010).
Hence, recognition memory is disrupted with entorhinal and hippocampal removals. However, short term and immediate memory remains intact.
SHORT VS LONG TERM MEMORY LOSS, RETRIEVAL & HIPPOCAMPAL DAMAGE
When the hippocampus has been damaged the ability to convert short term memories into long term memories (i.e. anterograde amnesia), becomes significantly impaired in humans (Eichenbaum et al. 1994; Gloor, 2011; MacKinnon & Squire, 1989; Nunn, et al., 2013; Squire, 1992; Victor & Agamanolis, 2010) and primates (Zola-Morgan & Squire, 1984, 1985a, 1986). Lesions of the hippocampus can also disrupt time sense and temporal sequencing such as involving timing tasks (Meck et al. 1984). And, memory for words, passages, conversations, and written material is also significantly impacted, particularly with left hippocampal destruction (Frisk & Milner, 2010; Squire, 1992).
Moreover, spatial memory is significantly impaired among a variety of species with hippocampal lesions (Mishkin, et al. 1984; Nunn et al., 2013; Weiskrantz, 1987). In fact, the capacity to cognitively map, or visualize one's position and the position of other objects and individuals in visual-space is dependent on the hippocampus (Nadel, 2012; O'Keefe, 1976; Wilson and McNaughton, 1993). The hippocampus contains "place" neurons which are able to encode one's position and movements in space. Specifically, O'Keefe, Nadel, and colleagues, found that hippocampal pyramidal cells became sensitive to particular spatial coordinates and the location of objects in visual space, but that these spatial maps were also very plastic. These authors also found that as the subject moves about in that environment, entire populations of cells would fire but only when in a particular spot, whereas other cells would fire when in a different location. Moreover, some cells respond not just when moving about, but in reaction to the speed of movement, or when turning in different directions. Moreover, some cells are responsive to the movements of other people in that environment and will fire as that person is observed to move around. (Nadel, 2012; O'Keefe, 1976; Wilson and McNaughton, 1993).
The hippocampus, therefore, can create a cognitive map of an individuals environment and their movements within it. Presumably it is via the hippocampus that an individual can visualize themselves as if looking at their body from afar, and can remember and thus see themselves engaged in certain actions, as if one were an outside witness (Joseph, 2008b, 2013d).
Therefore, patients with hippocampal destruction may demonstrate severe visual-spatial memory disturbances and may easily lose their way, forget where they place items, or where things are located (Nunn et al., 2013; Squire, 1992); particularly with right hippocampal injury. Nevertheless, initially individuals with even bilateral removal of the hippocampus demonstrate good initial retention and short-term memory (Horel, 1978; see commentary in Eichenbaum et al. 1994).
BILATERAL HIPPOCAMPAL DESTRUCTION & AMNESIA
Given the powerful effect of unilateral lesions on memory, bilateral destruction of the anterior hippocampus results in striking and profound disturbances involving almost all aspects of cognitive and recognition memory and new learning (i.e. anterograde amnesia), as well as retrograde amnesia (RA) for events that may extend 2-3 years in time (Marslen-Wilson & Teuber, 1975; Milner, 1966; Murray, 1992; Penfield & Milner, 1957; Scoville & Milner, 1956, Squire, 1992), particularly if the posterior hippocampus has also been injured or removed (Fedio & Van Buren, 1974). In the famous case of H.M. who underwent bilateral hippocampal and amygdala removals, retrograde amnesia was found to extend over 11 years (Corkin, 1984). However, as H.M. also had severe epilepsy, this extensive loss of pre-surgery memory may due to this preexisting lesion. For example, in other cases, patient have been able to describe and recall remote memories and are little different from normals in this regard (Horel, 1978; MacKinon & Squire, 1989; Squire, 1992).
Nevertheless, there may be a permanent loss of memory extending backwards several years in time. A 37-year old Lt. Cornel who had sustained bilateral posterior temporal lobe and hippocampal destruction secondary to prolonged anoxia in 1969 (after being injured in Viet Nam), had no memory of his injury, and no memory of major events that had transpired two years prior to his injury. Although he could recall the death of President J.F. Kennedy, he could not recall the assassination of his brother, Robert Kennedy, though he could recall that Robert Kennedy had been attorney general and was running for president. As part of his "memory retraining" I had him read and reread short newspaper style articles including those describing the death of Robert Kennedy, and would then ask him questions about the story. Each time he read this latter article he had the same exact emotional and behavioral reaction, expressing surprise, shock, and sorrow, He had absolutely no memory of having read the same exact article just 30 minutes earlier--though he had answered all questions correctly immediately following each reading. Although I worked with him three times a week, for an hour a day, for over 3 months, he had absolutely no memory of who I was or of ever having met me.
In yet another case, an 18 year old boy who sustained bilateral inferior-anterior-medial temporal lobe and hippocampal destruction (following a car accident immediately after high school graduation), thought he was still sixteen an a "junior or a senior" in high school "I'm not sure." He also suffered a profound anterio-grade amnesia. His father described this boy's memory as "like a sieve. You fill it up with information and 30 minutes later its all leaked away. If you leave him alone for half an hour he will completely forget where he is, how he got there, and so on. Then he begins to panic for he forgets he was in a car accident and has lost his memory." In fact, while examining this boy he suddenly looked up at me in astonishment and had no idea who I was, where he was, why he was there, and so on. Nevertheless, in this particular boy's case, his anterograde memory loss was not global as he was capable of learning new motor routines and was able to get a job performing electronics assembly. However, he has also had to carry a note pad with "reminders" to tell him where he was, why he was there, etc.
Presumably, in part, loss of memory and the inability to acquire new memories following bilateral hippocampal injury is due to an inability to consolidate new information, such that short term memory remains intact whereas long-term memory is disrupted. In fact, with hippocampal injuries, the creation of LTP is disrupted (Lynch, 1986).
LEARNING AND MEMORY IN THE ABSENCE OF THE HIPPOCAMPUS
As noted, the 18-year old mentioned above was capable of learning simple motor tasks. Similarly, monkeys with hippocampal destruction, including those with bilateral destruction, although demonstrating severe memory impairments are capable of learning motor skill tasks and acquiring habits (Squire, 1992; Zola-Morgan & Squire, 1984, 1985a). Indeed, it has been repeatedly demonstrated that various aspects of learning and memory are retained in the absence of the hippocampus (see Eichenbaum et al. 1994; Horel, 1978; Seldon et al., 2012; Squire, 1992; Zola-Morgan, et al. 1986). This includes the learning of skilled and coordinated motor programs -as these "memories" appear to be dependent on the basal ganglia (Heindel et al. 2012; Packard, et al. 1989; Wang et al. 2010), cerebellum (Schmahmann, 2011; Thompson, 1986), inferior parietal lobule, the supplementary motor areas, and the lateral frontal motor areas (chapters 19, 20). The role of the hippocampus is minimal in these forms of learning (see Squire, 1992).
Classical conditioning is also independent of the hippocampus which may be a function of the repeated nature of stimulus presentation -the hippocampus soon ceases to respond in repetitive experiences (see chapter 30). In contrast, single learning associations are more dependent on the hippocampus (Squire, 1992).
The learning of emotional information also appears to occur independent of the hippocampus -being dependent on the amygdala (Gloor, 2011; LeDoux, 2014; Seldon et al., 2012). As noted, LTP has been induced in the amygdala and amygdala pathways (Chapman, et al. 2010; Clugnet & LeDoux, 2010), which is presumably a function of its involvement in most aspects of emotional memory formation (Gloor, 2011, Halgren, 1992; Kesner, 1992; LeDoux, 2014; Rolls, 1992), and the creation of emotional memory neuronal networks.
It has also been claimed that amnesics can aquire what Tulving (1972) calls semantic memory (a "mental thesaurus") which includes factual information and knowledge of words. However, the savings is so minimal as to be insignficant (Gabrieli et al., 2012). Hence, in general, hippocampal injury results in profound memory loss.
INTERACTIONAL MEMORY SYSTEMS
There are all types of memories; i.e. verbal, visual, short-term, long-term, episodic, explicit, implicit, tacit, procedural, etc., and so on--and some investigators use different labels to describe the same type of memory; e.g., conscious/declarative/explicit vs unconscious/nondeclarative/implicit. Hence, some memories are easily consciously accessible and are therefore easy to declare because they are explicit, whereas there are other memories which are unconscious such that the individual may have no idea as to how they acquired these memories. For example, amnesics may display implicit knowledge and memory, though they may have no idea as to how they obtained that information (Penfield & Milner, 1958; Scoville & Milner, 1957; Squire, 1992).
