Rhawn Gabriel Joseph, Ph.D.
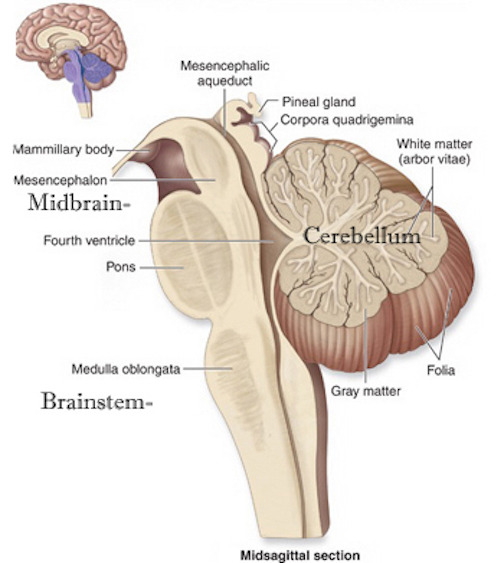
The brainstem can be subdivided and consists of the medulla (myelencephalon), pons (metencephalon) and midbrain (mesencephalon), the reticular formation, and monoaminergic neurotransmitter systems as well as the cranial nerves and associated nuclei.
Straddling the dorsal surface of the pons is the cerebellum and spanning the length of the brainstem are the nuclei for cranial nerves 3-12 (see Carpenter 1991; Parent 1995) which will be briefly reviewed below.
The pons represents the most rostral portion of the "hindbrain" and it is separated from the midbrain via the superior pontine sulcus, and from the medulla by the inferior pontine sulcus. The two components of the vestibulocochlear nerve (VIII), and the facial (VII), abducens (VI) -which also lies in the floor of the fourth ventricle- and the trigeminal nerve (V) are the cranial nerve nuclei associated with the pons.
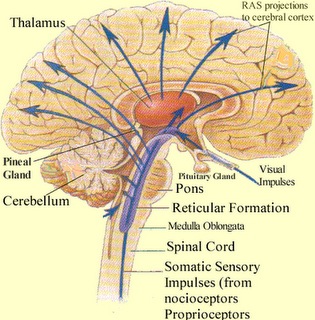
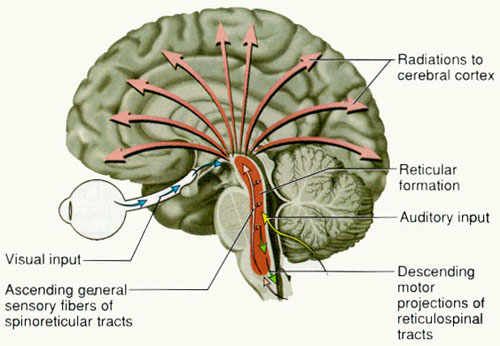
Spanning the medial and lateral lengths of the brainstem and pons is a complex reticulum of richly interconnected cells with long ascending and descending axons, collectively referred to as the reticular activating system, which is concerned with generalized and selective arousal and activation of the neuroaxis. The pontine tegmentum and the more dorsal portion of the midbrain tegmentum is also considered part of the reticular formation through which course ascending sensory fibers and descending motor fibers.
Neurons of the reticular formation receive specific sensory input from the skin, muscles, joints, and vestibular system which they then integrate. That is, the reticular formation as a whole, is not only concerned with arousal, but the integration and coordination of behavior in response to arousal, such as through the integration of trunkal, limb, arm, head and eye movements. Hence, the reticular formation exerts both generalized and specific activating influences and is thus concerned with sensory-motor integration (mediated by the excitatory and inhibitory neurotransmitters, glutamate and GABA) and modulatory functions (mediated by norepinephrine and serotonin).
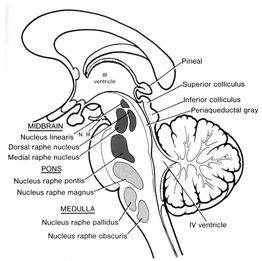
In part, the activating influences of the reticular formation are subserved by clusters of chemically active neurons which secrete and distribute norepinephrine (NE), serotonin, (5HT), and acetylcholine (ACh). Specifically, clusters of NE neurons (designated A1 through A7 by Dahlstrom & Fuxe, 19), are scattered throughout the lateral, ventral, and anterior reticular formation and tegmentum.
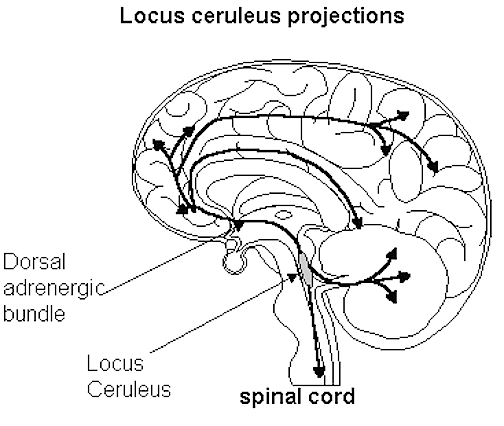
NE neurons project throughout the brain and spinal cord are concerned with emotional arousal, promote neuroplasticity, and also serve a neural protective function when subject to considererable and prolonged emotional stress (see chapter 30). Specifically, the more anteriorally located cell cluster, the locus ceruleus (A4 and A6) projects to the spinal cord, the brainstem, cerebellum, and cerebral cortex. Other NE cell clusters project throughout the brainstem, hypothalamus, limbic system and limbic striatum (A1-A3), as well as to the spinal cord and lower brainstem (A5 and A7).
5HT neurons are also concerned with emotional arousal, as well as with sensory filtering. 5HT neurons are clustered throughout the brainstem, forming the raphe nuclei in the midline, dorsal and median portions of the reticular formation (B1-B9). 5HT these neurons project throughout the forebrain.
Brainstem ACh neurons are clustered in the pons, forming the pendunculopontine nucleus and the laterodorsal tegmental nucleus. These neurons project throughout the brainstem, diencephalon, and limbic forebrain. Additional clusters of ACh manufacturing and secreting neurons are located in the septum and substantia innominata ("extended amygdala").
THE BRAINSTEM RETICULAR FORMATION
The pontine and medullary brainstem significantly contributes to cortical arousal, attention, sensory and motor facilitation and inhibition, and the orienting reaction. This is made possible through ascending limbic, thalamic, and neocortical influences, and descending inhibitory influences on brainstem and spinal sensory and motor nuclei. In this manner the brainstem is able to selectively arrest and activate specific movements (NE/ACh), while simultaneously filtering out distracting or irrelevant sensory stimuli (presumably via 5HT influences) so that attention may be selectively directed and maintained (see Klemm, 2011; Steriade & McCarley, 2011).
Brainstem motor and sensory filtering and inhibition is also made possible via reticular neuronal modulatory control over arousal; i.e. the reticular activating system. The brainstem reticular formation consists of a number of morphologically distinct nuclei and neurons (see Blessing, 2011; Steriade & McCarley, 2011; Vertes, 2011) which differ in regard to size, shape, number and orientation of dendrites, and axonal patterns of projection. It is via these nuclei that the reticular formation can exert widespread influences on motor and sensory functioning as well as arousal.
For example, cranial nerve and sensory fibers originating in the spinal cord give off collaterals as they ascend or terminate in the brainstem reticular formation (Brodal, 1981; Pompeiano, 1973). This includes auditory and visual fibers originating in the midbrain tectum (Edwards, 1980). The more lateral portions of the reticular formation consists of short axons which richly innervate adjacent brainstem nuclei and which are connected to the collaterals of the various sensory tracts.
