Rhawn Joseph, Ph.D.
BrainMind.com
Parietal Area 7
The parietal lobes are not a homologous tissue but consists of cells which are responsive to a variety of divergent stimuli, including movement, hand position, objects within grasping distance, audition, eye movement, pain, heat, cold, as well as complex and motivationally significant visual stimuli (Aoki et al., 2014; Cohen et al. 1994; Deibert et al., 2016; Dong et al. 1994; Lam, et al., 2014; Lebedev et al. 1994; Lin & Sessle 1994; Pred'Homme & Kalaska 1994; Remy et al., 2016; Snyder et al., 2017).
For example, area 7 of the superior parietal lobe receives considerable visual input, particularly from the lower visual fields and periphery--information which it receives not only from the visual cortex but from extra-retinal pathway. Area 7 also receives complex somesthetic stimuli regarding the hand and objects beyond and within reaching distance. Moreover, different neurons in different regions of area 7 perform somewhat different functions. Even the subareas within the primary somesthetic neocortex, areas 3ab,1, 2, respond to different stimuli and receive projections from different subregions within the thalamus, which in turn receive different forms of input from skin, joints and muscles.
PARIETAL TOPOGRAPHY
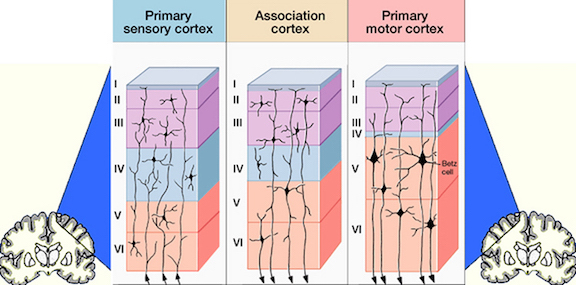
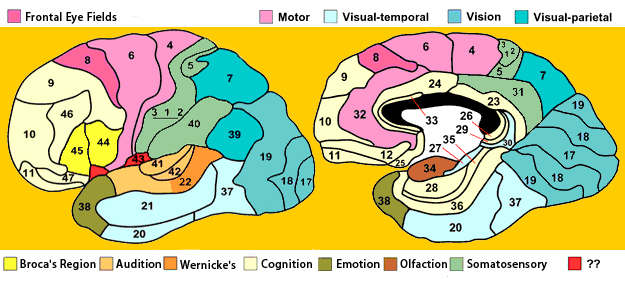
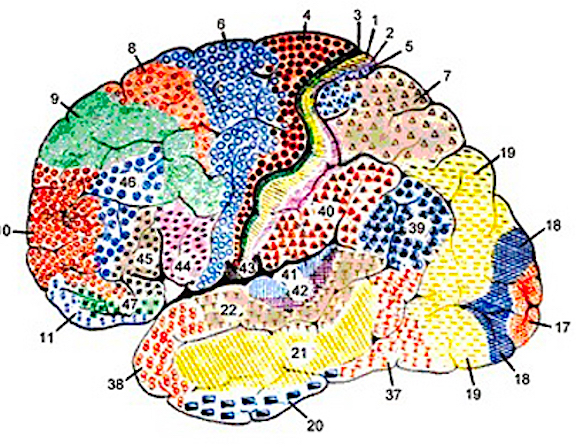
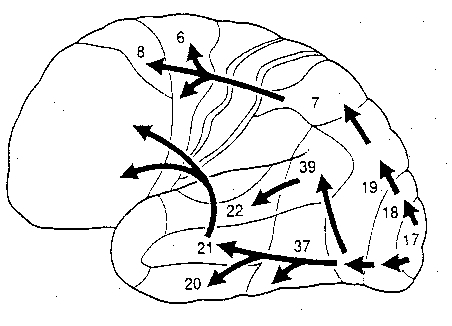
There are nine major somesthetic areas within the parietal lobe, such that the primary, association, and assimilation areas actually consist of numerous subareas. Broadly, and most generally, however, the parietal lobe may be subdivided into a primary receiving area (involving Brodmann's areas 3ab,1,2) within the post central gyrus, an immediately adjacent somesthetic association area (Brodmann's area 5ab), a polymodal (visual, motor, somesthetic) receiving area located in the superior-posterior parietal lobule (area 7ab), a granular insular area which is located in the inferior convexity and encompasses part of the marginal gyrus, and a multimodal-assimilation area within the inferior parietal lobule (areas 7, 39, 40) which encompasses the angular and supramarginal gyrus.