Sigmund Freud and Carl Jung were among the first to study and theorize as to these distinct types of memories, and cognitive psychologists, such as Tulving, Schacter, and others, and a number of neuroscientists, such as Mishkin (1978), Squire (1992) and others, have essentially verified, experimentally, the existence of distinct memory systems such as those maintained by the amygdala and hippocampus.
For example, it has been shown that hippocampal lesions produce mild deficits in recognition memory (Mishkin, 1978, 2010), though if the lesion includes the adjacent medial temporal cortex including the perirhinal cortex, recognition memory is severely impaired--which suggests that mesial structures play their own unique role in memory. It has also been reported that recognition memory is severely disrupted with damage to the entorhinal cortex but is spared when the hippocampus is intact (see Murray, 1992; Squire, 1992). However, since these structures are contiguous, and as the entorhinal cortex projects to and receives input from the hippocampus, and acts as a major relay to and from the neocortex to this structure, it seems rather impossible to damage the entorhinal cortex without inducing structural and profound functional abnormalities in the hippocampus and vice versas (see Gloor, 2011, for related discussion).
Recognition memory has also been reported to be somewhat (but not always) spared when the medial thalamic nuclei and/or the frontal lobe destruction are the source of the amnesic disturbance (Graff-Radford, et al. 2010; Jetter et al. 1986); in which case retrieval may be severely disrupted. In most instances, however, recognition memory is significantly effected with lesions in this area.
Moreover, Mishkin (1978) showed that lesions of the hippocampus and amygdala results in severe recognition memory deficits. However, Mishkin also proposed that the hippocampus and amygdala form two parallel memory systems, which together form the main memory system (Mishkin, 2010).
The "main memory system" however, extends beyond the hippocampus and amygdala, and includes numerous structures including the dorsal medial nucleus, the frontal lobes, mammillary bodies, etc., with some aspects of this "memory system" serving to store and others to search and recall or recognize specific memories. As noted, recognition memory appears to be more dependent on the hippocampus and adjacent nuclei as well as the entorhinal cortex and amygdala (Mishkin, 1982; Murray, 1992; Squire, 1992), as well as the dorsal medial nucleus. Retrieval may be more dependent on frontal-thalamic interactions (Aggleton & Mishkin, 1983; Graff-Radford et al. 2010; Squire, 1992; Victor et al. 1989) with the right frontal lobe becoming activated when searching for visual and episodic memories, and the left frontal when searching for verbal memories (Brewer et al., 2008, Wagner et al., 2008). The hippocampus appears to be more involved in the actual storage of non-emotional memories, including those which are visual, spatial, and involving language.
Because there are so many memory systems, and although they are interactional, it is therefore possible to lose certain types of memory, whereas retaining those that would normally be associated with those which are forgotten. For example, an inability to remember how to get to the dentist's office, whereas his name and address are retained. Or, reexperiencing feelings of severe emotional upset when returning to scene of a crime, but being unable to clearly recall what transpired (e.g., Christianson & Nilsson, 1989).
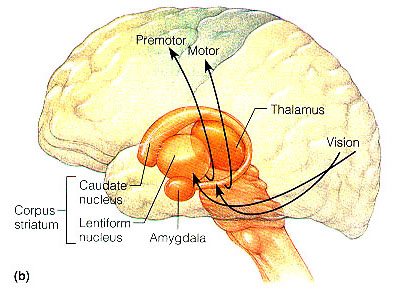
THE FRONTAL LOBES & THE DORSAL MEDIAL & ANTERIOR THALAMUS
The dorsal medial nucleus of the thalamus, as well as the frontal lobes are exceedingly important in memory functioning and information retrieval (Aggleton & Mishkin, 1983; Brewer et al., 2008; Dolan et al., 2011; Graff-Radford et al.2010; Hasegawa et al., 2008; Squire, 1992; Wagner et al., 2008; Victor et al. 1989), and both structures are intimately linked (see chapter 19) and interact with the temporal lobe and the amygdala and entorhinal cortex/hippocampus. For example, when engaged in episodic memory retrieval, there is increased activity in the frontal lobes and thalamus (Dolan et al., 2011) as well as the temporal lobes (Brewer et al., 2008; Wagner et al., 2008). In fact, the ability to remember a visual or a verbal experience, is directly correlated with activation of the right or left frontal lobe and the temporal lobes during that experience (Brewer et al., 2008; Wagner et al., 2008).

According to Wagner et al., (2008, p. 1190), as per verbal memories, "what makes a verbal experience memorable partially depends on the extent to which left prefrontal and medial temporal regions are engaged during the experience." Moreover, according to Brewer et al., (2008, p. 1185) the degree of this activity can be used to predict which experiences will be "later remembered well, remembered less well, or forgotten." Hence, activity increases in the right frontal lobe as a function of retrieval effort (Kapur et al., 2015), or a function of effort exerted at preventing retrieval.
As first fully proposed and detailed elsewhere (Como, Joseph, Fiducia, et al., 1979; Joseph, 1986a; Joseph et al., 1981) the frontal lobes play a signficant role in human memory including memory retrieval failure (Joseph, 2013a), and the inability to remember traumatic events (see chapter 29). The frontal lobes are also implicated in the failure to transfer these unpleasant memories from the right to left hemisphere (Joseph, 1992b; see also Hasegawa et al., 2008). That is, it has been proposed that although a memory may be recalled by the right hemisphere, through frontal lobe inhibitory action the memory may be suppressed or prevented from transferring to the left, such that the language regions of the brain remain amnesic. The individual thus knows, yet does not know, simultaneously, and may demonstrate the presence of unconscious memories.
Specifically, it appears that the frontal lobes, in conjunction with the hippocampus (Goldman-Rakic, 2010), reticular activating system, the amygdala and the dorsal medial thalamus (DMT), acts to gate and direct perceptual and cognitive activity occurring within the neocortex (Como et al., 1979; Joseph, 2013a; Joseph et al., 1981). That is, the frontal lobes, DMT, amygdala, and hippocampus interact when determining which neuronal circuits are created, linked together, inhibited or activated.
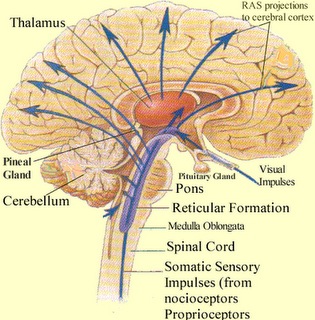
The frontal lobes, in fact, are highly concerned with arousal, attention, activation and neuronal inhibition (Como et al., 1979; Joseph, 1986a, 2013a; Joseph et al., 1981) as well as sustaining the creation of specific reverberating neural circuits. The frontal lobes are also particularly important in the capacity to retrieve previously stored information (Brewer et al., 2008; Wagner et al., 2008), as well as prevent access to those memories and perceptions deemed irrelevant or undesirable (see chapters 19, 29). Presumably it can retrieve or inhibit particular memories by acting directly on the neurons and neural network where they are maintained or indirectly via the thalamus and reticular formation through which it can selectively effect arousal. In this manner, the frontal lobes can selectively sample, deactivate, or enhance neural activity depending on the information it is searching for (chapter 19).
Conversely damage to the frontal lobes can result in difficulty retrieving memories (Joseph, 1986a) but has little effect on recogniton memory (Jetter, et al. 1986) where search, activation, and retrieval are not as important. Recognition memory can be activated by direct stimulation of the neurons and the neural network which have stored similar information in the past.
THE DORSAL MEDIAL THALAMUS
The frontal lobe, hippocampus, and the amygdala appear to share major roles in regard to neocortical activation and thus memory storage and retrieval, and all appear to partly relie on the dorsal medial thalamus (DMT) and anterior thalamus in this regard. The DMT (and the thalamus in general) are highly important in transferring information to the neocortex, and is involved in peceptual filtering, and exerting steering influences on the neocortex so as to direct attention (see Chapter 19). When damaged, there can result a dense verbal and visual anterograde amnesia (Graff-Radford, et al. 2010; von Cramon et al. 1985) as memories cannot be selectively stored or activated.