Specifically, the ascending reticular activating system consists of a multi-neuronal, polysynaptic core that conveys, via long central axons with numerous laterally projecting collaterals, nonspecific impulses to the forebrain. These long axons ascend via the central tegmental tract which projects to the intralaminar nuclei of the thalamus. Many of these core reticular neurons are concerned with motor activation and inhibition as well as alerting and arousal functions so that motor functioning is coordinated with forebrain sensory processing. By contrast, fibers from the medial reticular formation project to the limbic system.
COMA, LETHARGY & THE BRAINSTEM
The sensory tracts, including those involved in transmitting noxious stimuli, are exceedingly important in maintaining arousal. If these sensory tracts are severed at the brainstem level, patients may become exceedingly lethargic, and even apathetic. Similarly, destruction of the olfactory, auditory, and visual pathways, even at a peripheral level, can result in prolonged lethargic states (see Steriade & McCarley, 2011). This indicates that sensory input, from the periphery to the brainstem, and up through the thalamic level is exceedingly important in maintaining arousal and cortical alertness, as well as the waking state.
However, destruction or interruption of these long ascending sensory pathways (i.e. the lemniscal systems) that span the spinal cord and terminate in the thalamus, does not eliminate the cortical arousal response. This is because forebrain arousal is dependent on the reticular formation which receives collateral sensory fibers from these ascending systems.
Thus, whereas lateral brainstem injuries may create lethargic and reduced states of arousal, destruction of the core brainstem reticular formation can result in a loss of forebrain arousal and create a permanent comatose state such that the patients or animals may not even respond to noxious stimuli (French & Magoun, 1952; Lindsely, et al. 1950). Indeed, transection of the brainstem just a few millimeters below the visual and auditory colliculus also induces coma.
However, this is not only a consequence of a loss of reticular input to the forebrain, but a loss of forebrain (and frontal lobe) and midbrain input to the brainstem reticular formation (Bremer, 1975; see also Steriade & McCarley, 2011). Just as the complexity of motor programming increases as one ascends the brainstem, so to does control over arousal.
For example, if the transection is in the upper brainstem (i.e. at the pons, trigeminal level) well below the tectum, the subject continues to demonstrates EEG signs of alertness, as well as visual tracking eye movements (reviewed by Steriade & McCarley, 2011), although they are otherwise in a coma. However, if the lesion is more anterior at the midbrain level, the subject becomes completely comatose and unresponsive --which indicates an important role for the midbrain reticular formation in maintaining cortical and forebrain arousal.
However, if the lesion is anterior to the midbrain, rather than coma and a loss of arousal, there results a loss of control over arousal such that environmental stimuli may be ignored or neglected. For example, if the immediately adjacent thalamus is lesioned (which is the terminal junction of many reticular fibers), this may result in complete neglect of auditory, visual, and tactual space, depending on the laterality of the damage. Presumably this is a consequence of an inability to transmit or relay sensory stimuli or to activate the neocortex, for with lesions to the thalamus, cortical EEG desynchronization is usually absent regardless of brainstem activity (Steriade & McCarley, 2011).
Conversely, if the lower brainstem is lesioned, the patient not only become comatose, but cardiovascular and respiratory disturbances will result. Hence, the patient may die.
FRONTAL-THALAMIC & AMYGDALOID CONTROL OVER BRAINSTEM AROUSAL
When thalamic output increases, the amplitude of cortical responses also increases (Steriade & McCarley, 2011). However, the thalamus, in turn is also under the control of the lateral and orbital frontal lobes which also exerts significant modulatory influences on the brainstem reticular formation while it simultaneously monitors neocortical and thalamic activity (Joseph, 2014a). Via the frontal lobe, the thalamus and the reticular formation can be activated or inhibited selectively or globally so that specific sensory modalities are attended to while others are filtered or suppressed so that the organism can engage in selective attention and information processing.
Similarly, the amygdala remains capable of inducing arousal as well as EEG desynchronization (Kriendler & Steriade, 1964; see chapter 14). This is because the amygdala is also intimately interlinked with the upper brainstem reticular formation (Takeuchi et al., 1982), as well as the frontal lobe and thalamus. Hence, when stimulated, the amygdala can induce alerting reactions and arousal (chapter 14), as well as PGO waves (Calvo, et al. 2005), which in turn are associated with the onset of paradoxical sleep and brainstem activity.
In addition, the medial and lateral portions of the posterior hypothalamus also project to the midbrain reticular formation (Steriade & McCarley, 2011), as does the preoptic regions of the hypothalamus (Swanson, et al. 2005) -a region intimately involved in sexual behavior and sexual posturing (chapter 14). The hypothalamus is also intimately linked with the orbital frontal lobes and the amygdala. Therefore the hypothalamus, being exceedingly involved in all aspects of rudimentary motivational and emotional functioning is also important in arousal and the waking state (Ranson, 1939), and if the posterior regions are damaged there can result severe apathetic states.
Hence, these findings inplicate the forebrain (i.e. the amygdala, frontal lobes, thalamus, hypothalamus) in maintaining forebrain arousal. Nevertheless, even in this regard the forebrain is dependent on the midbrain and brainstem. It is the brainstem which contain nuclei and cell clusters which manufacture specific neurotransmitters which are directly responsible for maintaining and promoting behavior, emotional, cognitive, and cerebral arousal including sleep and dreaming.
DOPAMINE, NOREPINEPHRINE & SEROTONIN
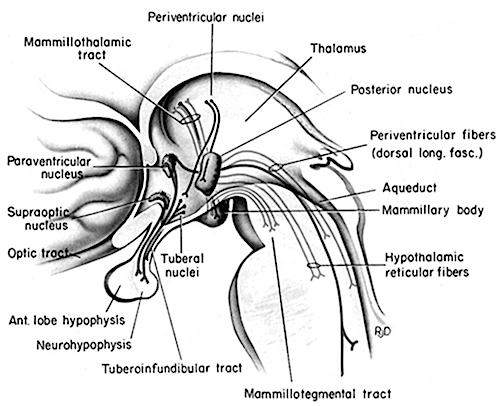
The pons contains neurons and nuclei that are responsible for manufacturing three principle monamine neurotransmitters, i.e. dopamine (DA), norepinephrine, (NE) -also referred to as catecholamines, and serotonin (5HT) which is an indolamine. DA is produced predominantly within the midbrain substantia nigra and tegmentum, whereas NE is manufactured and distributed by neurons located predominantly in the locus ceruleus, which is located in the pons. 5HT producing neurons and cell groups are found in both the pons and medulla (e.g. the dorsal and median raphe nucleus).
NOREPINEPHRINE
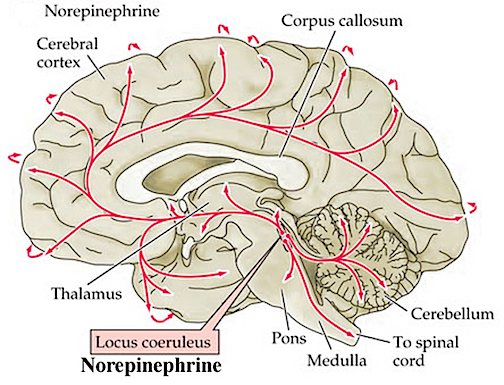
The NE neurons system is composed of three main neuronal clusters located in the pons and medulla; i.e. the dorsal medullary (A-2 cells), the locus ceruleus (LC, A-4, A-6 cells), and the subcoeruleus (A-1, A-3, A-5, A-7 cells). These nuclei give rise to ascending and descending monosynaptic projections which make diffuse and specialized selective contact with a variety of brain area via their highly branching axons (Dahlstrom & Fuxe 1965; Fuxe 1965; Levitt & Moore 1979). It has been estimated that via their diffuse projections a single NE neuron can make contact with up to 75,000 other neurons, and can contribute fibers to both the cerebral cortex, limbic system, and cerebellum (Cooper et al. 2014). However, this has also been disputed.
