Rhawn Joseph, Ph.D.
BrainMind.com
The frontal lobes serve as the "Senior Executive" of the brain and personality, acting to process, integrate, inhibit, assimilate, and remember perceptions and impulses received from the limbic system, striatum, temporal lobes, and neocortical sensory receiving areas (Fuster 1997; Joseph 1986a; Koechlin et al., 2017; Milner and Petrides 1984; Passingham 1993; Selemon et al. 2015; Shallice and Burgess 1991; Stuss 1992; Stuss and Benson 1986; Strub and Black 1993; Van Hosen et al., 1996).
Through the assimilation and fusion of perceptual, volitional, cognitive, and emotional processes, the frontal lobes engages in decision making and goal formation, modulates and shapes character and personality and directs attention, maintains concentration, and participates in information storage and memory retrieval (Dolan et al., 1997; Joseph, 1986a, 1988a, 2017a; Kapur et al., 2015; Passingham, 1997; Posner & Raichle, 1994; Tulving et al., 1994).
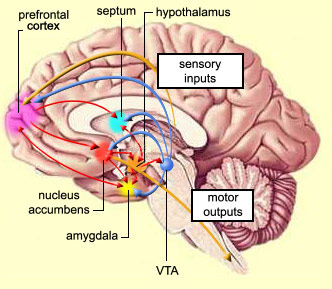
Indeed, the frontal neocortex is "interlocked" with the limbic system, striatum, and the primary and secondary receiving areas via converging and reciprocal connections, and receives verbal and ideational impulses transmitted from the multi-modality associational areas including Wernicke's area and the inferior parietal lobule (Cavada 1984; Fuster 1997; Jones and Powell 1970; Goldman-Rakic 2015, 1996; Passingham, 1993, 1997; Pandya & Yeterian 1990; Petrides & Pandya 1988). It is thus able to act at all levels of information analysis, including, via the orbital frontal lobes, at the level of the limbic system so as to inhibit and regulate emotional, sexual, and related behaviors.
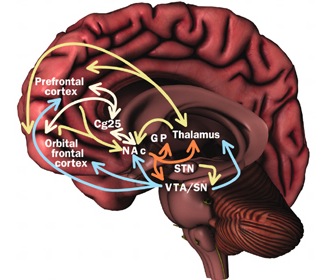
The orbital frontal lobes act as the Senior Executive of the social-emotional brain and exert tremendous inhibitory as well as expressive influences on emotion and generalized arousal through its massive interconnections with various limbic nuclei, the dorsal medial nucleus of the thalamus and the reticular formation.
When the orbital area is injured all aspects of emotional and inhibitory behaviors may be compromised, and patients may display disinhibition, manic-excitement, and internal utilization behaviors (increased sexuality, orality).With complete destruction of the orbital area, emotional and social functioning is abolished, but with less extensive damage, rather than a loss of emotion there is a loss of emotional control.
With massive injuries localized to the orbital/inferior areas, and/or which functionally disrupts the frontal-striatal pathways, patients may have difficulty inhibiting unwanted movements or thoughts, and may develop perseverative, obsessive compulsive abnormalities (Baxter et al. 1987; Breier et al. 2012; Buchanan et al. 1993; Iverson and Mishkin 1970; Luria 1980; Malloy, et al 2013; Rappoport 2011; Rauch et al. 1994; Swayze et al. 1992).
The orbital region also seems to coordinate and integrate emotional and perceptual activity. For example, fibers from the sensory association areas including the inferior parietal lobule project into the orbital frontal cortex. Hence, the orbital region is continually informed as to the perceptual processing being performed in the neocortex and can thus integrate and assign emotional-motivational significance to cognitive impressions; the association of emotion to ideas and thoughts. It is in this manner that thoughts can come to be upsetting or emotionally arousing. It is also in this manner that disturbances such as anxiety can result (Gray, 1981).
THE ORBITAL FRONTAL LOBES AND INFERIOR CONVEXITY
The Orbital Frontal Lobes
The orbital frontal lobes receive higher order sensory information from the sensory association areas throughout the neocortex and maintain rich interconnections with the lateral convexity, anterior cingulate, inferior temporal and inferior parietal lobes, the medial walls, the parahippocampal gyrus (Fuster, 1980, 1997; Johnson et al. 1968; Jones & Powell, 1970; Pandya & Kuypers, 1969; Van Hoesen 2015; Van Hoesen et al. 1975, 1996) and with the hypothalamus and amygdala (see chapter 13). Presumably, because of its extensive limbic interconnections, and the fact that it evolved from the limbic system, neurons in the orbital frontal lobes increase in activity when presented with rewarding stimuli (Tremblay & Schultz, 2017).
