Rhawn Gabriel Joseph, Ph.D.
Brain Research Laboratory
BrainMind.com
THE RIGHT CEREBRAL HEMISPHERE
It has now been well established that the right cerebral hemisphere is dominant over the left in regard to the perception, expression and mediation of almost all aspects of social and emotional functioning (e.g. Borod, 1992; Cancelliere & Kertesz, 2017; Freeman & Traugott, 1993; Heilman & Bowers 1995; Heilman et al. 1985; Joseph 1988a; Tucker & Frederick, 2017; see below), including the recall of emotional memories (Cimino et al., 2013; Rauch et al., 2016; Shin et al., 2013). This emotional dominance extends to bilateral control over the autonomic nervous system, including heart rate, blood pressure regulation, galvanic skin conductance and the secretion of cortisol in emotionally upsetting or exciting situations (Rosen et al. 1982; Wittling, 2017; Wittling & Pfluger, 2017; Yamour et al. 20011; Zamarini et al. 2017). However, this dominance does not appear to extend to the immune system (Meador et al., 2009).
Further, the right hemisphere is dominant for most aspects of visual-spatial perceptual functioning, the recognition of faces including friend's loved ones, and one's own face in the mirror. Faces, of course, convey emotion.
Recognition of one's own body and the maintenance of the personal body images is also the dominant realm of the right half of the brain. The body image, for many, is tied to personal identity; and the same is true of the recognition of faces.
Visual-spatial, facial recognition, and body image dominance appears to be tied to a greater representation of these functions in the right vs the left hemisphere.
In part, it is believed that the right hemisphere dominance over social and emotional functioning is due to more extensive interconnections with the limbic system (Joseph, 1982, 2018a), including the fact that limbic system appears to be functionally and structurally lateralized (see chapter 13). For example, the appear to be more axonal connections between the neocortex of the right hemisphere and subcortical structures as the white matter connections are more extensive. The neocortex of the right hemisphere is also about 4% greater in size as compared to the left, the right amygdala is significantly (9%) larger than the left (Caviness, et al., 2013), whereas the left amygdala contains heavier concentrations of dopamine (Bradbury, Costall, Domeney, & Naylor, 1985; Stevens, 1992).
It has also been theorized that over the course of evolution and development, limbic social-emotional functions have come to be hierarchically subserved by the right cerebrum due in part to the initial earlier maturation of the non-motor portions of the right cerebral neocortex and due to limbic laterality (Joseph, 1982, 2018a, see chapter 13). This right hemisphere limbic dominance came to include the expression and representation of limbic language, thus providing the right cerebrum with a functional dominance in regard to the expression and comprehension of emotional speech as well as all aspects of emotional perceptions, such as the image of the body and the recognition of friends and loved ones.
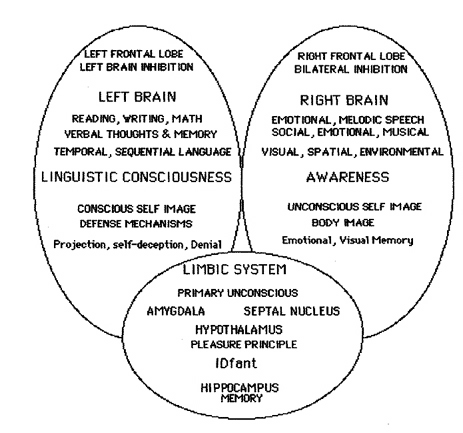
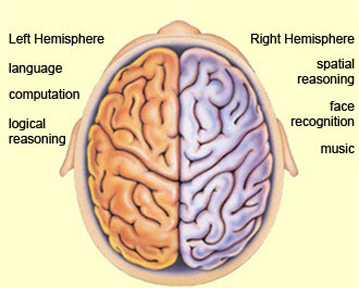
The right hemisphere, in fact, appears to maintain a realm of conscious-awareness which is completely different from that of the left. Right hemisphere mental functioning is more visual, spatial, emotional, personal, and non-verbal. By contrast, the mental realm of the left is more tied to language, including thinking in words.