Broadly considered, the NE system projects to the midbrain colliculi and lateral and medial geniculate of the thalamus, the amygdala (which contains very high concentrations of NE), as well as throughout the brainstem where it influences auditory and visual functioning and is a major transmitter involved in facial expression. Indeed, the NE system subserved a wide range of functions, including regulation of cardivascular activity, micturation, respiration, sleep and dreaming, motivation, emotional expression, and mobilization in response to pleasure, fear, or stress (reviewed in chapter 30).
The locus ceruleus (LC)-NE system is clearly implicated in emotional functioning and contributes fibers (via the medial forebrain bundle) to the hypothalamus, amygdala, septal nuclei, hippocampus, thalamus, cingulate gyrus (Dalhstrom & Fuxe, 1965; Fuxe 1965) as well as the midbrain including the lateral geniculate and pulvinar of the thalamus, and the frontal and occipital lobe (see Foote et al. 1983; Morrison & Foote, 1986; Sakaguchi & Nakamura, 2005).
However, it maintains predominantly ipsilateral connections, including those which diffusely project to the neocortex, and there is some evidence which indicates that whereas DA is more greatly concentrated in certain left cerebral nuclei and brain regions, NE concentrations are greater in the right hemisphere, particularly within the right somesthetic nucleus of the thalamus (Oke et al. 1978). In this regard it is noteworthy that whereas left cerebral lesions will reduce left cerebral NE levels within the areas of injury, right hemisphere lesions can induce diffuse and bilateral NE reductions (Robinson 1979).
Locus ceruleus-NE neurons become highly active in response to novel or motivationally signficant somesthetic, visual, and auditory stimuli and NE levels are effected and rapidly metabolized by stressful and emotional experience (Foote et al. 1980), especially fear. Indeed, the neural circuitry involving the NE system makes it "uniquely situated to subserve all the known physiological correlates of fear" (Redmon & Huang 1979; p. 2154).
Hence, electrical stimulation of the LC can trigger severe anxiety and fear reactions. Conversely, ablation of the locus ceruleus results in a significant decrease in fear and anxiety reactions even in response to threatening stimuli (Redmod & Huang 1979). Similarly, NE-reuptake blockers (tricyclics) can reduce feelings of anxiety, whereas NE activators, such as piperoxane, can induce anxiety reactions in humans (Klein et al. 1978).
The NE system is also involved in the mediation of pleasure and reward, and is rapidly synthesized when subjects are engaged in pleasurable activities (Bliss & Zwanziger 1966). Conversely, low levels of NE are associated with a lack of pleasurable feeling; i.e. depression. Hence, NE is essential to the maintenance of the "pleasure circuit."
The NE system is also highly involved in arousal, including the sleep wake cycle, and the generation of REM, PGO activity, and dreaming (Chu & Bloom 2014). NE levels, therefore, fluctuate in cycles and demonstrate specific circadian rhythms which are also related to sleep and dreaming. For example, in humans and other primates, NE levels are lowest at 3 AM and gradually increase throughout the day until around 3 PM (Ziegler et al. 1976), which in some cultures corresponds to "nap" or "siesta" time.
SEROTONIN
Serontonergic (5HT) neurons exert widespread and often tonic and inhibitory influences on a variety of brain areas (Applegate, 1980; Jacobs & Azmita 1992; Soubrie, 1986; Spoont, 1992). This includes the modulation of fear and pain and emotional states such as depression, the inhibition of incoming sensory input, and the modulation of motor expression. That is 5HT restricts perceptual and information processing and in fact increases the threshold for neural responses to occur at both the neocortical and limbic level.
For example, in response to arousing stimuli, 5-HT is released (Auerbach et al. 2002; Roberts, 1984; Spoont, 1992) which acts to aid attentional and perceptual functioning so that stimuli which are the most salient are attended to. That is, 5HT appears to be involved in learning not to respond to stimuli that are irrelevant and not rewarding (See Beninger, 1989). These signals are filtered out and suppressed.
It has also been demonstrated that 5HT acts to suppress activity in the lateral (visual) geniculate nucleus of the thalamus and synaptic functioning in the visual cortex as well as the amygdala and throughout the neocortex (Curtis & Davis 1962; Marazzi & Hart 1955). By contrast, substances which block 5HT transmission -such as LSD- results in increased activity in the sensory pathways to the neocortex (Purpura 1956), which induces complex hallucinatory experiences.
As will be detailed below, 5HT is also implicated in the generation of sleep and dreaming, as well as the production of motor paralysis during sleep. For example, 5HT via descending influences, facilitates motorneuronal activity, while simultaneously decreasing or inhibiting sensory input (Morrison & Pompeiana 1965). In this manner, selective attention (via sensory filtering) can be maintained while the organisms is engaged in goal directed motor behavior. However, while sleeping and dreaming, internal sensory filtering is reduced (due to decreased 5HT) whereas motor functioning is inhibited -which prevents the subject from walking about and acting out their dreams (see below).
THE DORSAL & MEDIAN RAPHE-5HT NUCLEI
Serotonin (5HT) is produced by the raphe nuclei located in the pons and medulla, with the highest percentage located in the dorsal raphe (79%) with the bulk of the remainder produced by the median raphe. Thus, the main source for limbic and neocortical 5-HT is the dorsal (DR) and median raphe (MR) nuclei (Azamita, 1978; Azamitia & Gannon, 1986; Wilson & Milliver, 2018ab). Specifically, the amygdala, hypothalamus, basal ganglia, primary and association receiving areas, and the frontal lobe are innervated by the DR, whereas the hippocampus, cingulate gyrus, and septum receive their 5-HT from the MR nuclei (Azamita, 1978; Azamitia & Gannon, 1986; Wilson & Milliver, 2018ab).
The MR, however is diffusely organized and appears to exert a non-specific and global influence on arousal and excitabilty (Wilson & Molliver, 2018ab). The DR is much more discretely organized and can exert highly selective inhibitory or excitatory influences and plays a role in the coordination of excitation in multiple functionally related areas including the frontal lobes and amygdala (Wilson & Molliver, 2010ab). Because of the manner in which they are organized, the DR and MR can exert select inhibitory influences so as to engage in perceptual filtering in one or a variety of areas while simultaneously exciting yet other multiple regions which in turn aid selective attention and in the creation of specific neuronal networks.
NE & 5HT INTERACTIONS: FEAR, PAIN, STRESS
The 5HT and NE systems appear to interact in regard to a number of different functions including arousal, sensory and emotional processing, and sleep and dreaming, and (in conjunction with brainstem enkephalin) the experience and modulation of painful experience including depression. For example, it has been repeatedly and widely reported that altered brain 5HT turnover and reduced 5HT levels are often discovered postmortem among individuals who have committed suicide (e.g. Arranz et al. 2009). Moreover, some individuals commit suicide because of feelings of chronic stress and emotional turmoil, and an inability to experience pleasure in living -which in turn implicates the NE as well as 5HT system.
PAIN & STRESS
Somatic and visceral sensations are transmitted to the cerbrum by way of the spinal cord and brainstem. At the spinal level these signals are transmitted via the dorsal horns where collateral fibers from the peripherally located somatic receptors enter and then terminate near the substantia gelatinosa, thereby forming and giving rise to the lateral spinal tract which transmits pain and thermal impulses to the somesthetic (VPL) thalamus which contains high concentrations of NE -particularly the right VPL (Oke et al. 197).
However, descending LC-NE and raphe-5HT fibers also terminate in the substantia gelatinosa -which is also rich in opiate receptors- (Dalhstrom & Fuxe 1965). In response to nocioceptive impulses, both NE and 5HT (as well as the brainstem enkephalins) act to reduce and inhibit the transmission of these impulses (Headley et al. 1978). Similarly, stimulation of the raphe nucleus induces anylgesis and direct application of 5HT depresses dorsal horn reactivity to noxious stimuli.