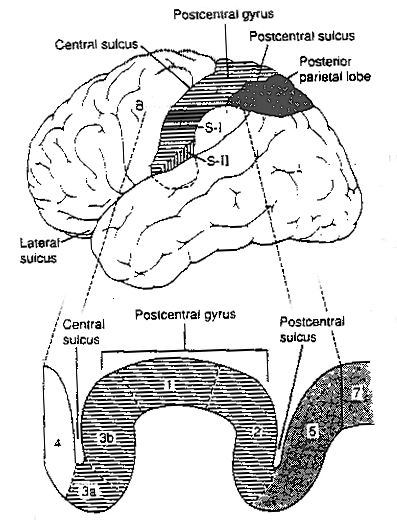

AREA 7 & THE SUPERIOR-POSTERIOR PARIETAL LOBULE
POLYMODAL INFORMATION PROCESSING
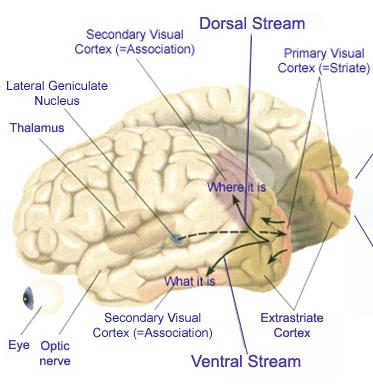
Information processed and analyzed in the primary and association somesthetic areas is then transmitted to area 7 where polymodal analyses take place (Dong et al. 1994; Lam et al., 2014; Previc 2003). Jones & Powell (1970) considered area 7 to be concerned with the highest levels of somesthetic integration as it receives massive input from the visual receiving areas in the occipital and middle temporal lobe, from the motor and non-motor areas in the lateral frontal convexity as well as from the inferior parietal and temporal lobe including the parahippocampal gyrus (Cavada & Goldman-Rakic 1989; Dong et al. 1994; Jones & Powell, 1970; Previc 2003; Wall et al. 2012). Cells in area 7 also have auditory receptive capacities, including the ability to determine sound location (Hyvarinen 2012).
Hence, area 7 is heavily involved in the analysis and integration of higher order visual, auditory and somesthetic information, and single neurons often have quite divergent capabilities. Moreover, cells in area 7 process both body-referenced and world-referenced signals, which provide information for the control of gaze and navigation and movement in space (Synder et al., 2017).
THREE-DIMENSIONAL ANALYSIS OF BODY-SPATIAL INTERACTION
A single area 7 neuron via the reception of converging input from the primary and association somesthetic regions can monitor activities occuring in many different body parts simultaneously; for example, the position and movement of the arms, trunk, and legs (Leinonen et al. 2009). Via the reception of auditory, somesthetic, and visual input, area 7 is thus also able to create a three-dimensional image of the body in space (Lynch, 1980).