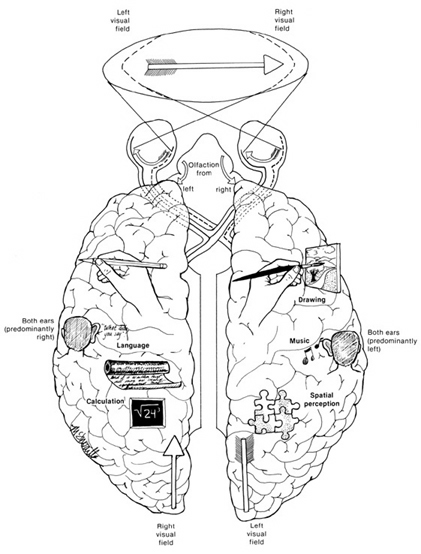
For example, expressive and receptive speech, linguistic knowledge and thought, mathematical and analytical reasoning, as well as the temporal-sequential and rhythmical aspects of consciousness, are associated with the functional integrity of the left half of the brain in the majority of the population (Frost, et al., 2009; Goodglass & Kaplan, 2009; Heiss, et al., 2009; Pujol, et al., 2009; Wagner et al., 1998). In contrast, the right cerebral hemisphere is associated with visual spatial, non-verbal and emotional memory (Cimino et al., 2013; Nunn et al., 2009; Ploner et al., 2009), and in fact demonstrates an increase in activity when recalling traumatic memories (Rauch et al., 1996, Shin et al., 2013) or when presented with stimuli which trigger positive or negative feelings (Teasdale et al., 2009), and when reporting sad memories or expressing feelings of depression (Abrams & Taylor, 1979; Cohen, Penick & Tarter, 1974; Deglin & Nikolaenko, 2005; Shagass et al.,1979).
The right hemisphere is also associated with nonlinguistic environmental awareness, visual-spatial perceptual functioning including analysis of depth, figure-ground and stereopsis, facial recognition, the maintanance of the body image (Bradshaw & Mattingley, 1995; Joseph, 2018a; Sterzi et al., 1993) and even the sense of smell.
As indicated by functional imaging, when presented with various odors, the right orbital area becomes highly active (Zatarre et al., 1992) whereas electrical stimulation of the right orbital frontal lobe evokes olfactory hallucinations (Munari & Bancaud, 1992).
The right hemisphere is dominant in the perception, expression and mediation of almost all aspects of emotional intelligence (Borod, 1992; Cimino et al., 2013; Joseph, 2018ab; Ross, 1993), including emotional vocalization and comprehension (Lalande et al. 1992; Ross, 2011; Shapiro & Danly, 1985; Tucker et al., 2017). In fact, although the left hemisphere is dominant for language, the right hemisphere continues to participate in language processing by evoking or sensing feeling, as demonstrated by functional imaging studies (Bottini et al., 2004; Cuenod, et al., 1995; Price et al., 1996). For example, the right temporal and parietal areas are activated when reading (Bottini et al., 2004; Price et al., 1996), and the right temporal lobe becomes highly active when engage in interpreting the figurative aspects of language (Bottini et al., 2004). This activity increases if the language is emotional.

LEFT HEMISHERE OVERVIEW
The left is also dominant in regard to most aspects of expressive and receptive linguistic functioning (Evers et al., 2009; Frost, et al., 2009; Heiss, et al., 2009; Pujol, et al., 2009) including grammar, syntax, reading, writing, speaking, spelling, naming, verbal comprehension, and verbal memory (Albert et al. 2001; Carmazza & Zurif, 1976; DeRenzi et al. 1987; Hecaen & Albert, 1979; Heilman & Scholes, 1976; Kertesz, 2013ab; Milner, 1970; Njemanze, 2013; Vignolo, 2013; Wagner et al., 1998; Zurif & Carson, 1970).
In addition, the left hemisphere has been shown via dichtoic listening tasks, to be dominant for the perception of real words, backwards speech, and consonants, as well as real and nonsense syllables (Blumstein & Cooper, 1974; Kimura, 1961; Shankweiler & Studdert-Kennedy, 1966, 1967; Studdert-Kennedy & Shankweiler, 1970).