In this regard, not only does NE and 5HT serve to counteract the reception of painful stimuli, they also act to arouse and mobilize the organism when experiencing pain or fear. As noted, stimulation of the LC can induce fear reactions as well as defensive behavior including flight (Redmond & Huang 1979). In this manner fear can also become associated with stimuli that induce pain and both can be associated with escape. However, in response to chronic fear, pain, or stress, 5HT and NE levels begin to rapidly decrease (see chapter 30).
STRESS & PSYCHOSIS.
In response to highly stressful, fearful, or painful stimuli, 5-HT levels, at least initially are increased which results in numbing, analgesia, and a loss of pain perception (Auerbach et al. 2002; Roberts, 1984; Spoont, 1992). Under conditions of prolonged stress, pain, or fear, 5-HT is rapidly depleted. In consequence, the individual may begin to feel overwhelmed and unable to appropriately process incoming sensory stimuli. Soon thereafter (i.e. following chronic stress) they may also demonstrate a heightened fear and startle response (Davis, 1984) as well as social withdrawal (Raleigh et al. 1983).
With continued high stress, sensory filtering is reduced, neurons in the amygdala, hippocampus, and inferior temporal lobe cease to be inhibited, which in turn can result in dream-like states, hallucinations (Joseph, 1988a, 1992a, 1998b, 2014d; Spoont, 1992), depersonalization (Sallanon et al. 1983) and associated psychotic and emotionally disturbed states (see chapter 30).
Under conditions of chronic emotional stress, 5-HT begins to be depleted. However, not just trauma, but repeated and chronic sexual activity is also associated with significant reductions and depletions in 5-HT (Spoont, 1992; Zemlan, 1978), which might explain why individuals who are repeatedly sexually molested or assaulted, may develop PTSD, dissociative and amnesic states, and psychosis (chapter 30).
In addition, stress, be it due to combat, cold, prolonged swimming, or pain, is associated with the depletion of NE (Bliss et al. 1968). With the exception of strenuous physical activity, fear and stress can reduce NE concentrations by up to 50%.
In fact, even a single trauma may result in alterations in NE and 5HT neurtransmitter turnover and release, which in turn can influence and increase the size of both the pre and postsynpatic substrates (Goelet & Kandel, 1986; Krystal, 2011) and even gene expression. This may induce not only alterations in learning and memory, but increased emotional reactivity and alarm responses that may persist indefinitely (Charney et al. 1993; Goelet & Kandel, 1986).
Under chronic conditions, effected neurons and associated neural networks mayalso become adapted to reduced 5-HT and NE levels, thereby increasing the likelihood of enhanced startle reactions, social withdrawal, feelings of depersonalization, hallucinatory states, sleep disturbances, increased REM latency, and other associated symptoms associated with a chronic post traumatic stress disorder (Carlson & Rosser-Hogan, 2018; Cassiday et al. 1992; Charney et al. 1993; Foa et al. 2015; McNally et al. 2011; Putnam, 2002; Wilcox, et al. 2016; Van der Kolk, 2005). It is thus noteworthy that blockage of 5-HT reuptake (which increases the amount of 5-HT in the synapse) acts to reduce the symptoms of post-traumatic stress disorder (Hollander et al. 2011).
DEPRESSION, 5HT & NE
As is now well known, major factors in the pathogenesis of depression include disturbances in the LC-NE and 5HT systems (Arranz et al. 2009; Delgado et al. 2009). For example, a disturbance in NE synthesis and metabolism, regardless of its cause, will disrupt limbic and cortical functioning and arousal, reduce neuroendocrine activity, and thus the organisms ability to mobilize its defenses in response to stress, adverse experience, or even self-defeating thoughts. Every component of social-affective expression will be altered, especially that related to the experience of pleasure and reward -a condition conducive to depression.
Similarly, when 5-HT levels are reduced individuals may become depressed. There is also an increased tendency to respond to non-rewarding situations (Sourbrie, 1986) and to continue to respond regardless of punishment (Spoont, 1992). Effected individuals may fail to avoid or may even be drawn to abusive or potentially frightening or traumatic situations, and may seem helpless to alter their behavior, e.g. learned helplessness (see Charney et al. 1993; Krystal 2011). Indeed, reduced 5-HT has been repeatedly noted in the brains of those who have committed suicide and taken their lives in a violent fashion (e.g. Brown et al. 1982; Cronwell & Henderson 1995).
Although it is difficult to classify a depressive episode or psychosis strictly in terms of a biochemical abnormality, it has been known for over two decades that there are at least two subgroups who differ in regard to depressive symptoms and NE and 5HT activity levels (Goodwin et al. 1978; Van Dongen 1982; Van Praag 1982). For example, those with an NE depression may behave in a more emotionally labile and expressive manner, whereas those with a 5HT depression may demonstrate a much more profound motor retardation coupled with social withdrawal and confusion due to sensory overload.
Moreover, both groups respond differently to pharmacological treatment. For example, some respond to best to impramine which inhibits NE reuptake (thus increasing NE levels in the synapse), whereas others react best to amitriptylline which potentiates 5HT (Goodwin et al. 1978; Van Praag 1982) and inhibits NE reuptake. This suggests that among some individuals NE and 5HT may simultaneously contribute to a distinct form of depression.
It has also been argued that reduced 5HT does not cause depression per se and is not linearly related to the level of depression (Delgado et al. 2009). Rather 5HT may be a predisposing factor in depression which in turn may be related to a postsynaptic deficit in 5HT utilization or binding affinity (Arranz et al. 2009; Delgado et al. 2009).
DREAMING, SLEEP & RHYTHMIC & OSCILLATING BRAINSTEM ACTIVITY
As noted, many of the functions performed by the brainstem are not only reflexive and stereotyped but characterized by specific oscillating rhythms. For example, the sex act involves the temporal-sequential insertion and movement of the male penis and rhythmic motion of the hips, whereas walking involves the rhythmic control of the extremities. As noted in chapter 5 (and above) over the course of neuronal evolution dorsally situated neurons, including those which came to form the pineal body, and portions of the thalamus and midbrain, were highly responsive to light and in consequence developed circadian and rhythmic cycles of activation.
Hence, a considerable number of neurons located in the midbrain and pons display a bursting, oscillating rhythm -a pattern that also accompanies controlled locomotion and a host of other cyclic activities. This includes 5HT and NE neurons which are also involved in controlling a wide variety of rhythmic functions, including respiration, chewing, and micturation (Skinner & Garcia-Rill, 2011) as well as sleep and dreaming. Presumably, these interconnected monoamine, cholinergic and catecholaminergic neuronal clusters are out of phase which induce cyclic oscilations (Skinner & Garcia-Rill, 2011) including those involved in sleep and dreaming.
REM-OFF & REM-ON NEURONS
It is now well known that the visual-emotional hallucinatory aspects of dreaming occur during REM, whereas more thought-like and verbal ideational patterns are produced during NREM. A variety of nuclei and brain regions appear to be involved in the production of REM, i.e. the amygdala, hippocampus, right temporal lobe, and especially brainstem nuclei located in the lateral and medial pons.
Specifically, cholinergic neurons located in the lateral pons, and neurons located in the medial pontine reticular formation appear to be the locus for REM-on neurons which initiate and/or maintain the production of REM sleep (see Steriade & McCarley, 2011; Vertes 2011). That is, during the production of REM and paradoxical sleep, there is increased cholinergic activity in these regions, whereas with the termination of REM, these same neurons have greatly reduced their activity.
Similarly, when cholinergic agnonists are injected into the medial pons, REM sleep is produced (Steriade & McCarley, 2011). When cholinergic agnonists are injected into the lateral pons, muscle atonia is produced -a common characteristic of paradoxical sleep. Thus, REM on cells tend to be cholinergic and become very active during paradoxical sleep, induce muscle atonia, but markedly decrease their activity during slow wave and non-REM sleep at which point atonia also tends to disapear.