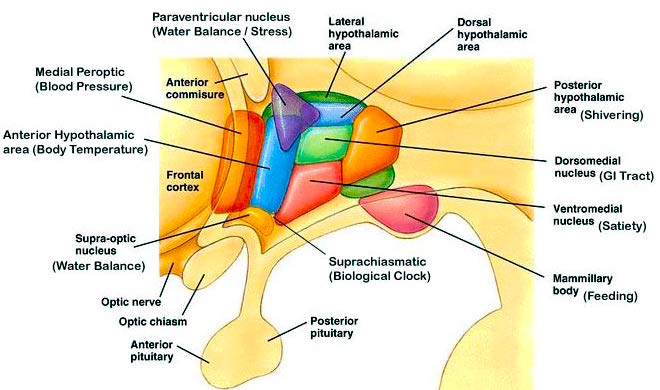
The DMT and anterior thalamus are able to perform these functions via inhibitory and excitatory influences (mediated by the frontal lobe and to a lesser extent, the hippocamus) on neurons located in the neocortex, limbic system, and reticular activating system. For example, the DMT and frontal lobes monitor and control, at the neocortical level, all stages of information analysis, and insure that relevant data and associations are shifted from primary to association to multi-modal associations areas so that further processing can occur. This is made possible since the frontal lobes are interlocked with the first, second, and third stage of neocortical information processing, and simultaneously projects to these neocortical regions and the DMT (see chapter 19) which enables it to exert inhibitory, facilitory influences so as to selectively gating information reception and processing.
Hence, the frontal-thalamic system can act to inhibit further processing and/or prevent this information from reaching the neocortex in the first place; i.e. at the thalamus prior to transfer to the neocortex, and at the level of the neocortex to prevent further processing. In this way, distractions and irrelevant stimuli may be filtered so that salient events may be attended to.
DMT, FRONTAL LOBES, KORSAKOFF'S SYNDROME, SEARCH & RETRIEVAL Through the widespread interconnections maintained by the frontal lobes, hippocampus, and thalamus, specific neuronal networks and perceptual fields can be selectively inhibited or activated, so that information may be processed or ignored, and so that conjunctions between different neocortical areas can be formed. These nuclei therefore directly mediate information processing, memory storage, and the creation of or additions to specific neural networks via the control of neocortical activity.
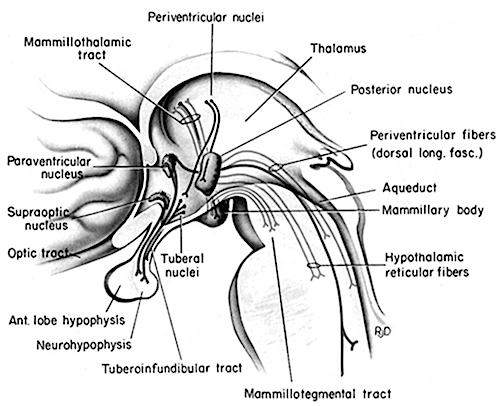
The frontal lobe and anterior and dorsal medial thalamus are also involved in search and retrieval, for these aspects of memory are severely disrupted with damage to the frontal lobes and dorsal medial nuclei, whereas recognition memory may remain much more intact (Graff-Radord, et al. 2010; Squire, 1992). However, recognition memory may be impaired as well. For example, one such patient (who, however, also had atrophy involving the mammillary bodies) was not only unable to recall that he had been a patient at the hospital for over a month, but repeatedly claimed to be "working" as a "janitor," and that he would be "going home at quitting time." However, when he was shown his hospital room and clothes, although he recognized the items as his, he persisted in claiming he was at work and that was the reason his clothes were in the hospital room was so he could change before leaving to go home (Joseph 1986a). As per the mammillary bodies (a structure also associated with memory), the hippocampus also maintains interconnections with this tissue (Guillary, 1956; Nauta, 1958), and "diencephalic" amnesia is a not uncommon manistations of mammillary injury (Victor & Adams, 1993; Victor et al., 1971).
Hence, when the frontal lobes and DMT and anterior thalamus are damaged, the ability to shift from one set of perceptions to another, or to selectively retrieve a specific memory, or to inhibit the activation of irrelevant ideas, associations, memories, and thoughts, is significantly impacted, and the patient may confabulate and produce "false memories" (Joseph, 1986a, 2012a, 2013a). The ability to alternate between and thus selectively activate specific neural networks is also disrupted, such that patients have difficulty shifting from one train of thought to another. That is, they may tend to become stuck in-set and to engage in recurrent perseveration (Joseph, 1986a, 2013a; Vikki, 1989) such that the same speech patterns or "memories" may be repeated unless something occurs to arrest and shift their attention.
Hence, with frontal-thalamic damage, retrieval is impaired as is the ability to selectively recall of shift between old versus recent memories, and to keep track of the order in which they may have been formed e.g. morning, evening, yesterday, last week, five years ago and so on (see Graff-Radford, et al. 2010; Squire, 1992; Talland, 1961). Therefore, a patient may deny possessing a certain memory of an event which occurred five weeks ago, because he is searching his memory store for events which were experienced five years ago or five days ago.
For example, one patient with severe frontal lobe damage claimed to have never been married and laughingly indicated that he never wanted to get married and that marriage was a bad idea. When pressed as to why, he exclaimed that "the wife may just up and die on you," and then began to laugh quite loudly and then just as suddenly began to sob and cry. When asked why he was crying, he claimed not to know why. However, when he was reminded that he had been married, he was suddenly able to recall that his wife (of two years) had been killed in the same accident that resulted in his injuries.
Another patient with anterior and dorsal medial thalamic damage erroneously stated that the president was Lyndon Johnson. However, when told the correct date ("1982") she correctly named Ronald Regan (Graff-Radford, et al. 2010).
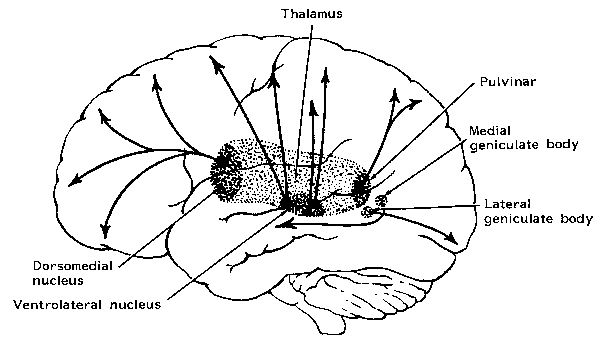
Thus "time sense" and capacity to selectively search memory may be disrupted with frontal-dorsal medial dysfunction; a condition which is also characteristic of Korsakoff's syndrome. For example, Talland (1961, p. 375) described one woman with Korsakoff's disease who "after ten year's hospitalization continually maintained that she has been brought in the previous day for observation." When questioned she also argued that she still lived with her husband in a fancy hotel, where she had lived "the life of a well-to-do lady of leisure. She seemed unaware that the conditions of that very agreeable life had long since ceased to operate."
Individuals suffering from Korsakoff's syndrome demonstrate profound disturbances of memory, including a dense verbal and visual anterograde amnesia (Aggleton & Mishkin, 1983; Graff-Radford, Tranel, Van Hoesen & Brandt, 2010; Victor et al. 1989; von Cramon et al. 1985) and in some cases a more temporally graded memory loss for the remote past (Squire, 1992) --although the hippocampus is intact. Rather, in Korsakoff's syndrome, damage typically involves the dosal medial nucleus of the thalamus and atrophy of the frontal lobes (Aggleton & Mishkin, 1983; Graff-Radford, et al, 2010; Lishman, 1981; Mair et al., 1979; Victor et al. 1989; von Cramon et al. 1985; Wilkinson & Carlen, 1982) as well as the mammillary bodies (Victor & Adams, 1993; Victor et al., 1971); i.e. "diencehalic amnesia".
Nevertheless, recognition memory may be partially intact, and with selective reminders and reorientation, the retrieval deficits associated with frontal/dorsal medial damage can sometimes be partly overcome (Graff-Radford, et al. 2010).
As noted, the mammillary bodies are interconnected with the hippocampus also maintains interconnections with this tissue (Guillary, 1956; Nauta, 1958), as well as to the hypothalamus and brainstem reticular formation, and are relayed to the septal nucleus--which is also implicated in memory (Gage et al., 1983; Olton, 2010). Injury to the mammillary bodies is typically associated with "diencephalic" amnesia (Victor & Adams, 1993; Victor et al., 1971).