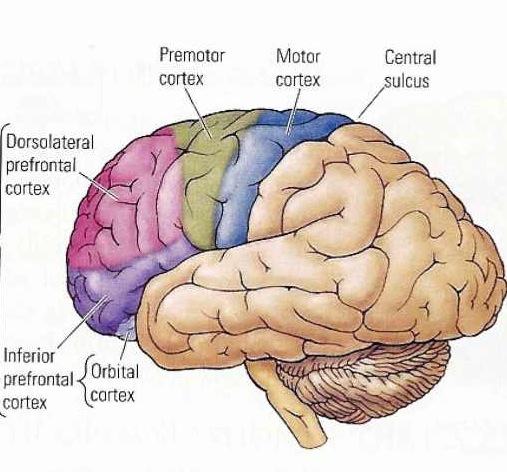
The orbital cortices also sends fibers directly to the reticularis gigantocellularis of the reticular formation, including the reticular inhibitory regions within the medulla and the excitatory areas throughout the pons (Kuypers, 1958; Rossi & Brodal, 1956; Sauerland et al., 2016). There are also rich interconnections with the medial magnocellular dorsal medial nucleus of the thalamus (Pribram et al., 1953; Siegel et al., 2007), a major relay nucleus involved in the gating and filtering of information destined for the neocortex (Skinner & Yingling, 1977; Yingling & Skinner, 1977) and limbic system.
The medial magnocellular dorsal nucleus of the thalamus appears to exert modulating influences on information reception and processing as well as limbic arousal, and in this manner the orbital frontal lobes are also able to act to modulate activity in widespread areas of the brain, the limbic system in particular.
For example, because the inferior-orbital areas are involved in inhibitory functions (Konishi, et al., 2017), this area of the brain can selectively activate or inhibit specific cortical and subcortical tissues, and can therefore discriminate between salient vs irrelevant stimuli (Joseph, 2007a; Tremblay & Schultz, 2015). For example, Tremblay and Schultz (2017, p. 704), found that the orbital frontal lobe is not only crucially involved in the motivational control of goal-directed behavior, but that orbital neurons can discriminate between stimuli which are more or less rewarding, and that these discriminations "reflect the animal's relative preference among available rewards, as expressed by their choice behaviour, rather than intrinsic physically rewarding properties."
Because of its role in regulating arousal and the limbic system, as well as its involvement in discriminating between rewarding vs non-rewarding stimuli, even minor damage to the orbital tissues is often accompanied by rather severe alterations in emotion, personality, arousal, motivational, and inhibitory function, due in part to the loss of inhibitory control over the limbic system and other forebrain structures as well as the destruction of orbital neurons.
ORBITAL MEDIATION OF LIMBIC AROUSAL
The medial portion of the magnocellular dorsal medial thalamic (MDMT) nucleus (like the orbital region) receives fibers from the reticular formation and amygdala (Aggleton, 2012; Chi, 2010; Krettek & Price, 1974; Siegel et al. 2018). However, this portion of the dorsal medial thalamus is subject to orbital control (Joseph 2017a). Indeed, the orbital region via it's interconnections with all three regions (i.e. reticular formation, limbic system, MDMT), is able to exert a considerable degree of influence on the interactions which take place in these nuclei, including control over various forms of limbic, behavioral and emotional arousal.
For example, electrical stimulation of the orbital area can cause a hungry animal to stop eating, walk away from its dish, lie down, and even fall into slow-wave synchronized sleep (Lineberry & Siegel, 2001). It can inhibit monosynaptic spinal reflexes (Clemente et al., 1966; Sauerland et al., 1967), as well as reduce and inhibit arousal throughout the neocortex (Lineberry & Siegel, 2001), limbic system (Steriade, 1964), including the reticular formation (Lineberry & Siegel, 2001; Siegel & Wang, 1974). Indeed, the orbital region appears to exert hierarchical control over the MDMT, reticular formation, limbic and autonomic nervous system, thus mediating generalized arousal throughout the neuroaxis. As pertaining to emotional arousal, it has also been postulated that the orbital area exerts a major influences on the experience of anxiety (Gray, 1981) including the development of ulcers (Freeman & Watts, 1942).