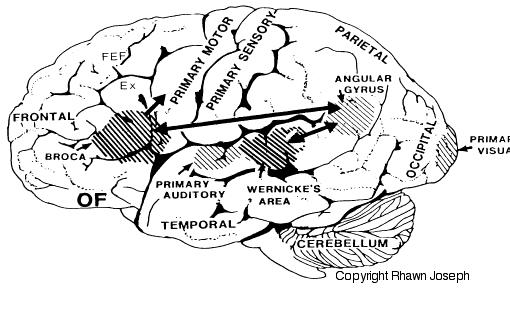
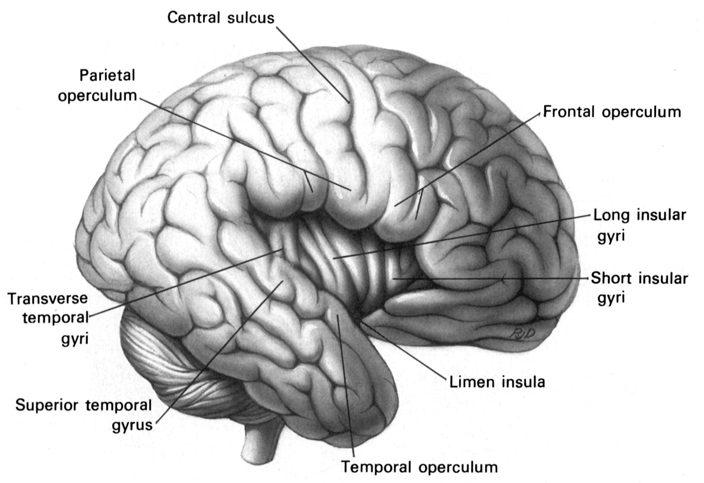
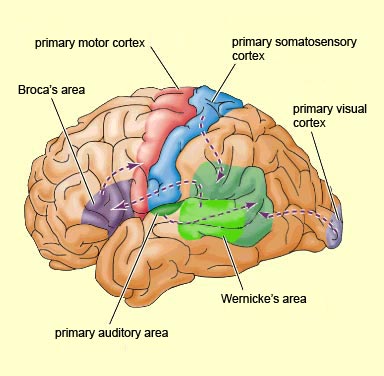
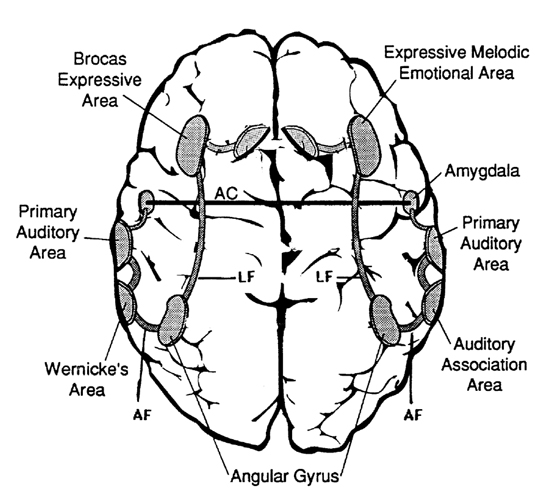
As is generally well known, within the neocortical surface of the left hemisphere there is one area that largely controls the capacity to speak, and another region that mediates the ability to understand speech (Frost, et al., 2009; Goodglass & Kaplan, 2009; Heiss, et al., 2009; Pujol, et al., 2009). Specifically, Broca's expressive speech area is located along the left frontal convexity, whereas Wernicke's receptive speech area is found within the superior temporal lobe and becomes coextensive with the inferior parietal lobule.
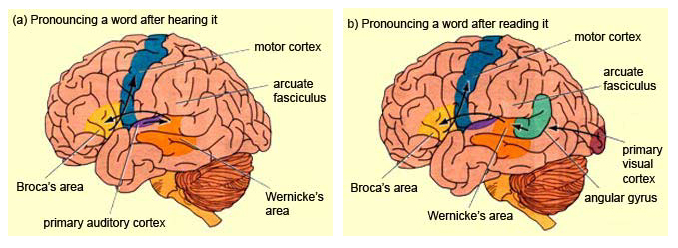
BROCA'S APHASIA
If an individual were to sustain massive damage to the left frontal convexity, his or her ability to speak would be curtailed dramatically. Even if only partially damaged, disturbances that involve grammar and syntax, and reductions in vocabulary and word fluency in both speech and writing result (Benson, 1993; Goodglass & Berko, 1960; Hofstede & Kolk, 2004; Milner, 1964). However, the ability to comprehend language is often (but not completely) intact (Bastiaanse, 1995; Tyler et al. 1995). This disorder is called Broca's (or expressive) aphasia (Benson, 1993; Goodglass & Kaplan, 2009; Levine & Sweet, 2013) and has also been referred to as "motor aphasia."