In contrast, REM-off neurons, which tend to be located in the medial raphe nucleus (which contain 5HT neurons) and in the locus coeruleus (located at the midbrain-pons junction and which contain NE neurons) are highly active during waking but then begin to reduce their activity during slow wave sleep and then significantly decrease their activity with the initition and onset of REM and the production of pontine, lateral geniculate, occipital activation; i.e. PGO waves (see Steriade & McCarley, 2011; Vertes 2011). Conversely, experimental cooling (and thus deactivation) of the LC consistently and repeatedly induces REM sleep (Cespuglio et al. 1982).
Hence, these REM-Off neurons appear to suppress REM and PGO activity and interfere with the onset of dreaming and paradoxical sleep, whereas REM-on neurons initiate the opposite sleep phase in which case the brain appears to be highly active (e.g. the amygdala, pons-geniculate,-occipital lobe) and the individual begins to dream.
REM-on and REM-off neurons, therefore, appear to oscillate in a rhythmic fashion thus inducing sleep, dreaming, and waking. When the REM-off neurons cease to fire, the REM-on neurons (which are predominately cholinergic) become highly active until a REM episode is produced. However, as REM-on neuron activity decreases, REM-off activity increases, thus setting into motion a continuous 90 minute cycle of REM - Non-REM sleep alteration.
In addition, and as noted above, whereas NE and 5HT (i.e. sensory motor neurons) play a significant role in the generation of dream (paradoxical) sleep, GABA and glutamate (modulatory) pace maker neurons, also play a significant role in the oscillation between REM and NREM, in part by acting on REM-off neurons. That is, REM-off neurons are turned off by modulatory inhibition (GABA), whereas REM-on neurons are turned on by modulatory excitation (glutamate).
However, these latter oscillating on/off activities appear to take place not only in the brainstem, but in the hypothalamus. That is, because the hypothalamus (via the suprachiamatic nucleus) is also involved in pace maker functions, whereas the posterior hypothalamus is involved in sleep (Sherin, et al., 1996) there is some evidence to suggest that this GABAergic inhibition may actually begin in the hypothalamus thus initiating sleep onset and the first sleep stage (NREM). GABAergic inhibitory actions then slowly spreads to and then throughout the brainstem only to become self-inhibiting and replaced by glutamate (excitatory) secretion which triggers REM-on neurons, and thus the next stage of sleep (REM).
Because of these on/off excitatory/inhibitory activities, during NREM NE and 5HT levels decline (Hobson, et al., 1975; Lydic, et al., 2018). However, ACh levels actually increase (Hobson, et al., 1975; Lydic, et al., 2018) and in fact remain at high levels during REM and the production of pontine, lateral geniculate, occipital activation; i.e. PGO waves (see Steriade & McCarley, 2011; Vertes 2011). As ACh is also implicated in memory, this may well explain whey recent memories tend to become incorporated in dreams.
PGO WAVES
Paradoxical sleep and the production of dream states are associated with the development of high levels of activity within the pons, the lateral geniculate nucleus of the thalamus, the occipital lobes, and association and motor cortices (Steriade & McCarley, 2011) -activity collectively referred to as PGO waves (see Hobson et al. 1986). Presumably, as these neurons are activated, visual imagery is produced as these regions are concerned with analyzing visual input as well as visual scanning of the environment.
However, PGO waves, although generated in a variety of brainstem reticular neuronal clusters, are also induced by amygdala activation (Calvo, et al. 2005). In addition, during REM, the hippocampus begins to produce slow wave, theta activity (Jouvet, 1967; Olmstead, Best, & Mays, 1973; Robinson et al. 1977), which is associated with long-term potentiation which is associated with learning and memory (see chapter 14).
The amygdala, in concert with the hippocampus, therefore appears to contribute emotional and visual imagery and experiential impressions to the dream state--a theory first proposed by this author (Joseph 1982, 1988a, 1992a) and which has subsequently been embraced by numerous investigators. Indeed, via the extensive interconnections the amygdala maintains with the brainstem and the geniculate and occipital lobes, and its capacity to induce high levels of arousal, this nucleus is ideally situated to initiate if not produce (again in conjunction with the hippocampus) the auditory-visual imagery characteristic of the dream state including all aspects of related emotion (see chapter 13). Moreover, the amygdala also receives 5HT and NE from the brainstem (with which it is intimately interconnected), thereby creating an elaborate feedback circuit.
The lateral pontine reticular formation is also involved in producing hippocampal theta during REM (Vertes, 1984). As detailed in chapter 14, hippocampal theta is associated with arousal, and theta activity may represent the introduction and transformation of hippocampal based memory-imagery into dream imagery. However, the production of hippocampal theta may also help insure that whatever is experienced during REM sleep is stored in such a fashion that dream stimuli do not become committed to memory.
Nevertheless, in regard to dream sleep, the amygdala and hippocampus appear to be largely dependent on the brainstem, for when disconnected (see below), although the brainstem continues to demonstrate a sleep wake cycle, the forebrain will cease to demonstrate paradoxical sleep activity. Moreover, although the amygdala can induce PGO activity, in general, neurons in the upper medial pontine reticular formation begin to fire well in advance of the development of PGO waves (McCarley & Ito, 1983). It is also these pontine neurons (in conjunction with the cranial nerves and midbrain) which initiate eye movements during REM. Typically, PGO waves reach their maximum amplitude in the lateral geniculate nucleus of the thalamus which (like the amygdala) are also cholinergically responsive (Steriade & McCarley, 2011).
It is noteworthy that PGO-on neurons also become active during waking when eye movements are made (Nelson et al. 1983). Therefore, PGO waves are not strictly a phenomenon associated with sleep, but can occur when the subject is emotionally aroused and perhaps looking anxiously around for the source of stimulation -which again implicates the amygdala in their production.
Characteristically, however, PGO waves and their transfer to the thalamus has two distinct states (Steriade & McCarley, 2011). That is, they first appear during the last stages of synchronized sleep just prior to REM, and then again, accompanying the development of desychronized sleep and REM.


SYNCHRONIZED, SLOW WAVE SLEEP
The defining feature of synchronized (or quiet, Non-REM) sleep is large amplitude rhythmic EEG waves of varying frequencies (from slow to fast). Even during waking, however, and when highly aroused or alert, some cortical areas may display synchronized EEG activity, whereas even during REM sleep some cortical areas display synchronized activity (rviewed in Steriade & McCarley, 2011). Presumably in that some cortical areas demonstrate slow wave activity whereas yet others are highly aroused, is a reflection of selective attention and discrete neocortical activation. Some cortical regions becomes suppressed thus narrowing the range of incoming stimuli whereas other specific regions remain activated in order to process incoming information and/or for the purposes of contributing to whatever mental process may be taking place.
Steriade and McCarley (2011) propose that the development of synchronized sleep is dependent on the removal or lessening of brainstem input and is a passive rather than an active process. That is, fluctuations and decreases in cholinergic and non-cholinergic activity within the reticular formation decreases thalamic activity thus producing sleep spindles in the reticular thalamic nucleus followed by synchronous activity. However, with increases in cholinergic and non-cholinergic activity, thalamic activity increases, and synchronous EEG activity is replaced by desynchronization and the production of REM (see also Hobson et al. 1986; Vertes 2011).
THALAMIC CONTRIBUTIONS
Given that the thalamus acts to relay sensory input to the neocortex, not surprisingly thalamic neurons are also involved in synchronized and paradoxical sleep. Specifically thalamic neurons inhibit sensory transfer to the neocortex during the synchronized sleep stage as well as during the transition from wakefulness to sleep and when drowsy; otherwise the individual would keep waking up due to neocortical activation.
Some thalamic neurons, however, act to enhance sensory transfer during desynchronized sleep (Steriade & McCarley, 2011) as well as during waking. Presumably this allows for sensory stimuli to become incorporated into and to in fact induce dream states.