AUTONOMIC INFLUENCES
The orbital frontal lobes participate in the regulation of the autonomic nervous system. Electrical stimulation of the orbital cortex can slow or arrest respiration, alter arterial blood pressure, inhibit pyloric peristalsis, increase salivation, dilate the pupils, decrease gastric motility, and increase skin temperature (Bailey & Sweet, 1940; Chapman et al. 1950; Kaada, 1951, 1972; Livingston et al. 1948; Wall & Davis, 1951).
Hence, when the orbital cortex is severely damaged or following an orbital lobotomy, the autonomic nervous system is liberated from higher order inhibitory control (Rinkel et al. 1950). Blood pressure and skin temperature are lowered, sweating is increased, and widespread disturbances involving gastrointestinal activities, salivation, micturation, diuresis, and sexual arousal can occur (Bailey and Sweet 1940; Chapman et al. 1950; Delgado and Livingston 1948; Kaada 1951, 1972; Langworthy and Richter 1939; Mettler et al. 1936; Rinkel, et al. 1950; Wall and Davis 1951).
Urinary bladder control is also diminished resulting in a disturbance of micturation (Bianchi 1922; Fulton, Jacobsen, Kennard 1932; Freeman and Watts 1942; Langworthy and Richter 1939; Greenblatt 1950; Rose 1950; Watts and Fulton 1934). It is not uncommon for those with orbital injuries to freely urinate on themselves. Patients may urinate or even defecate while lying in bed, sitting in front of the T.V. during dinner, or while sitting in a restaurant. One patient was observed to empty her bladder while waving goodbye to a friend. She stopped on request but completed the act as she walked to the toilet (Rose 1950). In fact, the bladder may become spastic and hypertonic (Rinkel et al. 1950).
ALTERATIONS IN APPETITE
As noted, orbital neurons respond selectively to rewarding stimuli, and show particular food preferences (Tremblay & Schultz, 2017). With bilateral or anterior orbital damage this discriminatory capacity may be abolished and appetite may become exceedingly excessive and patients (or animals) may eat two or three times their normal amount (Bianchi, 1922; Anand, Dua, & Chhina, 1958; Fulton et al., 1932; Langworthy & Richter, 1939). One patient I examined who suffered a series of small bilateral strokes gained over 150 pounds within 8 months.
A second patient with a degenerative disturbances involving the inferior medial and orbital area, in addition to perseveration and compulsive utilization, gained over 200 pounds within a year and a half. Indeed, it has been reported that in some patients the craving for food, became so intense they repeatedly crammed their mouths with food, became cyanotic, and died with their mouth still full (Greenblatt, 1950).
In other instances patients might eat non-nutritive objects such as cigarette butts or drink enormous amounts of fluid. Hence, hyperphagia extreme orality, even coprophagia may occur (Butter & Snyder, 1972; Butter, Snyder, & McDonald, 1970). Similar disturbances result from hypothalamic/limbic damage.
APATHY & EMOTIONAL UNRESPONSIVENESS
Usually immediately following orbital or surgical destruction of the frontal lobes (e.g. frontal lobotomy), there results severe reductions in activity and emotional/motivational functioning, including apathy, indifference to loud noises or threats, as well as extreme reductions in arousal and motor functioning (Butter et al., 1970; Freeman & Watts, 1942, 1943).
With extensive orbital destruction involving portions of the medial wall and anterior tip of the cingulate, reduced responsiveness persists and patients will sit quietly and silently nearly motionless, making little or no attempt to communicate as if mute. The ability to respond socially and emtionally seems abolished. Rather, if sufficiently stimulated humans and animals seem capable of reacting only in an irritable and aversive manner (Butter et al. 1968) and many may appear depressed (Grafman et al. 1986).
In free ranging monkeys as well as those reared in enclosed settings, global social disintegration is observed following orbital destruction. Animals cease to groom or produce appropriate vocalizations, adults attempt to completely avoid members of their social group, and mothers neglect and rebuff their infants (Myers, Swett, & Miller, 1973; Raleigh, 1976, cited by Kling & Steklis, 1976). Similarly, human females with surgical destruction of this vicinity will neglect and/or strike or beat their children without provocation (Broffman, 1950).