THE HIPPOCAMPUS AND DORSAL MEDIAL NUCLEUS
In addition, the hippocampus acts directly on the DMT, and on the orbital frontal lobes, and via the entorhinal cortex can act on the neocortex. In this manner the hippocampus can transmit to and keep track of where memories are stored, while simultaneously acting to store them in select neocortical regions. This is accomplished by simultaneously exerting influences on the DMT while sharing reciprocal influences with the orbital frontal lobe which also projects to the DMT.
Specifically, layer 2 of the hippocampus consists of pyramidal neurons which provide excitatory output and thus act to activate and arouse target tissues; via the transmitters glutamate and aspartic acid. In addition, the entorhinal cortex provides excitatory input into the hippocampus--input which is derived from the neocortex; using again, aspartic and glutamate acid (reviewed in Gloor, 2011). It thus appears that the hippocampus can interact with the neocortex is regard to arousal and memory storage via the dorsal medial nucleus of the thalamus and the entorhinal cortex, and can excite, for example, inhibitory circuits in the DMT (in conjunction with the frontal lobe) so as to direct neocortical activity, and perhaps coordinate this activity in regard to memory storage. In other words, these structures interact to insure that a certain neocortical area is selected for memory storage (via excitation), while simultaneously inhibiting and preventing information access in other areas. Memories can be stored and the hippocampus and frontal lobes, via the DMT, can keep track of where they are stored.
HIPPOCAMPUS & NEOCORTICAL AROUSAL
The frontal lobes, DMT, amygdala, hippocampus, entorhinal cortex, and the neocortex of the overlying temporal lobe all appear to interact in concert during various aspects of memory storage and retrieval as well as in the temporal placement of events in regard to time and even place (Brewer et al., 2008; Gloor, 2011; Graff-Radford et al. 2010; Talland, 1961; Wagner et al., 2008). This is why retrieval and even memory storage may become abnormal with damage to any of these nuclei.
There are reciprocal connections between the DMT and the amygdala, and temporal allocortex and mesocortex including the entorhinal cortex, as well as the lateral and orbital frontal lobes. This relationship suggests that the DMT upon receiving converging input, processes this material, and then at the behest of the frontal lobes, amygdala, and entorhinal cortex/hippocampus, stores this material in select areas of the neocortex such as the temporal lobe.
As discussed in chapter 13, the hippocampus exerts desynchronizing or synchronizing influences on various thalamic nuclei which in turn augments or decreases thalamic and neocortical activity (Green & Adey, 1956; Guillary, 1955; Nauta, 1956, 1958). As the thalamus is the major relay nucleus to the neocortex and is richly interconnected with the frontal lobes and amygdala, the hippocampus therefore appears able to act in concert with these nuclei so as to block or enhance information transfer to various neocortical areas where memories and perceptual experiences are presumably stored.
For example, when the neocortex becomes desynchronized (indicating cortical arousal), the hippocampus often (but not always) develops slow wave, synchronous theta activity (Grastyan et al. 1959; Green & Arduni, 1954) such that it appears to be functioning at a much lower level of arousal--at least in lower mammals. Conversely, when cortical arousal is reduced to a low level (indicated by EEG synchrony), the hippocampal EEG often becomes desynchronized and thus highly aroused (Grastyan et al., 1959; Green & Arduni, 1954). However, when this occurs, theta activity disappears and learning and memory are also disrupted--at least in lower mammals.
With the exception of the orbital frontal lobe, neocortical interconnections with the hippocampus are indirect and relayed by the entorhinal cortex,. Nevertheless, these interconnections (coupled with those of the DMT) enable the hippocampus to not only activate select regions--as its pyramidal projection system and that of the entorhinal cortex is excitatory (reviewed in Gloor, 2011)--but to sample information after it has been partially processed. This is accopmplished via the entorhinal cortex which projects back and forth from the association areas to the hippocampus. In this manner the hippocampus can also influence the processing that takes place via the DMT/Entorhinal cortex, and keep tract of what takes place as well.
The hippocampus consists of 3 layers, layer 2 consisting of pyramidal neurons which provide excitatory output. The hippocampus can act to activate and arouse target tissues; via the transmitters glutamate and aspartic acid. In addition, the entorhinal cortex provides excitatory input into the hippocampus--input which is derived from the neocortex; using again, aspartic and glutamate acid (reviewed in Gloor, 2011). The hippocampus can therefore exert significant activating influences on target tissues.
Specifically, it appears that the hippocampus interacts with the neocortex is regard to arousal via the orbital frontal lobe, dorsal medial nucleus of the thalamus, the septal nuclei, the hypothalamus, amygdala and brainstem--structures with which it maintains direct interconnections. As per the neocortex, this sheet of tissue is also innervated by these same limbic, thalamic, and brainstem structures, and by the entorhinal cortex.
Hence, the hippocampus serves as a major component of an excitatory interface and can be aroused by neocortical activity (via orbital frontal lobes, and aroused by the entorhinal cortex which provides excitatory input to the hippocampus). And the hippocampus can provide excitatory input directly to subcortical structures and indirectly to the neocortex (via the entorhinal cortex and dorsal medial nucleus) as well as directly influences the orbital frontal lobes and DMT.
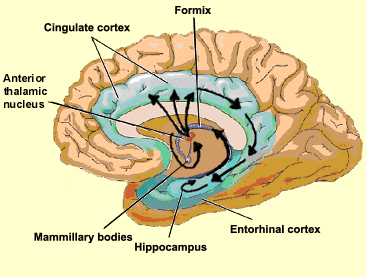
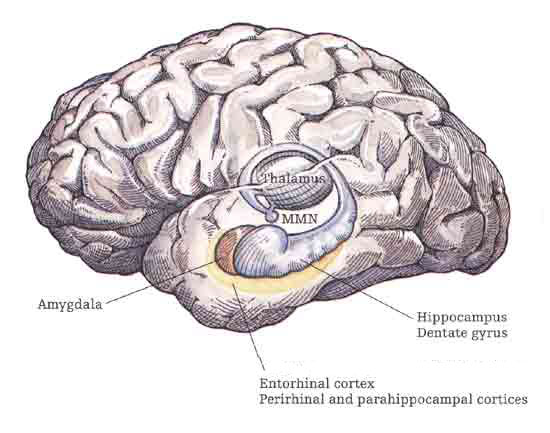
Again, presumably these reciprocal interconnections may enable the hippocampus to keep track of this activity so as to form conjunctions between different brain regions which process associated memories (Rolls, 2010; Squire, 1992). By sampling the activity occurring in different regions and via its rich, indirect interconnections with these neocortical areas, the hippocampus (or entorhinal cortex) may also be able to determine which perceptions are the most relevant and which should be stored vs inhibited so that relevant memories need not compete with irrelevant sense data (Joseph, 1992a; Rolls, 2010a).
Presumably the hippocampus acts to aid in the creation of these networks and to thus protect memory and the encoding of new information during the storage and consolidation phase, by the gating of afferent streams of information and the filtering/exclusion (or dampening) of irrelevant and interfering stimuli; i.e. by reducing or increasing arousal in select regions and via inhibitory and excitatory influences on the DMT.
In consequence, if the hippocampus is damaged or is overwhelmed, there may be input overload, the neuroaxis may be also overwhelmed, and the consolidation phase of hippocampal memory formation is disrupted such that cognitively relevant information is not properly stored or even attended to (Joseph, 2008b, 2013d; Squire, 1992). Consequently, the ability to form non-emotional associations (e.g. between stimulus and response) and to create new neural networks, or to alter preexisting cognitive schemas and neural circuits is attenuated (Douglas, 1967). These individuals appear to be amnesic.
Again, however, this is a role the hippocampus also shares with the frontal lobes, which, in humans, is clearly dominant over the hippocampus in this regard. That is, the frontal lobes have taken over many functions that the hippocampus mediates in lower mammals.
EXCESSIVE HIPPOCAMPAL AROUSAL & MEMORY LOSS
As described in chapter 13, when lower mammals are exposed to novel stimuli or when engaged in active searching of the environment, hippocampal theta appears (Adey, et al. 1960) as does LTP (Lynch, 1986). There is thus a direct correlation between hippocampal theta and the development of hippocampal LTP (Lynch et al. 2010)--at least in lower mammals. However, with repeated presentations of a novel stimulus or when familiar stimuli are presented, the hippocampus habituates and theta disappears (Adey et al., 1960). Possibly, the reason the hippocampus and other brain structures appear to respond preferentially to novel stimuli and to then cease, at least during learning tasks, is because the continual processing of familiar stimuli in short-term memory is a waste of energy and attentional space.