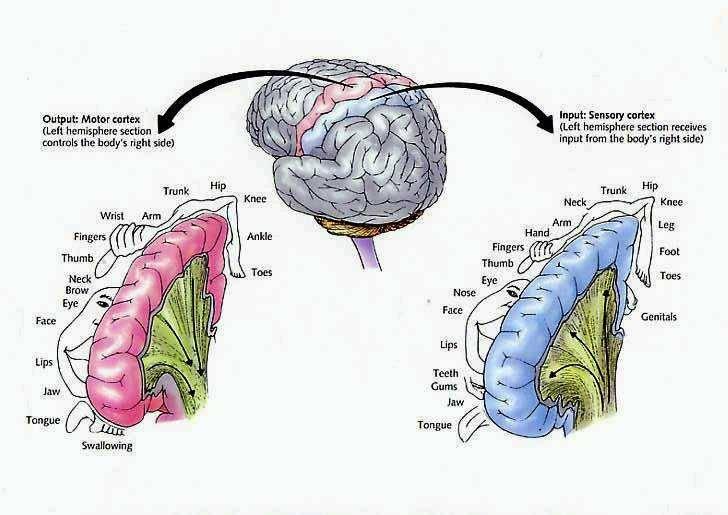
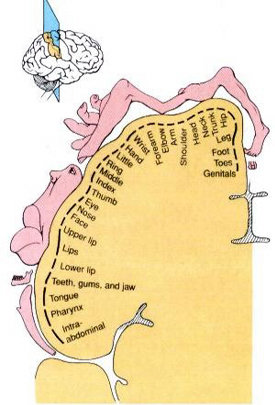
Moreover, cells in this area not only receive information about body part interrelationships (such as is maintained by area 5), but the interaction of the body with external objects and events in visual space (Snyder et al., 2017; Stein 1992). Indeed, many cells in this vicinity become highly active when the hand is moved toward or while reaching for and/or manipulating objects (Mountcastle et al. 1975; Robinson et al. 2008; Yin & Mountcastel, 1977). They also act to coordinate and guide gaze and whole body-positional movement through visual and auditory space (Snyder et al., 2017).
As stated by Mountcastle, Motter & Andersen, (1980), "the parietal lobe, together with the distributed system of which it is a central node, generates an internal neural construction of the immediately surrounding space, of the location and movements of objects within it in relation to body position, and of the position and movements of the body in relation to that immediately surrounding space. The region appears in general to be concerned with continually updating information regarding the relation between internal and external coordinate systems" (p.522).
VISUAL-SPATIAL PROPERTIES
As noted in chapter 5, the parietal lobe, in part, evolved from the hippocampus, and maintains rich interconnections with this nucleus via the parahippocampal gyrus. Given that the hippocampus is concerned with the body-in-space, and with the position of various objects and stimuli in visual space (Nadel, 1991; O'Keefe, 1976; Wilson and McNaughton, 1993), not surprisingly, the parietal lobe performs similar functions.
Although cells in area 7 receive and transmit data to the hippocampus via the parahippocampal gyrus, these neurons also accomplished these functions via the convergence of somesthetic information from area 5, visual input from the visual association areas, as well as via the reception of midbrain and vestibular signals (Cavada & Goldman-Rakic 1989; Gross & Graziano 1995; Hyvarinene, 2012; Kawano & Sasaki, 1984; Previc 2013). Like some hippocampal neurons, single parietal neurons are also involved in the representation of space (Gross & Graziano 1995; Stein 1992) as well as the body in space (Synder et al., 2014) including topographic learning.
Area 7 neurons also have quite large visual receptive fields, sometimes occupying a whole quadrant, hemifield, or the entire visual field (Robinson et al., 2008). However, the receptive visual fields of these neurons do not usually include the fovea and are more sensitive to objects in the periphery and lower visual fields (Motter & Mountcastle, 1981; Previc 2003); i.e. where the hands, feet, and ground are more likely to be viewed. In this regard, many of these cells are not concerned with the identification of form but rather place, position, and reaching distance as well as depth perception and the coordination of the body as it moves through space. To accomplish this, however, requires that this area of the brain also participate in memory and place this information in short-term memory so that comparison can be made and information retained.