In the free ranging situation, orbital animals in fact ran away from their social groups and remained solitary until their deaths (Myers et al., 1973). In contrast, lateral convexity lesions do not result in changes involving social proximity, grooming, or bonding (Kling & Mass, 1974). Hence, complete orbital destruction results in complete abolition of most forms of emotional and social behavior.
EMOTIONAL DISINHIBITION
With less extensive damage, or as swelling and ischemia become reduced so that neighboring structures are not impacted, this initial unresponsive phase may pass. Rather than a loss of emotion there is a loss of emotional control and the subject becomes disinhibited, hyperactive, euphoric, extroverted, labile, overtalkative, and develops perseveratory tendencies (Butter, 1969; Butter et al., 1970; Daz et al. 1948; Greenblatt, 1950; Kennard et al. 1941; Kolb et al. 1974; Reitman, 1946, 1947; Ruch & Shenkin, 1943). Patients are frequently described as markedly irresponsible, antisocial, lacking in tact or concern, having difficulty planning ahead or foreseeing consequences, and suffer from generalized disinhibition. There can result tendencies toward impulsive actions, to laugh inappropriately and make trivial jokes, or to behave in a demanding or transiently aggressive manner. Proneness to criminal behavior, promiscuity, gradiosity, and paranoia have also been observed (Blumer & Benson 1975; Benson & Geschwind, 2001; Lishman, 1973; Luria, 1980; Stuss & Benson, 1984). In this regard it is noteworthy that murderers show reductions in frontal lobe glucose metabolism (Raine et al. 2004).
Much of this inappropriate behavior is a consequence of a loss of control over reticular and limbic structures. That is, the orbital region appears as a kindof "censor", or even "conscience", such that when these "superego"-like influences are removed, there results heightened levels of generalized and emotional arousal and the patient responds in a childish, inappropriate and unrestrained manner (Joseph 1986a, 2017a).
With less extensive damage, or when the lesions are confined to one hemisphere the long term effects are less drastic. However, right orbital damage seems to result in the most severe alterations in mood and emotional functioning (Grafman et al. 1986).
THE INFERIOR & ORBITAL FRONTAL LOBES
DISINHIBITION & RESPONSE SUPPRESSION
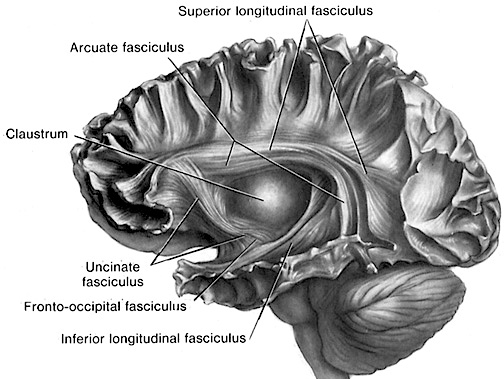
The inferior and lateral convexity appears to be highly involved in the inhibition of behavior and the ability to withhold or delay responses, which in turn is in part a function of its involvement in controlling neocortical perceptual activity and arousal (Como et., al, 2009; Fuster, 1997; Joseph et al. 1981; Joseph 2017a). Hence, electrophysiological analysis of cellular activity within the lateral convexity indicates that many neurons alter their discharge rates when a subject is required to wait before responding to a signal. Yet others increase of decrease their activity as the time interval between the onset of the delay and the release of the response increases (Fuster, 1997; Fuster et al. 1982). The majority of these delay neurons are found within the inferior convexity. However, a number of neurons in the superior convexity show similar pr
operties (Pragay et al., 2007). Similarly, high frequency electrical stimulation of the lateral and inferior convexity has been shown to disrupt the ability to inhibit, delay, and withold responses (Goldman et al. 1970; Gross & Weiskrantz, 1964; Stamm & Rosen, 1969), whereas low frequency stimulation actually improves performance on delayed response tasks and enhances behavioral inhibition (Wilcott, 1974, 1977). Interestingly, electrical stimulation of the right frontal region, as compared to the left, more greatly disrupts delayed response performance.