Specifically, the reticular thalamic nucleus appears to also act as a "pacemaker" center for the development of sleep spindles (i.e. EEG waves which wax and wane between 7 and 14 Hz) and which herald the onset of sleep (Steriade & McCarley, 2011). Spindles are typically associated with loss of consciousness and thalamic inhibition and thus loss of information transfer to the cortex. Moreover, spindles originate in the reticular thalamus, which in turn projects to other thalamic neurons. These thalamic nuclei are indirectly under the control of the frontal lobe and dorsal medial thalamus (see chapter 19).
MOTOR INHIBITION DURING SLEEP
The frontal lobe as well as brainstem motor areas become activated during REM (see Steriade & McCarley, 2011). However, although motor commands may be initiated and even transmitted from the neocortical motor centers, they cannot be acted out during REM due to the production of muscle atonia. Muscle atonia is produced, in part, via descending 5HT influences on the spinal cord (see above).
Specifically, during the transition from slow wave to paradoxical sleep motor neurons in the brainstem and the spinal cord come to be actively inhibited (Chandler, et al. 1980; Chase & Morales 2002). The result is muscle atonia, or what has also been referred to as sleep paralysis. This condition is almost exclusively associated with REM sleep (Chase & Morales 2002).
Presumably these inhibitory influences arise within the pontine and bulbar brainstem reticular formation and via descending 5HT influences so as to prevent an individual from motorically acting out their dreams and possibly injuring themselves (e.g. sleep walking). Indeed, the dorsal lateral portion of the pontine tegmentum has been repeatedly implicated as responsible for muscle atonia during sleep (see Steriade & McCarley, 2011).
By contrast, lesions of the pontine reticular formation (with sparing of the locus coeruleus) abolishes muscle atonia (Hendricks, et al. 1982; Jouvet, 1979). In consequence, during REM sleep, the animal will move about and engage in semi-purposeful behaviors, including walking, orienting movements, and attack -as if the animal were acting out its dream (Hendricks, et al. 1982).
BRAINSTEM SLEEP DISORDERS: NARCOLEPSY & CATAPLEXY
NARCOLEPSY
Narcolepsy is a syndrome that is often chateracterized by excessive daytime sleepiness, and may be accompanied by partial or complete and sudden attacks of REM sleep where the patient was quite awake just moments before (Broughton 2011; Guilleminault, 1989). In some instances patients may also experience sleep paralysis, hypnagogic hallucinations, and cataplexy. These daytime attacks sometimes follow the 90 minute cycle characteristic of REM sleep. Abnormalities involving rapid eye movements (REM) and motor disturbances such as muscle twitching and persistence of muscle tone are not uncommon (Schenck & Mahowald 1992), and the latency of REM sleep is often very short.
This condition is often treated with amphetamines and with tricyclic anti-depressants. It is also believed to be secondary to abnormal concentrations of NE and DA. However, narcolepsy has several different components.
Excessive Daytime Sleepiness (EDS). EDS involves the sudden sleep onset of REM involving dreaming. These sleep isodes tend to last from a few minutes up to half an hour. Sleep Paralysis (SP). SP usually occurs upon waking, such that the person experiences a paralysis of the voluntary muscles. They cannot open their eyes, move their arms or legs, and cannot speak. Some are terrified by these experiences. Presumably, many people have experienced this at least once in a mild form. The condition tends to last just a few minutes.
Presumably SP is due to the abnormal prolongation of activity within the dorsal lateral portion of the pontine tegmentum, a region which is normally responsible for producing muscle atonia during sleep (see above).
Hypnogogic Hallucinations. Some patients experience hypnogogic hallucinations upon waking and while seemingly completely conscious. These can be exceedingly vivid and realistic and involve auditory, kinesthetic, and somesthetic as well as visual hallucinations. The effected individual may see what they believe to be ghosts or even intruders attempting to attack them. Hypnogic hallucinations (and sleep paralysis) appear to represent the intrusion of REM into wakefulness.
SOMNAMBULISM
Sleep walking is most common among children, and presumably up to 15% of all children sleep walk at one point in their lives (see Kavey, et al. 2011). Among adults, it occurs in less than 1% of the population.
Somnambulism occurs during the first third of the night during NREM sleep. Usually patients have no recollection or only a vague memory of walking about (Kavey, et al. 2011). While sleep walking some patients may behave in an aggressive or agitated manner, scream and engage in fighting or fleeing behaviors and may fall or strike and injure themselves. Fragmentary memories may include being chased.
Adults who sleep walk often have a history of sleep walking during childhood (Kavie et al. 2011). Some patients demonstrate epileptiform EEGs and/or suffer form atypical nocturnal complex partial seizures (Pedley & Guilleminault 1977).
REM Sleep Behavior Disorder.
REM sleep behavior disorder is associated with narcolepsy and REM sleep (Schenck & Mahowald, 1992), and is a form of motor disinhibition accompanied by often violent behaviors. Patients appear to be acting out their dreams. It usually effects men over 60, though it is also seen in children with obvious brainstem dysfunction (see Schenck & Mahowald, 1992). However, when it accompanies narcolepsy this disorder tends to have it's onset in men in their late 20's (Schenck & Mahowald, 1992).
Presumably somnambulism and REM sleep behavior disorder are a consequence of disinhibition of the brainstem and spinal motor neurons which are normally inhibited during REM. That is, it is likely that the dorsal lateral portion of the pontine tegmentum, a region which is normally responsible for producing muscle atonia during sleep, fails to induce muscle atonia such that the person begins to walk about and act out their dreams. As noted above, destruction of this nuclei also results in motor disinhibition during REM such that the subject begins to move about as if actively engage in purposeful behavior.
CATAPLEXY
Cataplexy involves a sudden loss or decrease of muscle tone; atonia. Hence the effected individual may suddenly fall to the ground, sometimes injuring themselves. In mild cases the jaw may suddenly become slack and the knees become weak and may buckle. In severe cases the patient immediately falls to sleep, and enters REM which is accompanied by dream mentation. Often cataplexic attacks are induced by sudden surprise or attacks of emotion, including anger or laughter.
Cataplexy is generally considered a component of narcolepsy and the production of REM and the loss of muscle tone indeed suggest a brainstem loci for this disorder. However, the influence of emotion in inducing cataplexic attacks and the fact that some individuals become rigid and stiff, almost catatonic, suggests that the amygdala and the basal ganglia and perhaps, indirectly, the medial supplementary motor areas (SMA) of the frontal lobe may also be contributory. That is, in some respects this disorder is reminiscent of abnormalities within the basal ganglia and SMA -nuclei which are significantly influenced by the amygdala.