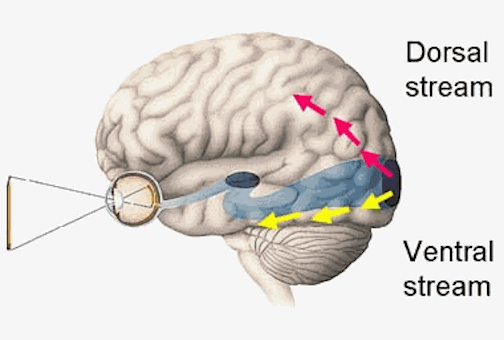
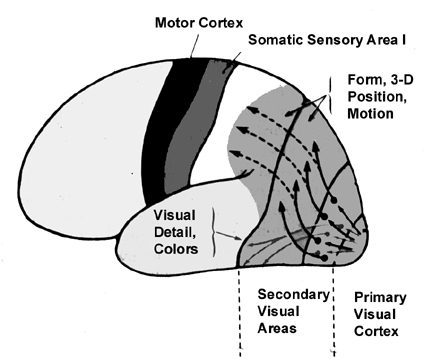
Because many of the neurons in area 7 receive highly processed visual (as well as limbic) input, including information regarding ocular movement and direction of gaze (Snyder et al., 2017), they are also responsive to and can determine a variety of visual-object qualities and interrelationships, such as motivational significance, direction of movement, distance, spatial location, figure-ground relationships, and depth, including the discrimination and determination of an objects 3-dimensional position in space, (Andersen et al. 2015; Gross & Graziano 1995; Kawano,et al., 2014; Lynch, 1980; Previc 2003; Sakata et al. 2008; Snyder et al., 2017; Stein 1992). They are largely insensitive to velocity or speed of movement. In addition, many cells respond most to stimuli which are within grasping distance, whereas others respond most to stimuli which are just beyond arms reach.
Neurons in this supramodal region are able to accomplish this by responding to somesthetic positional information provided by area 5, visual input from areas 18,19, and the inferior and middle temporal lobe and hippocampus, as well as from extraretinal signals regarding convergence and accommodation of the eyes, and the position and movement of the eyes while tracking (Gross & Graziano 1995; Previc 2003). Indeed, electrical stimulation of this area elicits eye movements as well as convergence, accommodation, and pupil dilation (Jampel, 1960). By integrating these signals these cells are able to monitor and mediate eye movement and visual fixation, map out the three-dimensional positions of various objects in visual space, and determine the relation of these objects to the body and to other objects.
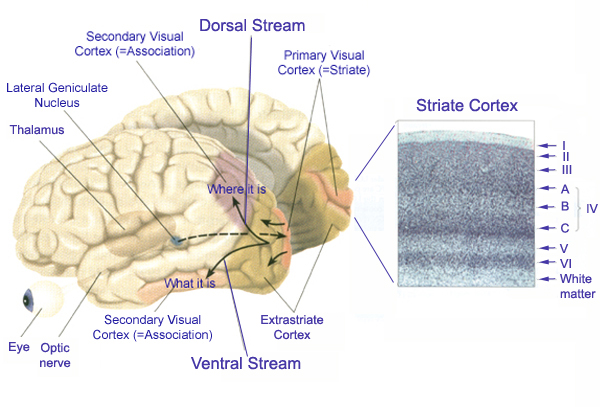
VISUAL ATTENTION
Many neurons in area 7 can act so as to increase or decrease visual fixation, direct attention to objects of motivational significance, and promote the maintanance of visual grasp such that a moving object continues to be visually scanned and followed (Lynch et al., 1977; Previc 2003). Electrical stimulation of this area induces lateral eye movements due to interconnections maintained with the frontal eye fields, visual cortex, and subcortical visual centers.
In contrast, lesions to this area often disrupt attentional functioning, such that in the extreme (for example, following a right parietal lesion) patients fail to attend to the left half of not only visual space, but the left half of the body; i.e. neglect. Neglect is most common with right cerebral lesions but in some instances may be seen initially following massive left cerebral destruction (Chapter 10; see also Umilta 1995).