As noted above, when the neocortex is highly stimulated, the hippocampus (in order to monitor what is being received and processed), functions at a lower arousal level in order not to become overwhelmed. However, at extremely high levels of arousal, what is being experienced may not be learned, or it will be learned independent of the hippocampus due to diminished hippocampal activity.
In situations where both the neocortex and the hippocampus become highly aroused and desynchronized, there results distractibility and hyperresponsiveness such that the subject becomes overwhelmed, confused, and may orient to and approach several stimuli (Grastyan et al., 1959); a condition that also occurs following hippocampal lesions (Clark & Issacson 1965; Douglas, 1967; Ellen et al. 1964)--at least in non-humans. Under conditions of abnormal or incoherent cortical arousal, the ability to think or respond coherently may be disrupted as is attention, learning, and memory functioning, i.e. memories are stored haphazardly, incompletely, or not at all.
Situations inducing high levels of arousal, perceptual disorganization, and memory loss sometimes also occur when individuals are highly anxious, frightened, or emotionally upset and traumatized (chapters 2, 30), e.g. during a prolonged and brutal rape or physical assault, or during horrendous battle field conditions -in which case hippocampal participation in memory formation dramatically decreases as neocortical and limbic arousal increases (Joseph, 2008b, 2013d). If the hippocampus is overwhelmed and deactivated, the victim will experience amnesia.
For example, in cases of transient global amnesia there is evidence for temporary mesial temporal inactivation (Hodges & Warlow, 2010), including bilateral hypofusion of the hippocamus (Evans et al., 1993). These structures also become inactivated following the seizure- or electrode induced- postictal anterograde amnesia (Brazier, 1966; Chapman et al. 1967; Halgren, et al., 2012). However, these memory deficits may shrink over time (Brazier, 1966; Squire 1992), as do those following mild or moderate head injuries up to the moment of impact.
Under emotionally traumatic or stressful conditions the hippocampus may also become highly and overactivated and it may in fact be injured (Lupien & McEwen, 2011; Sapolsky, 2014). Hence, it is not at all unusual for victims to profess a complete or partial amnesia for the event. However, because the amygdala may continue to function normally the victim may later experience flashbacks, heightened startle reactions, and intrusive emotional images (chapter 30). In these instances of extreme emotional stress, other brain structures, such as the amygdala probably play a more important role in memory and learning.
THE AMYGDALA & EMOTIONAL NEURAL NETWORKS
The amygdala (although dependent on the hypothalamus) is preeminent in the control and mediation of most higher order emotional and motivational activities including the formation of emotional memories (Davis et al., 2011; Gloor, 1992, 2011; Halgren, 1992; LeDoux, 1992, 2014; Rolls, 1992) and the recall of trauma-related memories and stimuli (Rauch et al., 2014; Shin et al., 2011). Amygdaloid neuron are able to monitor and abstract from the sensory array stimuli that are of motivational significance to the organism (Gloor, 1992, 2011; Ono & Nishijo, 1992; Rolls, 1992; Steklis & Kling, 1985) as well as add emotional attributes to perceptual and cognitive activities. This includes the ability to discern and express even subtle social-emotional nuances such as friendliness, fear, love, affection, distrust, anger, etc., and at a more basic level, determine if something might be good to eat (Fukuda et al., 1987; O'Keefe & Bouma, 1969; Ono & Nishijo, 1992; Ono et al., 1980).
The lateral amygdala also has high concentrations of acetylcholine (Woolf & Butcher, 1982) as does the hippocampus (Kuhar, 1975) and acetylcholine (ACh) activity within the amygdala as well as hippocampus is correlated with learning and memory (Todd & Kesner, 1978). Indeed, a portion of the "extended amygdala"; i.e. the substantia innominata (of the limbic striatum) is a major source of neocortical ACh, and is therefore exceedingly important in memory. Alzheimers disease is associated with degeneration in the substantia innominata (see chapter 16), as well as a host of other abnormalities.
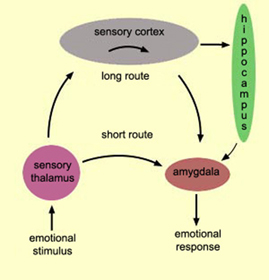
Single amygdaloid neurons receive a considerable degree of topographic input, and are predominantly polymodal, responding to a variety of stimuli from different modalities simultaneously (O'Keefe & Bouma, 1969; Perryman et al. 1987; Sawa & Delgado, 1963; Schutze et al. 1987; Turner et al. 1980; Ursin & Kaada, 1960; Van Hoesen, 1981). Hence, multi-modal integration and assimilation occurs within single amygdala neurons. Amygdala neurons are also involved in recognition memory, and the detection of faces, facial expressions, and visual stimuli involving context (Gloor, 1992; Murray, 1992; Rolls, 1992).
The amygdala, therefore is also capable of forming cross modal associations such that a neutral stimulus can come to be endowed with emotional attributes (Gaffan & Harrison, 1987; LeDoux, 1992, 2014; Rolls, 1992). This includes forming associations between primary as well as secondary reinforcers (Everitt et al. 2012; Gaffan & Harrison, 1987). In addition, there are single neurons in the amygdala which discriminate between stimuli which are rewarding or punishing (Rolls, 1992).
As noted, the amygdala is functionally related and richly (and reciprocally) interconnected with the hippocampus, inferior temporal lobe, and a variety of other limbic, brainstem, and neocortical regions that are important in memory functioning (Amaral, et al. 1992; Gloor, 1992, 2011; Halgren, 1992; Rolls, 1992). It also projects to all areas of the neocortex and receives highly processed visual, somesthetic, auditory, as well as olfactory input and is especially responsive to faces, when making eye contact, and can discriminate between males and females, strangers and those who are familiar, and can determine if someone is staring or looking away (see chapter 13).
Because of its involvement in all aspects of social-emotional and motivational functioning, activation of the amygdala therefore, can evoke highly personal and emotional memories as it is highly involved in remembering emotionally charged experiences (Gloor, 1992, 2011; Halgren, 1981, 1992; Halgren, et al. 1978; Rolls, 1992; Sarter & Markowtisch, 1985). In fact, the amygdala becomes particularly active when recalling personal and emotional memories (Halgren, 1992; Heath, 1964; Penfield & Perot, 1963), and in response to cognitive and context determined stimuli regardless of their specific emotional qualities (Halgren, 1992). Moreover, depth electrode activation of the amygdala can even evoke memories of sexual intercourse, and traumatic memories that had long ago been forgotten.
Consider, for example, a case described by Gloor (2011, pp. 7-9) in which the right amygdala of a 21 year old patient "was briefly stimulated for 2.8 seconds at a low intensity. Immediately upon its onset" the patient reported "a feeling like falling into the water. When he was asked to elaborate, he replied that it was as if something had covered his eyes, nose, and mouth." Again the stimulation was applied and the patient excitedly asked: "Could you do it again, Doctor? When asked why, he said he had the words on his lips to describe the feeling, but then could not." The stimulation was then applied without warning for 4.4 seconds. The patient "opened his mouth, with an astonished look on his face," for a terrifying memory had suddenly come back to him. It was when he was eight years old and had been at a picnic when "A kid was coming up to me to push me into the water.... I was pushed by somebody stronger than me... a big fellow..." who pushed him under the water and had kept his head under the water so that he couldn't breath.
As pondered by Gloor (2011, p. 9): "why did the first two stimulations elicit only a quasi-affective response, vaguely evocative of a past event, whereas the third brought out the reenactment or recollection of the specific event? One possibility is that because the third stimulation was 30% to 50% longer than the first two, more time was provided for initiating the activation of the neural systems encoding the experience. A second possibility is that the stimulations that were carried out a few minutes apart left in their wake some residual long-lasting neural facilitation."
As detailed in chapter 30, under conditions of prolonged fear and traumatic stress, the amygdala may process and store emotional experiences in memory in the absence of significant hippocampal participation. In consequence, the victim may become amnesic, though they may later experience a shrinking amnesia, and/or when presented with trauma-related cues, they may begin to remember, in bits and pieces, the original traumatic experience in a manner identical to that described by Gloor (2011).