Conversely, when the lateral and inferior convexity are damaged there results a consistent disturbance across tasks requiring the withholding and delay of a response (Brutkowski et al. 1963; Gross & Weiskrantz, 1964; Mishkin & Pribram, 1956; Stepien & Stamm, 1970). That is, subjects become disinhibited and impulsive --disturbances which in turn effect all aspects of behavior. Patients may spontaneously speak or make comments "without thinking", and act on sudden impulses without regard for consequences. Depending on the extent of the lesion both emotion and cognitive activity can be affected (to be discussed).

In addition, humans and animals tend to become hyperreactive and may demonstrate increased activity levels (Bradford, 1950; French, 1959; Fuster 2015; Joseph 1986a, 1988a, 2011a; Latto & Cowey, 2001a; Rose, 1950). For example, noise, threat, or novel stimuli result in significantly heightened activity in frontal animals coupled with distractability (French, 1959).
Among humans a perpetual shifting of attention may result as they are inordinantly distracted by noises in the hall or even specks on the testing table. However, if potential distractors are removed or the subject is placed in a darkened room, activity and hyperresponsiveness declines (French, 1964; French & Harlow, 1955).
With more restricted or lateralized lesions, long term effects are more mild. For example, right sided damage being more frequently associated with motor decontrol, such as edginess (Grafman et al., 1986).
ATTENTION The frontal lobes are highly involved in the maintenance of concentration and attentional functioning (Como et al. 1999; Crowne, 2013; Fuster, 1997; Luria, 1980; Joseph et al. 1981; Joseph 1986a, 1988a, 2017a; Knight et al. 1981; Pragay, et al. 2017), and attentional disturbances are frequently associated with frontal lobe lesions in general, and orbital injuries in particular. Among primates and mammals, disturbances involving visual, spatial, auditory, tactile, and olfactory discrimination are most commonly observed with orbital lesions (Fuster, 1997; Oscar-Berman, 1975).
Some patients, although fully alert and oriented are easily distracted and show wandering or a perpetual shifting of attention (Bianchi, 1922; Stuss & Benson, 1984). They may seem distracted by noises in the hall, specks on the testing table, or extraneous objects around the room.Others may seem easily overwhelmed by complexity or behave as if their sensory-perceptual capacities were significantly narrowed (Yarcozynski & Davis, 1942). In severe cases attention may be focused for only short time periods. For example if asked to count they may stop after reaching 10 or 15 (Rose, 1950) and then must be prodded to continue. In part, this distractibility is due to heightened generalized arousal levels and disinhibition such that the patient is overwhelmed and/or easily distracted by irrelevant stimuli.
In milder cases, although repetition of digits may be normal, or even well above average, when required to recite digits backwards performance often is abnormal (Partridge, 1950; Petrie, 1952). Hence, although able to attend (or at least echo what has been said), the ability to maintain sustained concentration is often disturbed.
Some patients display different types of attentional disturbance depending on the laterality of the lesion as well as the extent to which it involves the convexity or orbital areas. For example, some right frontal patients are impaired on sustained attention tasks when stimuli are presented at the rate of 1 stimulus per second. However, if presented with 7 stimuli per second performance is improved (Wilkins, Shallice, & McCarthy, 2007). This suggests that right frontal patients may be understimulated and show wondering attention if not fully engaged.
The left frontal lobe appears to be more concerned with verbal attentional functioning and the monitoring of temporal-sequential and detailed events (Milner, 2001; Petrides & Milner, 1982; Wagner et al., 1998), whereas the right is more attentive to non-verbal auditory, visual, tactual, and social-emotional stimuli (Brewer et al., 1998; Joseph, 1988a). For example, damage involving either the right or left frontal lobe may impair performance on the Picture Completion subtest of the WAIS-R (i.e. detecting a missing detail such as a dog leaving no footprints), but for different reasons. Left frontals may do poorly because of inattention to detail (e.g. "There's nothing wrong with this picture"). Right frontals may perform deficiently due to impulsive tendencies to say the first thing that comes to mind ("The dog doesn't have a leash"). Higher false positive rates also occur when the frontal lobes are damaged. However, both right and left frontals may do poorly because of memory loss (see below).
On the other hand, some patients seem remarkably able to maintain directed attention, at least when performing simple tasks. Hence, some frontal patients can perform tasks such as digit span without difficulty (Benson et al. 1976; Partridge, 1950; Petrie, 1952; Stuss et al. 1978). Indeed, patients may seem to be locked into this as if all potentially interfering stimuli were completely blocked out; a condition that may best be described as "perseverative attention".