As detailed below, with circumscribed parietal lesions involving area 7, neglect is more pronounced for the lower visual fields. The lower visual fields, in turn are a predominant sensory domain of the parietal lobes, for this is the area of visual space in which the hands and feet are most likely to view -therefore making movement of the body in space more efficient. The lower visual field is also the area in which objects are most likely to be physically explored by the hand, which in turn are guided by the parietal lobe.
MOTIVATIONAL & GRASPING FUNCTIONS
A number of cells in area 7 have been described as exerting "Command" functions (Mountcastle, 1976; Mountcastle et al., 1980), especially those located along the inferior lateral convexity. These cells are motivationally responsive, can direct visual attention, become excited when certain objects are within grasping distance and can motivate and guide hand movements including the grasping and manipulation of specific objects (Hyvarinene & Poranene, 1974; Lynch et al., 1977; Mountcastle, 1976). Most of these latter cells cease to fire when the object fixated upon is actually grasped, suggesting that they may be exerting some type of driving force or at least an alerting function so that objects of desire or of general (vs. specific) interest will be attended to (Rolls et al. 2009).
In other words, when an object is recognized as being of motivational significance (determined by the limbic system and visual form recognition neurons in the temporal lobe), this information is relayed to neurons in area 7. Although not concerned with form recognition, these cells will guide as well as monitor eye movement so that the object of interest is fixated upon. These cells then exert motor command functions so that the hand is guided toward the object until it is grasped.
ATTENTION & VISUAL SPACE
Lesions involving the superior, as well as the inferior parietal lobule (of which area 7 is part) and the parietal-occipital junction can greatly disturb the ability to make eye movements, maintain or shift visual attention, visually follow moving objects, and in the extreme result in oculomotor paralysis (Hecaen & De Ajuriaguerra, 1954; Previc 2003). Right parietal lesions are associated with deficiencies involving depth perception and stereopsis, including the abilIty to determine location, distance, spatial orientation and object size (Benton & Hecaen, 1970; Ratcliff & Davies-Jones, 1972). Visual constructional abilities may also be compromised (see Cowey, 1981; Critchley, 1953) and many patients suffer from visual-spatial disorientation and appear clumsy.
Individuals with right parietal lesions show defective performance on line orientation tasks (Warrington & Rabin, 1970; Benton et al. 1975), maze learning (Newcombe & Russell, 1969), the ability to discriminate between unfamiliar faces (Milner, 1968), or to select from the visual envrionment stimuli which are of importance is also a consequence of right parietal lesions (Critchley, 1953), whereas those with right parietal-occipital damage are deficient on tasks requiring detection of imbedded figures (Russo & Vignolo, 1967).
Others may also have severe problems with dressing (e.g. dressing apraxia) and may become easily lost or disoriented even in their own homes. One patient I examined with a gun shot wound involving predominantly the right superior posterior parietal area was unable to find his way to and from his hospital room (although he had been an inpatient for over 3 months) and on several occasions had difficulty finding his way out of the bathroom. Indeed, in one instance he was discovered feeling his way along the walls in his attempt to find the door.
Commonly right parietal injuries can result in a complete neglect of the left half of visual space. By contrast, left parietal injuries result in only minimal right sided neglect. Again, this is because the right parietal lobe not only attends to both halves of the body, but both halves of visual space (Joseph, 1986ab, 1988a).
LOCALIZATION OF OBJECTS IN SPACE
Patients with right parietal lesions perform defectively on visual localization tasks (Hannay, Varney, & Benson, 1976). However, Ratcliff and Davies-Jones (1976) found that the localization of stimuli within grasping distance is disrupted equally by lesions to the posterior region of either hemisphere. Hence, the right parietal lobe appears to play an important role in generalized localization, whereas the left exerts influences in regard to objects which may be directly grasped and manipulated.