FEAR, ANXIETY, STARTLE, & TRAUMATIC STRESS
The amygdala is the major site where conditioned fear, startle and autonomic reactions occur (Davis, 1992; Davis et al., 2011; Hitchcock & Davis, 2012; LeDoux, 1992, 2014) -reactions which may be stored in memory. Moreover, when recalling traumatic memories or when presented with traumatic-stimuli, the amygdala becomes highly active and subjects display all the signs of heightened autonomic activity (Rauch et al., 2014; Shin et al., 2011). Hence, in response to a certain stimulus that has been repeatedly associated with positive or negative outcomes, autonomic changes are triggered which may be accompanied by fear as well as memory recall. That is, although an emotional event may be seemingly forgotten, associated cues may trigger the remembrance of things past, including memories that may have been seemingly forgotten and "repressed." However, if the memory is not completely remembered, the patient may reexperience feelings and autonomic changes associated with the memory, as described above by Gloor (2008).
For example, the amygdala directly influences breathing and heart rate in response to emotional stimuli. It may in fact may create a circuit or neural network that triggers heart rate and autonomic changes, as well as neocortical desynchronization (low voltage fast activity) in response to a neutral stimulus that has been repeatedly associated with highly arousing pleasant or aversive emotional outcomes (Kapp et al. 1992; Ursin & Kaada, 1960). That is, emotional learning of a well defined conditioned response occurs within the amygdala which in turn influences autonomic activity such that both classical and instrumental emotional conditioning occurs.
However, this also makes possible the experience of anxiety and anticipatory fear in response to cues which might trigger the original emotional memory. Even if the original memory is not recalled, the victim may still experience the emotional and associated autonomic reactions--all of which may contribute to the development and maintenance of post-traumatic stress disorder (see van der Kolk, 1987).
In these particular instances, however, the more the cue is linked and associated with the original traumatic event, the more severe might be the reaction, and the more upsetting the recalled memory. Kolb (1986, p. 172), for example, in his discussion in the persistence of startle reaction in combat soldiers, noted that "it was remarkable 10, 15, or 20 years after the combat event to hear men tell about suddenly hearing a loud noise reminding them of battle sounds and then being in a panic state."
Kolb (1986, p. 172) also reports that when they "played a tape consisting of combat sounds, including those of helicopters, mortars, and screaming wounded... the patients immediately returned to a critical event in the battlefield. Many then experienced and reenacted scenes of fight, flight, or rage." For most of these individuals, reexperiencing the traumatic memory is, initself extremely traumatic and they avoid all forms of stimulation that can reawaken these memories.
Hence, an individual may become severely stressed or repeatedly frightened, and later experience an increase in heart rate, changes in breathing patterns, and other autonomic alterations, in anticipation or in response to associated emotional and contextual cues, which in turn may activate the neural network in which the original emotional memory is stored. However, at least in regard to heart rate changes, this circuitry is in part hard wired and involves a projection into the midbrain periaqueductal gray (as well as the basal ganglia) and then on to the lower brainstem into the vagal nucleus (Kapp et al. 1992).
According to Davis (1992, p. 283), "much of the complex behavioral pattern seen during fear conditioning has already been hard wired during evolution. In order for a formerly neutral stimulus to produce the constellation of behavioral effects used to define a state of fear or anxiety, it is only necessary for that stimulus to now activate the amygdala, which in turn will produce the complex pattern of behavioral changes by virtue of its innate connections to different brain target sites." Hence, the amygdala need only learn what stimuli are the most arousing, threatening, fearful, pleasurable and so on, and can then automatically trigger autonomic and behavioral responses which are already hard wired into the nervous system.
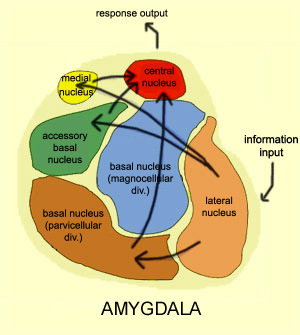
Amygdala activation will also increase the frequency of respiration and will trigger behavioral freezing and an arrest of ongoing behavior (Kapp et al. 1992; Ursin & Kaada, 1960). As detailed in chapter 16, the amygdala may also contribute to the frozen states common among those with Parkinson's disease. Initially, however, freezing behavior is part of the attention response, which in turn may be followed by an orienting reaction, sometimes with anxious glancing about, and/or with an approach or withdrawal reaction (Gloor, 1960; Ursin & Kaada, 1960). If amygdala stimulation continues fear and/or rage reactions are elicited (Ursin & Kaada, 1960).
In general, the right amygdala is more involved than the left in the production of fear and anxiety (Davis, 1987), particularly free floating fears (chapter 13), and the right hemisphere is more greatly involved in the fear potentiated startle response (Lang et al. 2010), as well as emotion in general (chapter 10).
FEAR, ANTICIPATION, & REPRESSION
As noted, the amygdala can make fine discrimination between stimuli that are closely vs not so closely associated with fear, so that startle and fear reactions are elicited to stimuli which most closely match (Davis, 1992). The amygdala, therefore, can also anticipate fearful stimuli based on only fragmentary cues or sensory stimuli, some of which might trigger startle reactions (Davis, 1992; Davis et al., 2011; Kurtz & Siegel, 1966; LeDoux, 2014) or memory recall.
The amygdala is thus very important in the production of anticipatory anxiety (Davis, 1992) as well as emotional memory (Gloor, 2011, Halgren, 1993; LeDoux, 2014; Rauch et al., 2014; Shin et al., 2011). Because it can anticipate based merely on fragmentary cues, individuals may come to feel upset, angry, frightened, and yet not know why or what might have triggered these emotions. This often occurs among individuals suffering from post traumatic stress disorder, as well as among those with repressed memories; i.e. a consequence of a single cue triggering a complex emotional memory or associated feelings of anxiety (van der Kolk, 1987).
In some respects this is a very adaptive arrangement, for if the individual were unable to inhibit or forget emotionally upsetting memories, their intrusion would crippled his or her ability to function. That is, perhaps it makes little evolutionary sense for an indivdual to be able to recall and act on extremely arousing and emotionally upsetting memories except when in a highly aroused state. For these memories to seep or burst into consciousness at other times may exert a deleterious effect on functioning --which appears to be the case in post-traumatic stress disorder.
Perhaps it is adaptive for the hippocampus to sometimes cease to participate in memory formation under extremely distressful conditions, and why only fragments of the emotional trauma may appear in consciousness under certain similar circumstances and conditions. From the standpoint of survival it makes sense for these memories to be forgotten but then recalled when in a similar state of high arousal or when presented with similar emotional cues. However, even under these conditions the forgotten memory may remain "repressed," and this too may be made possible via the amygdala (as well as the frontal lobes).
For example, the amygdala in response to a partial cues may respond with feelings of anxiety and fear before the actual memory or associated neural network can be fully activated (Gloor, 2011). In instances such as these, the actual memory might come to be inhibited, or repressed, in response to the anxiety that was triggered in advance.
FEAR, EMOTIONAL TRAUMA, HIPPOCAMPAL DEACTIVATION & MEMORY LOSS
Rhythmic slow activity (theta), is generated by both the hippocampus and entorhinal cortex (Alonso & Garcia-Austt, 1987) and theta has been found in the hippocampus of most species studied, including monkeys (Stewart & Fxx, 2010) and humans (Sano et al., 1970); though in primates it seems to differ from the theta rhythm of non-primates (see Gloor, 2011). Theta is an indication of hippocampal arousal (Green & Arduini, 1954; Petsche et al., 1965; Vanderwolf, 1992) and is associated with learning and memory (O'Keefe & Nadel, 1978). Moreover, LTP is generated in those neurons demonstrating theta or activity that is at the "theta frequency" (Staubli & Lynch, 1987).
Neurons of the septal nucleus which innervate the hippocampus fluctuate in activity in parallel with changes in the theta rhythm (Petsche et al., 1965). The septal nucleus, in fact, acts as an interface between the reticular formation and the hippocampus (Petsche et al., 1965) and thus appears to contribute to the development of theta by modulating hippocampal arousal thus promoting learning and memory. Hence, if the septal nucleus is lesionsed hippocampal theta is abolished (Green & Arduini, 1954) and significant memory deficits are produced. Moreover, the septal nuclei is directly implicated in the stress response and can be injured by prolonged stress (see chapter 30); conditions which also result in the loss of theta and LTP.