Moreover, the ability to shift attention is reduced, such that the patient appears "stuck in set" and may demonstrate perseverative attentional activity and difficulty shifting responses. The ability to alternate between and thus selectively activate specific neural networks is also disrupted, such that patients have difficulty shifting from one train of thought to another. Thus they may tend to become stuck in-set and to engage in recurrent perseveration (Joseph, 1986a, 2017a; Vikki, 1989) such that the same speech patterns or "memories" may be repeated unless something occurs to arrest and shift their attention. However, if the patient or orbitally lesioned animal is purposefully distracted, e.g. via a novel stimulus, this pattern of perseverative attention is momentarily halted and the ability to shift response and attention is briefly regained (Mishkin, 1964; Pribram et al., 1964).
PERSEVERATION
Based on neuropsychological studies of patients with frontal lobe lesions, this author proposed, in the first edition of this text, that the frontal lobes could be divided into anterior and posterior (as well as lateral and medial) motor areas, with the posterior areas subserving and maintaining the greatest concentration of motor-related functions. This distinction has been subsequently verified based on a variety of functional imaging studies (reviewed in Passingham, 1997).
Because the anterior and supposed non-motor areas of the frontal lobes also subserve motor functioning, and in fact often become activate just prior to and in anticipation of movement (Passingham, 1997), injuries to these tissue can in fact affect movement and speech, giving rise to retarded or excessive, and/or perseverative and compulsive movement and speech disorders (Joseph, 1988a, 2017a; Luria, 1980; Na et al., 2017; Sandson & Albert, 1984).
Although damage in the anterior, inferior, right and left frontal lobes can produce perseverative disorders (Luria, 1980; Na et al., 2017; Sandson & Albert, 1984), lesions involving the orbital frontal cortex and inferior convexity have consistently resulted in increased activity levels and an abnormal tendency to repeat previous responses in a repetitive perseverative fashion. Perseverative behavior has been observed even when the context is no longer appropriate or rewarded, and/or the response is punished and the individual realizes his responses are incorrect (Butter, 1969; Butter et al., 1963; Iverson & Mishkin, 1970; Jones & Mishkin, 1972; Kolb et al., 1974; Luria, 1980; Mishkin, 1964). In part, because the inferior-orbital areas are involved in inhibition (Konishi et al., 2017) and discrimination (Tremblay & Schultz, 2017), with orbital lesions the capacity to shift responses is attenuated. Thus, once a behavior is completed, particularly if it is repeatedly performed, the pattern continues to be involuntarily executed such that the ability to change to a different pattern of activity is disrupted. Similar disturbances are seen with inferior medial damage.
Perseverative, repetitive abnormalities can effect motor behavior. For example, a common feature of large lesions involving the neocortical regions is macrographia and expansiveness, which can be seen in drawings which are made abnormally large. Conversely, deep frontal lesions may produce micrographia, such that drawings become abnormally small; i.e. micrographia. However, in some instances, micro- or macro- graphia may be also be contaminated with perseverative abnormalities (see figures ).
Perseverative tendencies also influences speech. For example, patients may tend to repeat phrases: "Doctor, can I look at this, can I look at this, Doctor, can I look at this", or once a topic seems to have changed they may reintroduce and again repeat certain statements or words. During the administration of the Vocabulary subtest from the WAIS-R, for example, an orbitally damaged patient managed to use the word "summer" in five different definitions.
Another patient with damage and suspected subclinical seizures involving the mesial orbital cortex and possibily the basal ganglia, demonstrated a striking perseveration of movement. If asked to draw a star he would dash off four or five. When he attempted to sit, the downward motion continued in a repetitive machine-like fashion and he would slide to the floor.
In severe cases it is frequently found once a patient has performed a required action, such as drawing a particular figure several times, when asked to draw a different figure they continue to draw the first. If asked to draw a star, a cross, and a square, the patient may draw the first or even the second figure correctly but then impulsively draws again the star. In the less extreme they may simply inappropriately introduce components of a previous response in their next set of actions (Luria, 1980).
Conceptually, patients may have difficulty considering or even recognizing alternatives such that they seem to become locked into a particular mode of thinking or activity. For example, Luria (1980) describes "a patient with a wound of the frontal lobes, who, when working in the carpenters shop of the hospital, inertly went on planing a piece of wood until nothing of it remained. Even then he did not stop but continued to plane the bench" (p. 294).