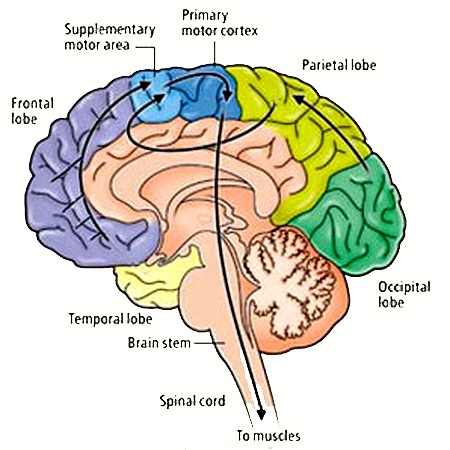
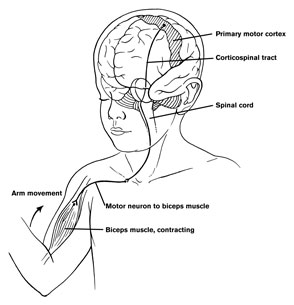
APRAXIA
The parietal lobes mediate hand, arm and body movements in space, as well as temporal-sequencing (Joseph 1993; Kimura 1993; Lynch et al. 1977). Hence, damage to this vicinity can also result in apraxia such that individuals have difficulty controlling or temporally sequencing the extremities. This is especially evident with left rather than right parietal injuries. There can also result gross inaccuracies as well as clumsiness when making reaching movements or when attempting to pick up small objects in visual space (Kimura 1993; Lynch, 1980). Tendon reflexes may be slowed, and hypotonia coupled with a paucity of and/or a slowness in movement initiation may result (Denny-Brown & Chambers, 1958; LaMotte & Acunam, 2008; Lynch, 1980). With left parietal damage there may result difficulty visually recognizing objects (agnosia), and left-right orientation may be grossly deficient.
EMOTION
Some of the effects of lesions in this region also include altered emotional-motivational functioning, body and visual-spacial neglect, as well as clumsiness and visual-spatial disorganization. With massive right parietal lesions involving area 7, many patients are often initially hypokinetic and seem very passive, inattentive, unresponsive and take very little interest in their environment (Critchley, 1953; Heilman & Watson, 1977). Moreover, when their disabilities are pointed out (e.g. paresis, paralysis), they may seem indifferent or conversely euphoric (Critchley, 1953).
Area 7, as well as 5 (and the inferior parietal lobule) receive auditory information (Cavada & Goldman-Rakic 1989; Hyvarinen, 2012; Lam et al., 2014; Roland et al., 1977) and are capable of discerning the emotional-motivational significance of this input as well as differentiating between different vocal-emotional characteristics --especially right parietal neurons. Hence, when the right parietal region is damaged, patients not only seem unconcerned about their disability, but they may have difficulty perceiving and differentiating between different forms of emotional speech (Tucker, Watson & Heilman, 1977).
PAIN: AREAS 5, 7, & THE SUPRAMARGINAL GYRUS
As noted, neurons in area 5, as well as those located in the insula, receive direct thalamic input from the ventral and posterior portion of VPL. The ventral portion in particular, however, in addition to somesthetic information, may also convey pain sensation to the parietal lobe. In fact, Penfield and Boldrey (1937) reported that electrical stimulation of the parietal lobe resulted in the sensation of pain, albeit about 1% of the time.
Some neurons located in area 5 and 7 of the parietal lobe also demonstrate pain sensitivity, with some are 7 neurons responding exclusively to thermal and nociceptive stimuli (Dong et al. 1994) and with area 5 presumably acting to localize the source of pain. Hence, in some instances, such as when the more inferior portion of area 5, 7 or the supramarginal gyrus (Broadmann's area 40) has been destroyed, patients may demonstrate a lack of emotional responsiveness to painful stimuli, become indifferent, develop an increased pain threshold, tolerate pain for an unusually lengthy time period and fail to respond even to painful threat (Berkley & Parmer, 1974; Biemond, 2016; Geschwind, 1965; Greenspan & Winfield 1992; Hyvarinen, 2015; Schilder, 1935) --particularly with right parietal destruction (Cubelli et al 1984). However, disturbance or lack of pain sensation has been noted to occur when lesions to either hemisphere.