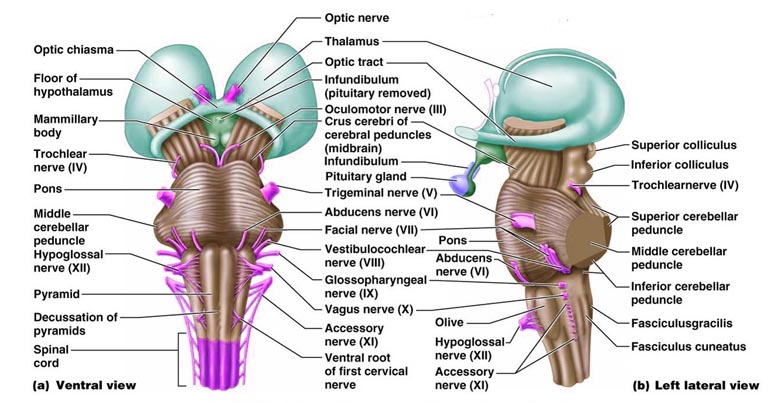
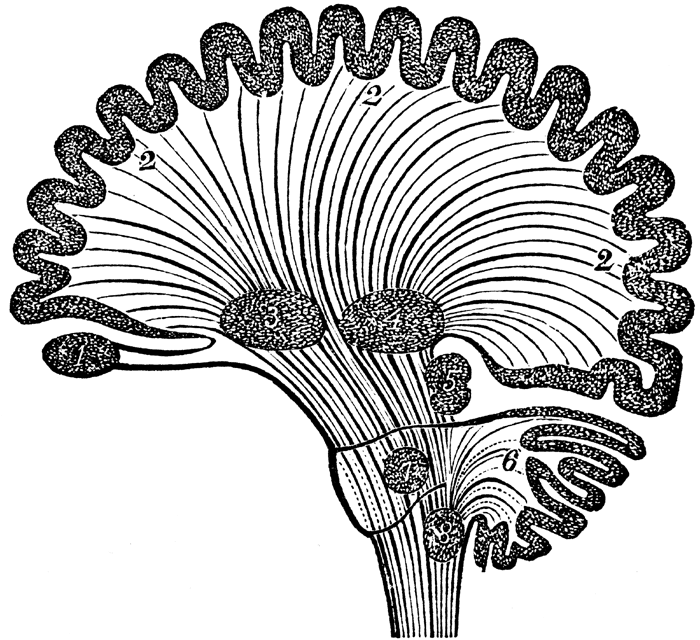
THE CRANIAL NERVES OF THE PONS
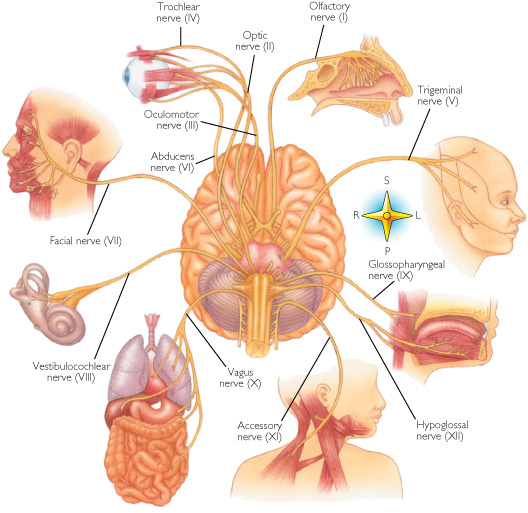
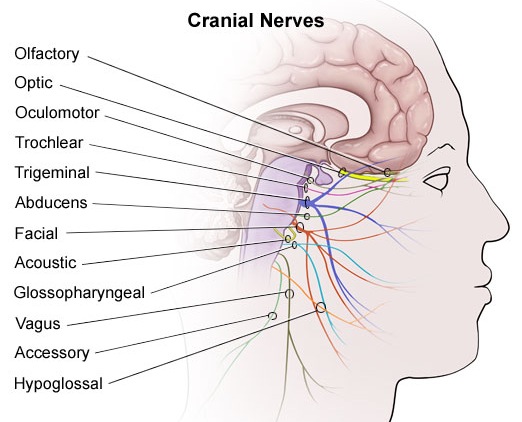
TINNITUS, DEAFNESS, DIZZINESS, VERTIGO
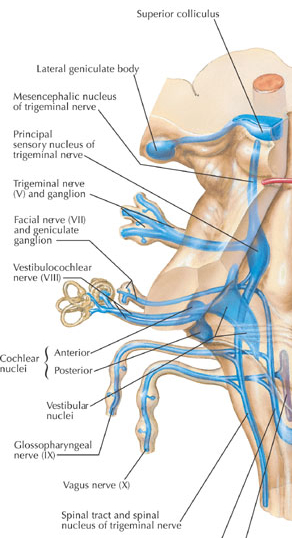
A portion of the 8th nerve arises from the cochlear nuclei so as to form the auditory branch of the 8th nerve which in turn bifurcates and innervates the ampulla, utricle, saccule and cochlear duct of the semi circular canals of the inner ear. It is via the acoustic nerve that auditory stimuli are relayed to the ventral and dorsal cochlear nuclei for further transduction prior to transfer to the inferior colliculus of the midbrain, and the medial geniculate nucleus of the thalamus. Hence, lesions involving this nerve or the cochlear nucleus can give rise to significant hearing problems, including deafness and tinnitus --ringing in the ears which may be reported as buzzing, humming, whistling, roaring, hissing, or clicking.
Tinnitus
There are two general types of tinnitus, vibratory, and non-vibratory. Non-vibratory tinnitus is actually quite common, being due to contraction of the muscles of the inner and middle ear, but is usually masked and may only be heard at night or when in a very quiet environment. In contrast, vibratory tinnitus is most often due to disorders of the inner ear, ossicles of the middle ear, tympanic membrane, or 8th nerve, and may be accompanied by deafness in one ear.
Deafness
If a patient complains of deafness, it must be determined if this is due to otosclerosis, chronic otitis, occulsion of the external auditory canal or eustachian tube, in which case the problem is conductive and is not related to nerve damage. By contrast, disease of the cochlear or of the cochlear division of the 8th nerve, or its central connections results in sensorineural (or nerve) deafness.
These two distinct disorder can be easily differentiated with the help of a tuning fork. When struck, it is placed at the center of the forehead or on the mastoid bone behind the ear so that the vibration can bypass the middle ear and can be mechanically conveyed to the inner ear so as to excite auditory impulses.. If the condition is due to nerve deafness, the sound is localized in the normal ear and fails to be perceived by the effected ear. If the disorder is conductive, the sound is heard as louder in defective ear. If there is an obstruction or a middle ear abnormality (vs a nerve disorder) then the sound is heard louder in that ear due to dampening of noise via the air conduction block. If there is a lesion of the nerve then the sound is heard in the opposite of normal ear only.
THE EIGHTH (VESTIBULAR) NERVE
The vestibular division of the 8th nerve arises from the vestibular nerve which innervates the labyrinth and the maculas of the saccule and utricle and the ampullae of the semicircular canals. As noted in chapter 5, the vestibular system was derived from the lateral line system, and in turn gave rise to the evolution of the cerebellum.
The major concern of the vestibular division is not hearing or vibratory perception, however, but determination of the body's position in visual-space so as to maintain equilibrium during movement. All sensory receptors for the vestibular system are located in the membranes of the labyrinth of the internal ear, and in the vestibular ganglion which is in the internal auditory canal. Thus it can determine changes in position via alterations in fluid balance within the semi-circular canals.
Lesions of the vestibular receptors, their nerves, or central connections cause abnormal sensations of movement, vertigo, nausea, tendencies to fall, dizziness, and motion sickness, etc.
The vestibular system also provides information about the position of the head and correlates head and eye movements with somatic muscle activity. Together with the descending medial longitudinal fasciculus (MLF) and vistibulospinal tracts, the vestibular system is able to mediate the postural reflexes. In part it is able to accomplish this via rich interconnections with those cranial nerve nuclei (via the MLF) which subserve eye movement (nerves VI, IV, & III). Disease involving these tissues can therefore cause nystagmus.
Following injuries to this system, patients may complain of to-and-fro or up-and-down movements of body, or floors, and noted that the walls seem to tilt or sink or rise. When walking there may be feelings of unsteadiness such that they veer to one side. Or there may be a feeling of being pulled or drawn--a feeling of impulsion. There also may be a disinclination to walk (particularly during an attack), a tendency to list to one side, and the condition may be aggravated by riding in a vehicle. Some disturbances may occur only for a few seconds, or after lying down or sitting up, turning, etc. When less severe the patient may merely veer to one side while walking.
The vestibular system is also concerned with eye movement, for it is also via ocular signals that the position of the head and body in space can be determined. Hence, vestibular dysfunction can include difficulty focusing or fixating on objects while walking, or when the object is moving. This is due to a loss of stabilization of ocular fixation by the vestibular system during body movement and is caused by an inability to integrate visual with vestibular input. These functions are normally made possible through rich interconnections (via the MLF) between the vestibular system and the 6th, 4th and 3rd nerves which subserve eye movement, as well as the bilateral interconnections between these regions and the cerebellum.