Aphasia & Depression
Individuals with expressive aphasia, although greatly limited in their ability to speak, nevertheless are capable of making emotional statements or even singing (Gardner, 2005; Goldstein, 1942; Joseph, 2018a; Smith, 1966; Smith & Burklund, 1966; Yamadori et al. 2017). In fact, they may be able to sing words that they cannot say. Moroever, since individuals with Broca's aphasia are able to comprehend, they are aware of their deficit and become appropriately depressed (Robinson & Szetela, 2011). Indeed those with the smallest lesions become the most depressed (Robinson & Benson, 2011) --the depression (as well as the ability to sing) being mediated, presumably, by the intact right cerebral hemisphere (Joseph 2018a) and the right frontal (Kelin et al., 2009) and right temporal lobe (Abrams & Taylor, 1979; Cimino, et al. 2013; Cohen, Penick & Tarter, 1974; Deglin & Nikolaenko, 2005; Shagass et al.,1979; Wexler, 1973).
For example, Abrams and Taylor (1979) and Shagass et al. (1979) have found that depressed patients demonstrated more right vs left temporal lobe electrophysiological activity and EEG abnormalities. Moreover, others have argued that ECT administered to the right vs left temporal lobe is more likely to alleviate depressive symptoms (Cohen, et al, 1974; Deglin & Nikolaenko, 2005) and more likely to result in euphoric reactions. Likewise, it has been found that repetitive transcranial magnetic stimulation of the right frontal lobe over a 10 day period, significantly diminishes depressive feelings as compared to those who received sham treatments (Klien et al., 2009).
Wernicke's Area
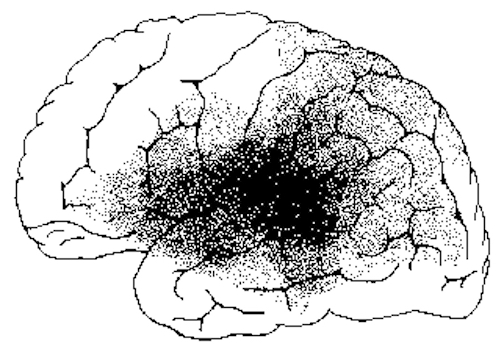
If, instead, the lesion were more posteriorly located along the superior temporal lobe the patient would have great difficulty understanding spoken or written language (Benson, 1993; Goodglass & Kaplan, 2017; Hecaen & Albert, 1978; Kertesz, 2013a; Sarno, 1998). Presumably this disorder is due in part to an impaired capacity to discern the individual units of speech and their temporal order. That is, sounds must be separated into discrete interrelated linear units or they will be perceived as a meaningless blur (Carmon & Nachshon, 1971; Christman, 2004; Efron, 1963; Lackner & Teuber, 1973).
Wernicke's area (in conjunction with the inferior parietal lobule) acts to organize and separate incoming sounds into a temporal and interrelated series so as to extract linguistic meaning via the perception of the resulting sequences (Efron, 1963; Lackner & Teuber, 1973; Lenneberg, 1967). When damaged a spoken sentence such as the "big black dog" might be perceived as "the klabgigdod." This is referred to as Wernicke's aphasia. However, comprehension is improved when the spoken words are separated by long intervals.

Patients with damage to Wernicke's area are nevertheless, still capable of talking (due to preservation of Broca's area and the fiber pathway linking these regions). However, because Wernicke's area also acts to code linguistic stimuli for expression, expressive speech becomes severely abnormal and characterized by nonsequiturs, neologism, paraphasic errors, sound and word order substitutions, and the ommission of pauses and sentence endings (Christman, 2004; Goodglass & Kaplan, 2009; Hecaen & Albert, 1978; Hofstede & Kolk, 2004; Kertesz, 2013a). That is, temporal-sequential expressive linguistic encoding becomes disrupted.
For example, one patient with severe receptive aphasia responded in the following manner: "I am a little suspicious about what the hell is the part there is one part scares, uh estate spares, Ok that has a bunch of drives in it and a bunch of good googin...what the hell...kind of a platz goasted klack..." Presumably since the coding mechanisms involved in organizing what humans are planning to say are the same mechanisms that decode what they hear, expressive as well as receptive speech becomes equally disrupted with left superior temporal lobe damage.