In response to continued, ongoing fear, emotional stress, and anxiety, the amygdala (as well as the HPA axis) begins to secrete large amounts of the amino peptide, corticotropin-releasing factor (CRF) which in turn potentiates the behavioral and autonomic reaction to fear and stress (see chapter 30). Cortisone levels also increase dramatically when stressed by restraint (see Davis, 1992) and amygdala activity will increase as well (Henke 1992). Moreover, high levels of cortisone exerts an inhibitory influence on the hippocampus and eliminates hippocampal theta by attacking hippocampal pyramidal cells (Joseph, 2008d, Spoont, 1992; see chapter 30) which normally provide excitatory output. In consequence, memory begins to decline (Lupien & McEwen, 2011; Sapolsky, 2014).
When fear follows the attention response, the pupils dilate and the subject will cringe, withdraw, and cower -which is associated with high levels of amygdaloid activity. This cowering reaction in turn may give way to extreme fear and/or panic and the animal will attempt to take flight. This may be accomplished via amygdala activation of the locus coeruleus which would release norepinephrine (NE), and activation of the raphe nucleus which would release serotonin (Magnuson & Gray, 2010), both of which facilitate motor responsiveness (White & Neuman, 1980). However, if arousal levels continue to increase, the hippocampus may cease to participate in memory functioning such that whatever is experienced and learned may subsequently appear repressed due to hippocampal deactivation (see chapter 30 for further details).
Among humans, the fear response is one of the most common manifestations of amygdaloid electrical stimulation (Gloor, 2010; Halgren, 1992; Williams, 1956). Moreover, unlike hypothalamic on/off emotional responses, attention and fear reactions can last up to several minutes after the stimulation is withdrawn. Apparently this is associated with the development of LTP within amygdala neurons which thus continue to reverberate even when the subject is frightened or in pain.
In addition, when the (human) amygdala is abnormally or highly activated, it is able to possibly deactivate the anterior hippocampus as well as overwhelm the neocortex and the rest of the brain so that the person not only forms emotional ideas (that later cannot be fully remembered) but responds to them in an abnormal manner.
In fact, during periods of extreme fear, such as when a subject freezes, hippocampal theta disapears and is replaced by irregular electrophysiological activity (Vanderwolf & Leung, 1983). Prolonged stress also inhibits long term potentiation in the hippocampus (Shors et al. 1989). Theta also disappears if a subject is placed in a situation where they are completely hopelessly surrounded by painful physical threat, such as an electrified grid floor (Vanderwolf & Leung, 1983). Similarly, under conditions of stress or when the body has been anesthetized (Green & Arduini, 1954), hippocampal theta disapears, as does LTP (Shors et al. 1989) and the threshold for hippocampal responding is significantly raised. In part it is overwhelmed and inhibited by the amygdala and the reticular activating system, which in turn reduces its participation in learning and memory.
The hippocampus maintains a reciprocal relationship with the reticular formation in regard to the modulation of neocortical activity except under conditions of high arousal in which case it can be inhibited by the reticular activating system (Adey, et al. 1957; Green & Arduini, 1954; Redding, 1967). This may occur in situations involving high levels of arousal and with repetitive stimulation (Redding, 1967) in which case theta and LTP may be prevented. Indeed, with persistent and repeated stimulation, instead of excitation there is synaptic depression with no evidence of LTP even under neutral and non-emotional conditions (Lynch, 1986). Presumably this is related to rapid calcium depletion, for LTP and synaptic plasticity are effected by calcium levels.
This loss of hippocampal theta and LTP under repetitive, stressfull, painful, or fearful conditions is not a function of lack of whole body movement, however. For example, theta appears when human subjects are sitting motionless but are mentally engaged in problem solving or learning tasks (Meador et al. 2012) and when the median raphe nucleus (which suppresses hippocampal theta and induces high states of arousal) is destroyed and subjects are immobilized (Vertes, 1981). Rather this loss of theta activity is a function of overwhelming fear and high levels of arousal, for although seemingly alert, the hippocampus is desychronized, and the subject may seem frozen and will be unable to respond to environmental stimuli, be it threat or painful stimuli (Redding, 1967). In consequence they are unable to recall what occurred as this data was stored by the amygdala in the absence of hippocampal participation.
Later, hippocampal retrieval mechanisms are unable to find the memory. The patient will appear amnesic when they are in fact relying on the wrong (albeit "normal") retrieval mechanisms; i.e. the hippocampus.
Indeed, under conditions of high hippocampal arousal or inhibition (Evans et al., 1993; Halgren, et al., 2012; Hodges & Warlow, 2010), or due to direct hippocampal stimulation, verbal amnesia results (Brazier, 1966; Chapman et al. 1967) and there is a loss or significant reduction in theta (Redding, 1967). This occurs even when there has been no loss of consciousness, and while the subject seems fully alert with no evidence of confusion.
Normally, as described by Squire (1992, p. 224, 208), "the possibility of later retrieval is provided by the hippocampal system because it has bound together the relevant cortical sites. A partial cue that is later processed through the hippocampus is able to reactivate all of the site and thereby accomplish retrieval of the whole memory... In the absence of the hippocampus, representations that had been established in short-term memory are literally lost, become disorganized, or achieve some abnormal fate."
As detailed in chapter 30, memories formed by the amygdala in the absence of hippocampal participation, such as in instances of extreme emotional stress and neocortical arousal, may appear to be forgotten as they cannot be accessed via normal hippocampal retrieval mechanisms. However, even when the hippocampus has been destroyed, this amnesia does not extend to fear or pain related stimuli (Seldon et al.2012). In fact, aversive conditioning is completely unaffected (Seldon et al. 2012), particularly when the conditions are highly aversive and painful or highly emotional as this form of learning is mediated by the amygdala (Cahill & McGaugh, 2010; Hitchcock & Davis, 1987; Kesner & DiMattia, 1987) as well as the hypothalamus (see chapter 13). Rather, what may be forgotten is the place and the circumstances around which the pain was experienced (Seldon, et al. 2012).
However, although the hippocampal amnesia may extend backward in time from minutes to days and even weeks, the amnesia may shrink over the course of the next several hours such that only those events which took place just prior and during the period of abnormal hippocampal excitation are seemingly forgotten (Brazier, 1966; Chapman et al. 1967). Nevertheless, when specific cues are provided, recall and memory recovery occurs (Seldon, et al. 2012). Recognition memory is activated and what was forgotten is suddenly recalled -sometimes to the disbelief of family or treating physicians.
MEMORY LOSS & AMYGDALA DYSFUNCTION
As discussed in chapter 13, the amygdala appears to be involved in all aspects of social-emotional memory functioning. Hence, destruction of the amygdala makes it exceedingly difficult to form associations between reinforcing stimuli, regardless of their being positive or negative (Jones & Mishkin, 1972; LeDoux, 1992; Rolls, 1992; Weiskrantz, 1956), and results in a dense social-emotional agnosia and amnesia such that even loved one's are no longer recognized. Similarly emotional learning becomes very limited, primitive and dependent on the hypothalamus (chapter 13).
For example, animals who have been amygdalectomized often demonstrate very poor performance on emotional learning and attention tasks (Kesner, 1992; Murray, 1992; Sarter & Markowitsch, 1985) as they are unable to assign emotional attributes to what they perceive. They also cannot learn associations between primary reinforcers and rewarded stimuli, including those where the subject has to approach or avoid stimuli which has either been rewarded or punished (Kesner, 1992; Murray & Mishkin, 1985; Rolls, 1985, 1992).
Neurosurgical removal of the right or left amygdala also results in significant memory disturbances, particularly in the realm of information retrieval and social-emotional perception and contextual information storage (Andersen, 1978; Gloor, 1992; Rolls, 1992; Sarter and Markowtisch, 1985). Moreover, a complete amnesia can result following electrical stimulation of the amygdala (Chapman, 1958; Jasper & Rasmussen, 1958), and difficulty recognizing people and objects has been repeatedly noted following amygdalectomy in humans (Sawa et al. 1954).
Moreover, Alzheimer's disease as well as other dementias and encephalitic conditions not only result in severe memory loss, but often attack the amygdala and the anterior commissure (which links the right and left amygdala) creating both neurofibrillary changes and senile plaques (see Mann, 1992; Sarter & Markowitsch, 1985, for review). With normal aging there is also a progressive memory loss and a significant drop out of neurons in the amygdala (Herzog & Kemper, 1980), as well as in other structures, particularly the substantia innominata which is part of the "extended amygdala" and which is directly implicated in the genesis of Alzheimer's disease.