Moreover, loss of sensation or an inability to react to pain may also occur from subcortical lesions, especially within the thalamus, and less often, with surgical destruction of the anterior cingulate--the so called center of "pain and misery." In this regard, there appears to be two major cerebral pain pathways, a subcortical medial pathway involving the thalamus and cingulate, and a neocortical pathway involving the parietal lobe.
At the neocortical level, although pain responsiveness may be diminished or absent following damage to these tissues, elementary sensation remains intact and the ability to differentiate, for example, between dull and sharp is retained. The deficit is usually bilateral.
In this regard, Schilder (1935), has argued that the loss of reaction to pain is due to disturbances in the image of the body. That is, the experience or threat of pain is no longer related to the body image. Geschwind, (1965), however, raises the possibility that this condition is due to disconnection from the limbic system (see Cavada & Goldman-Rakic 1989). If this were the case, somesthetic (painful) sensation would no longer be assigned emotional significance and would thus implicate the insular region of the parietal lobe, which also receives visceral as well as somesthetic information and funnels this data to the limbic system.
In fact, this same insular-limbic pathway may serve to promote tactile memory; that is, via the funneling of complex somesthetic information to the hippocampus and amygdala. Conversely, it may be this same pathway which when abnormally activated or injured, may give rise to abnormal emotional significance being attributed to bodily sensations.
PAIN AND HYSTERIA
Whereas destruction of the inferior portions of areas 7, 5, and 40 may result in loss of pain sensation, when the injury is secondary to tumor or seizure activity patients may instead report experiencing pain (Davidson & Schick, 1935; Hernandez-Peon et al. 1963; Ruff, 1980; Wilkinson, 1973; York et al. 2009). In addition patients may experience sensory distortions that concern various body parts due to abnormal activation of the parietal neocortex.
For example, one 48 year old housewife complained of diffuse, poorly localized (albeit intense) pain in her left leg, which occurred in spasms that lasted minutes. She subsequently was found to have a large tumor in the right parietal area, which, when removed, alleviated all further attacks. Head and Holmes (1911) reported a patient who suffered brief attacks of "electric shock"-like pain that radiated from his foot to the trunk; a glioma in the right parietal area subsequently was discovered.
Sometimes the pain may be related to abnormal sexual or genital sensations. For example, one 9-year-old boy seizure activity in the right parietal area experienced spontaneous attacks of intense scrotal and testicular pain (York et al.2009). Ruff (1980) reports two cases who experienced paroxsymal episodes of spontaneous and painful orgasm, which was secondary to right parietal seizure activity. In one patient the episodes began with the sensation of clitoral warmth, engorgement of the breasts, tachycardia, etc., all of which rapidly escalated to a painful climax.
It is important to note, however, that although the predominant focus for paroxysmal pain is the right hemisphere, pain also has been reported to occur with tumors or seizures activity that involves the left parietal region (Bhaskar, 1987; McFie & Zangwill, 1960).
Unfortunately, when the patient's symptoms are not considered from a neurological perspective, their complaints with regard to pain may be viewed as psychogenic in origin. This is because the sensation of pain, stiffness, engorgement, is, indeed, entirely "in their head" and based on distorted neurological perceptual functioning. Physical exam may reveal nothing wrong with the seemingly affected limb or organ. Thus such patients may be viewed as hysterical or hypochondriacle, particularly in that right hemisphere damage also disrupts emotional functioning.
REFERENCES
