Although orbital patients may have difficulty inhibiting emotionality, the perseverative abnormality is not due to disturbances involving impulsivity or the withholding of a behavioral response. For example, across tasks requiring delayed responding, orbital lesions have little or no effect on performance (Brutkowski, et al. 1963; Rosenkilde, 2009). Rather, the problem is in shifting sets and in inhibiting the recurrence of a previous response when the next action is initiated. Once a behavior occurs it tends to persist and contaminates the performance of unrelated actions.
Perseverative reactions, however, may occur with lesions situated well outside the orbital areas. For example, perseveration of speech (of which there are several subtypes), can also occur among individuals with aphasia and with lesions localized to the right or left frontal lobe or the anterior, superior, or inferior frontal lobes (Joseph, 1986a, 2017a; Peitro & Rigdrodsky, 1986; Sandson & Albert, 1987). With left or right convexity lesions, and as patients grope for words, the same word may be repeated in a number of erroneous contexts in a perseverative fashion.
Again it must be stressed, however, that lesions are seldom confined to a single quadrant of the frontal lobe, even following "frontal lobotomy" -which during the 1940's and 1950's were often perform as an in office procedure and were simply quite sloppily performed. Moreover, damage in one frontal area may result in abnormal activity in yet a different region of the frontal lobe due to disconnection and loss of input or inhibitory restraint. Hence, symptoms associated with orbital destruction may also appear with medial or lateral convexity injuries and vice versa. In addition, it appears that the right inferior-orbital region performs somewhat different functions than the left orbital areas (e.g. Grafman et al. 1986; Konishi et al., 2017) such that different symptoms may appear with right vs left lesions.
THE ORBITAL AND THE LEFT AND RIGHT FRONTAL LOBE
It is sometimes difficult to distinguish between an orbital and a right frontal injury, and in part this is due to the fact that both the right lateral and orbital frontal lobes are dominant over the left in the regulation of neocortical and limbic system arousal (Joseph, 2017a; Konishi et al., 2017). That is, the orbital and the right frontal lobes both exert inhibitory influences over arousal, serving what Posner and Raichle (1994) refer to as a altering/arousal vigilance network, such that when injured, patient's may become disinhibited and develop the classic "frontal lobe personality."
Of course, the orbital frontal lobes are also a subcomponent of the right and left frontal lobe. However, in this regard, just as the right frontal lobe in general is dominant in regard to arousal, the right orbital area appears to be dominant as well (Konishi et al., 2014), for right orbital injuries are more likely than left orbital injuries to result in gross emotional disinhibition (Grafman et al. 1986). Patients become disinhibited, hyperactive, euphoric, extroverted, labile, overtalkative, and may develop perseveratory tendencies (Butter 1969; Butter et al. 1970; Greenblatt 1950; Joseph 2007a; Kennard et al. 1941; Kolb et al. 1974; Malloy, Birhlr, and Duffy 1993; Reitman 1946 1947; Ruch and Shenkin 1943). By contrast, reductions in left orbital functioning are associated with psychomotor retardation, depression, and anxiety (Joseph, 2017a), and even PTSD (Rauch et al., 1996; Shin et al., 1997).
When coupled with abnormalities involving the right convexity, patients may become so disinhibited, delusional, grandiose, and emotionally labile that they develop what has classically been described as mania. However, because the orbital frontal lobes not only regulate but integrate and relay emotional input to the neocortex, with massive bilateral orbital destruction, rather than a loss of emotional control, there is a complete loss of emotion, and the patient will become severely apathetic, indifferent, and unresponsive (see below).
Likewise, with massive bilateral convexity lesions patients may also become severely apathetic and unresponsive. However, similar disturbances may result with massive left convexity or massive left orbital lesions (Joseph, 2017a).
Nevertheless, although there are parallels, the right and left convexity perform functions which are distinct from the right and left orbital frontal lobes. For example, whereas the orbital frontal lobes regulate limbic and autonomic nervous system activity (Joseph, 2017a), the right and left convexity regulate neocortical and thus cognitive and perceptual activity, including memory (Brewer et al., 1998; Koechlin et al., 2017; Wagner et al., 1998; Dolan et al., 1997; Tulving et al., 1994; Kapur et al., 2015).
REFERENCES
