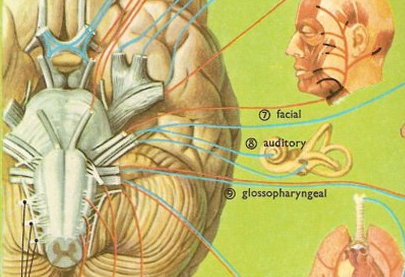
ORAL-FACIAL MOVEMENT & SENSATION
THE SEVENTH CRANIAL (FACIAL) NERVE
The 7th nerve, and the brainstem nuclei which it innervates, is concerned with facial movement, including elevation of the eyebrows, retraction of the lips, closure of the auditory canals, as well as with gustatory sensation. Injuries to the 7th nerve can therefore produce a lip retraction, eyebrow lifting or eyelid closure paralysis; i.e. Bell's palsy. Patients have difficulty or are unable to wrinkle the forehead, purse their lips and show their teeth,and the corner of the mouth may droop.
In addition, whereas cranial nerves 9 and 10 innervate the taste buds of the posterior 3rd of the tongue, cranial nerve 7 innervates the anterior 2/3s. Hence, a lesion of the 7th nerve can result a disturbance of taste sensation.
In addition to the muscles of the face, the 7th nerve also innervates the stapedius muscle, which acts to dampen excessive sound via inhibition of the movement of the ossicules. If the 7th nerve is injured, the stepedious may become paralyzed and the patient may report that sounds are uncomfortably loud. However, disturbances of hearing are most usually associated (at least at the brainstem level) with damage involving the 8th cranial nerve.
As to oral and facial movement including swallowing and speaking, the functional integrity and participation of the 5th nerve is also important for it controls the jaw. In this regard, the 5th, 7th, 9th, 10th, and 12th nerve and associated nuclei frequently act in concert regarding oral-facial, jaw, head and shoulder movement, and are richly interconnected.
THE FIFTH CRANIAL NERVE: TRIGEMINAL
The 5th nerve is the largest of the cranial nerves and innervates the trigeminal nucleus within the medulla. It is concerned with jaw closure, as well as chewing, grinding, and lateral movement of the jaw. In this regard the 5th nerve also acts in concert with the 7th nerve which innervates all the muscles involved in facial expression. These muscles control the size of every facial aperature, including the auditory canals. A lesion involving this nucleus, therefore, can result in difficulty chewing, and if severe, atrophy and complete paralysis of the left or right temporal and masseter muscles. Paralysis and atrophy are always ipsilateral to the lesion.
The general somatic afferent components of the 5th nerve also mediate the general sensory modalities for the face, teeth, and mouth, and the mucous membranes of the nose, check, tongue, and sinuses. These general sensory functions include proprioception, touch, pain, and temperature.
EYE MOVEMENT THE SIXTH CRANIAL NERVE: ABDUCENS
The abducens is a motor nerve that innervates the lateral rectus muscle of the eye, and is part of a collection of fibers which are found with the loop of fibers that also form the facial nerve. However, fibers from the abducens nuclei also ascend the brainstem and terminate on neurons belonging to the occulomotor complex which innervates the medial rectus muscle. It is therefore responsible for horizontal eye movements. In large part, the pontine center for lateral gaze and the abducens nuclei form a single entity (Carpenter 2018). In addition, the abducens is linked to the pontine/midbrain center for vertical gaze (the rostral interstitial nucleus of the MLF).
Thus abducens nerve and nuclei are the pontine centers which control lateral eye movements outward to the right or to the left. Injuries to the 6th nerve can therefore produce a lateral gaze paralysis as well as a paralysis of the lateral rectus muscle which results in double vision (horizontal diplopia).
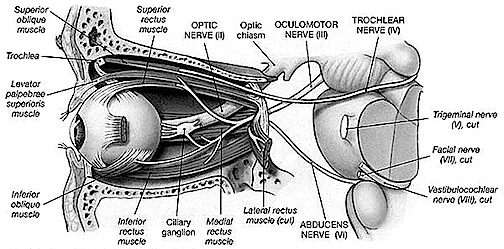
CRANIAL NERVES OF THE MIDBRAIN
EYE MOVEMENT
Movement of the eye is dependent on the functional integrity of the posterior parietal neocortex, the frontal eye fields of the lateral frontal lobes, the midbrain visual colliculi, cerebellum, and the cranial nerves and nuclei of the upper pons (cranial nerve VI) and midbrain (nerves IV & III). All pathways mediating saccadic, pursuit, and vestibulocular movements, including those originating in the forebrain and midbrain, then converge onto the pontine centers for horizontal gaze.
THE 4TH NERVE: TROCHLEAR
As noted, the 6th (abducens) nerve and nuclei are the pontine centers which control lateral eye movements outward to the right or to the left. By contrast, the 4th (trochlear) nerve and nucleus is located just caudal to the inferior colliculi, and innervates the superior oblique muscle of the eye; a muscle which has three actions: depression, abduction, intorsion. The 4th nerve therefore assists in moving the eye downward or inward.
THE 3RD NERVE: OCULOMOTOR
The 3rd (oculomotor) nerve is responsible for rotating the eye upward, downward, as well as inward and innervates all ocular rotary muscles (except for the lateral rectus and superior oblique). These include the medial, superior, inferior recti and inferior oblique.
The 3rd never also innervates the intraocular and smooth muscles of the pupil; i..e. the ciliary and pupilloconstrictor muscles. Lesions, therefore, may result in an inability to rotate the eye upward, downward, or inward, and the pupil (on the side of the lesion) may fail to respond to direct light.
In addition, the 3rd nerve innervates the levator palprebrae muscle which elevates the eyelid. When the third nerve has been injured this may result in levator palpebrae weakness and thus ptosis of the eyelid.
DISTURBANCES OF EYE MOVEMENT
Eye movement nuclei are all located in the pontine brainstem and receive input from (and transmit information to) the superior (visual) colliculus within the midbrain (Grantyn & Grantyn, 1976), and the frontal eye fields (Leichnetz, et al. 1984), which also projects to the superior colliculus (Segraves & Goldberg, 2005). As is well known, the frontal eye fields and the superior colliculus become activated immediately prior to the onset of an eye movement, and stimulation of these regions elicits eye movements and visual orienting reactions as well.
Hence, damage to these cortical areas or to any of these nerves or nuclei can result in significant visual disturbances. For example strabismus (or squint) is due to a brainstem lesion that results in muscle imbalance and thus improper alignment of the visual axes of the two eyes. A complete lesion of the oculomotor nerve causes ptosis (drooping of the upper eyelid) and an inability to rotate the eye upward, downward, or inward. Lesion of the 4th nerve results in weakness of downward movement of the affected eye and patients may complain of difficulty reading or walking downstairs. Head tilting to opposite shoulder is especially characteristic. Lesion of the 6th nerve results in paralysis of lateral or outward eye movement.
Most common causes of 3, 4, 6th nerve damage are tumors of the base of the brain, trauma to head, ischemic infarction of one of these nerves, and aneurysms of the circle of Willis. Sixth nerve palsies in children are due to neoplasm-pontine glioma. The fourth nerve is most commonly injured by head trauma. Third nerve damage is often due to compression by aneurysm, tumor, or temporal lobe herniation--enlargement of the pupil is an early sign.
As noted, cranial nerves 3, 4, and 6 are linked to the 8th (vestibular) nerve via the MLF. It is due to these rich interconnections that a lesion involving these regions and the MLF can cause vertigo, dizziness, as well as nystagmus and impairment of vertical fixation and pursuit. However, disturbances such as these also raise the possibility of cerebellar dysfunction.