AMYGDALA, HIPPOCAMPUS & DORSAL MEDIAL NUCLEUS
Both the hippocampus and amygdala maintain rich interconnections with the anterior thalamus (Ammaral et al. 1992; Krettek & Price, 1977; Nauta, 1971; Graff-Radford, et al. 2010). However, these two limbic pathways remain distinct and terminate in separate, albeit adjacent thalamic areas (Graff-Radford, et al. 2010). Indeed, a similar pattern of parallel organization characterizes amygdala and hippocampal input into the basal ganglia and other limbic and brainstem nuclei (see chapters 13, 17).
This suggests that the influences and contributions of the amygdala and hippocampal to memory, motor, and perceptual activity remain distinct within the brainstem, basal ganglia, and the neocortex such that perhaps two separate, albeit closely aligned neural circuits are created in parallel; one concerning the emotional the other the cognitive attributes of what has been attended to, learned, and stored in memory.
It is also noteworthy that although destruction of the anterior and dorsal medial thalamus can induce severe verbal and visual amnesic disorders, it appears that both the amygdala and hippocampal pathways must be destroyed for this to occur (Graff-Radford et al. 2010). Presumably this is because the amygdala and hippocampus interact at the level of the limbic system, and again within the thalamus, and probably again within the neocortex to insure that certain memories are formed. Hence, damage to the DMT may result in a disconnection syndrome. That is, the interacting influences of the hippocampus and amygdala is no longer possible within the DMT which negatively impacts memory formation within the neocortex.
In contrast, thalamic lesions which are more caudally located and which spare these limbic pathways, do not induce amnesia (see Graff-Radford et al. 2010; von Cramon et al. 1985). Because these limbic-thalamic pathways have been spared, the hippocampus and amygdala are able to continue to exert tremendous influences on neocortical information processing, including the gating of perceptual activity and neocortical arousal and the filtering vs selective attention to events which are emotionally, motivationally and cognitively significant.
These distinct, albeit co-dependent (emotional vs cognitive) memory traces are also probably mutually reinforcing and linked to some degree. However, because they are separate, it is possible to recall the cognitive attributes of an experience in the absence of its emotional significance, or conversely, to recall the emotional features of an event in the absence of memory for the event; e.g., an individual may recall having a horrible argument but be unable to remember what was said. In severe cases they may become severely amnesic.
OVERVIEW: THE AMYGDALA, HIPPOCAMPUS, & MEMORY
There is considerable evidence which strongly suggests that the hippocampus plays an interdependent role with the amygdala (and the frontal-thalamic system) in regard to many aspects of memory (e.g. visual, verbal, spatial), and that the amygdala plays a primary role in regard to all aspects of emotional memory. That is, the amygdala (in conjunction with the hypothalamus) extracts and stores the emotional attributes as well as mediating the emotional reactions and feelings triggered by particular events. It also acts on the anterior hippocampus and dorsal medial thalamus so that motivationally and emotionally significant information is attended to and stored via the creation of appropriate neural networks. The hippocampus performs likewise with verbal, visual-spatial, and cognitive material. Thus, these two nuclei work in concert.
The amygdala seems to reinforce and maintain hippocampal activity via the identification of social-emotional and motivationally significant information and the generation of pleasurable rewards -through interaction with the lateral hypothalamus and by "rewarding" the anterior hippocampus (chapter 13). That is, reward increases the probability of attention being paid to a particular stimulus or consequence as a function of its association with reinforcement (Douglas, 1967; Kesner & Andrus, 1982).
The same is true when an experience is personally significant and moderately emotionally arousing. It is perhaps for this reason that personal emotional events are often better recalled than those which are neutral and why learning and memory improves when rewarded; i.e. amygdala-hippocampal interactions are enhanced which leads to the establishment of cognitive-emotional neural networks and thus long term memory. Conversely, when both the amygdala and hippocampus are damaged, striking and profound disturbances in memory functioning result (Kesner & Andrus, 1982; Mishkin, 1978, 2010).
There is also evidence which indicates that these emotional and cognitive memories may be stored in separate, albeit aligned neural networks. As noted, the pathways leading from the amygdala and hippocampus to the anterior and dorsal medial thalamus terminate in separate, albeit adjoining thalamic neurons (Graff-Radford, et al. 2010) as well as in adjacent basal ganglia and brainstem neurons. Thus these two networks of emotional vs cognitive "memories" and associated neural pathways remain separate and may terminate on different dendrites and neurons. Hence, cognitive and associated emotional memories are maintained by semi-independent neural networks which might be activated or inhibited separately, partially, sequentially or in parallel, and conjointly.
Also, as noted, the amygdala and hippocampus act differentially in regard to the effects of positive vs. negative reinforcement. For example, whereas the hippocampus produces theta in response to noxious stimuli it soon ceases to participate in memory formation when negative conditions are continuous and ongoing, i.e., theta disappears (Vanderwolf & Leung, 1983) as does hippocampal LTP (Lupien & McEwen, 2011; Shors et al. 1989). Similarly, Heath (1977) reports that high emotional states, including fear, and rage, are associated with high amplitude spindle EEG wave forms within the hippocampus. In contrast, the amygdala increases its activity following the reception of a reward and in response to stressful stimuli (see Gaffan, 1992; Halgren, 1992; Rolls, 1992). It also develops LTP under these conditions.
The amygdala also mediates behavioral responsiveness under conditions of high arousal, so as to maintain attention, or to attack in anger (or hunger) or run away in fear. Hence, whereas the hippocampus diminishes its contribution under these highly arousing conditions, the amygdala continues to subserve perceptual and behavioral activity, and continues to monitor and store the emotional attributes in memory, even in the absence of hippocampal participation. From the standpoint of survival, this makes evolutionary sense. Consequently, later this information may not be accessible via normal "hippocampal retrieval strategies" and may appear to be forgotten or repressed.
This neurological relationship is quite adaptive for in some life threatening situations an immediate emotional reaction is called for and the recall and consideration of cognitively based alternatives might result in death. Hence, the hippocampus ceases to participate. Moreover, for much of limbic system and human evolution, under highly stressful and emotionally arousing conditions, the expression, consideration and storage of thought patterns and other cognitive processes may have been irrelevant and might even have interfered with the retrieval of survival related information. Consequently, irrelevant information is not stored in memory, whereas when highly emotionally aroused, relevant memories might be suddenly recalled.
In fact, as individuals may not be capable of (verbally)-thinking too clearly under certain highly stressful and fearful conditions, the generation and storage of these thoughts might result in their latter recollection when in a similar situation (which, for almost all of our ancestry was probably likely), thus disrupting the ability to engage in effective action (such as fleeing or fighting) or to recall the last successful strategy.
However, these emotional memories may be suddenly recalled if the person is later confronted with similar emotional and contextual cues; which enables them to act in the same manner which previously saved their life. This may also account for the effect of certain contextual cues in the elicitation of traumatic stress reaction; i.e. hearing a car backfire and suddenly dropping to the floor in response to a "flashback" of being in battle. That is, the amygdala responds to associated cues to trigger the recollection of associated emotional memories and relevant (and life saving) behaviors.
It is presumably due to these separate neural networks and the differential contributions of the amygdala and hippocampus, that a cognitive memory might be retrieved or recalled stripped of its original emotional attributes (which may be too troubling and upsetting to remember), or why emotional memories might be triggered in the absence of concrete auditory and visual cognitive cues. As such, an individual may feel exceedingly upset, anxious, angry, and so on, and not know why (chapters 2, 10, 13), or conversely verbally recall certain traumatic or negative events which are expressed and verbally described in neutral or even positive terms (see chapter 30), though the memory is actually quite negative if not traumatic. However, in some cases the individual may "recall" nothing at all and may not even be aware that something unpleasant has been repressed.
Again, this is very "adaptive" for otherwise an individual may be plagued by memories that may best be forgotten. Indeed, although resulting in modern day "neurosis" and brain damaged induced amnesias, it is presumably this dual amygdala-hippocampal relationship and the capacity to separately store and selectively recall emotional vs cognitive information which has promoted not just learning and memory, but individual and species survival (if not sanity) for much of human history.