Nevertheless, a peculiarity of this disorder is that these patients do not always realize that what they say is meaningless (Maher et al. 2004). Moreover, they may fail to comprehend that what they hear is meaningless as well (Cf. Lebrun, 1987). This is because when this area is damaged, there is no other region left to analyze the linguistic components of speech and language. The rest of the brain cannot be alerted to the patient's disability. Such patients are at risk for being misdiagnosed as psychotic.
Presumably, as a consequence of loss of comprehension, these patients may display euphoria, or in other cases, paranoia because there remains a nonlinguistic or emotional awareness that something is not right. That is, emotional functioning and comprehension remain intact (though sometimes disrupted due to erroneously processed verbal input). Hence, aphasic individuals are often able to assess to some degree the emotional characteristics of their environment including the prosodic (Monrad-Krohn, 1963), stress contrasts (Blumstein & Goodglass, 2001), and semantic and connotative features of what is said to them, i.e., whether they are being asked a question, given a command, or presented with a declarative sentence (Boller & Green, 2001).
For example, many individuals with severe receptive (Wernicke's) aphasia can understand and respond appropriately to emotional commands and questions (e.g., "Say 'shit'" or "Do you wet your bed?" (Boller et al. 1979; Boller & Green, 2001). Similarly, the ability to read and write emotional words (as compared to non-emotional or abstract words) is also somewhat preserved among aphasics (Landis et al. 1982) due to preservation of the right hemisphere. Indeed, the capacity to identify emotional words and sentences is a capacity at which the right hemisphere excells (Borod et al. 1992; Graves et al. 2011; Van Strien & Morpurgo, 1992).
Because these paralingusitic and emotional features of language are analyzed by the intact right cerebral hemisphere, the aphasic individual is able to grasp in general the meaning or intent of a speaker, although verbal comprehension is reduced. This, in turn, enables them to react in a somewhat appropriate fashion when spoken to.
For example, after I had diagnosed a patient as suffering from Wernicke's aphasia, her nurse disagreed and indicated the patient responded correctly to questions such as, "How are you this morning?" That is, the patient replied: "Fine." Later, when I re-examined the patient I used a tone of voice appropriate for "How are you today?", but instead said; "It's raining outside?" The patient replied, "Fine!" and appropriately smiled and nodded her head (Joseph, 2018a). Often our pets are able to determine what we mean and how we feel by analyzing similar melodic-emotional nuances.
Although language is often discussed in terms of grammar and vocabulary, there is a third major aspect to linguistic expression and comprehension by which a speaker may convey and a listener discern intent, attitude, feeling, mood, context, and meaning. Language is both emotional and grammatically descriptive. A listener comprehends not only the content and grammar of what is said, but the emotion and melody of how it is said -what a speaker feels.
Feeling, be it anger, happiness, sadness, sarcasm, empathy, etc., often is communicated by varying the rate, amplitude, pitch, inflection, timbre, melody and stress contours of the voice. When devoid of intonational contours, language becomes monotone and bland and a listener experiences difficulty discerning attitude, context, intent, and feeling. Conditions such as these arise after damage to select areas of the right hemisphere or when the entire right half of the brain is anesthetized (e.g., during sodium amytal procedures).
It is now well established (based on studies of normal and brain-damaged subjects) that the right hemisphere is superior to the left in distinguishing, interpreting, and processing vocal inflectional nuances, including intensity, stress and melodic pitch contours, timbre, cadence, emotional tone, frequency, amplitude, melody, duration, and intonation (Blumstein & Cooper, 1974; Bowers et al. 1987; Carmon & Nachshon, 1973; Heilman et al. 2005; Ley & Bryden, 1979; Mahoney & Sainsbury, 1987; Ross, 2011; Safer & Leventhal, 2017; Samson & Zatorre, 2018, 1992; Shapiro & Danly, 1985; Tucker et al. 2017). The right hemisphere, therefore, is fully capable of determining and deducing not only what a persons feels about what he or she is saying, but why and in what context he is saying it --even in the absence of vocabulary and other denotative linguistic features (Blumstein & Cooper, 1974; DeUrso et al. 1986; Dwyer & Rinn, 2011). This occurs through the analysis of tone and melody.
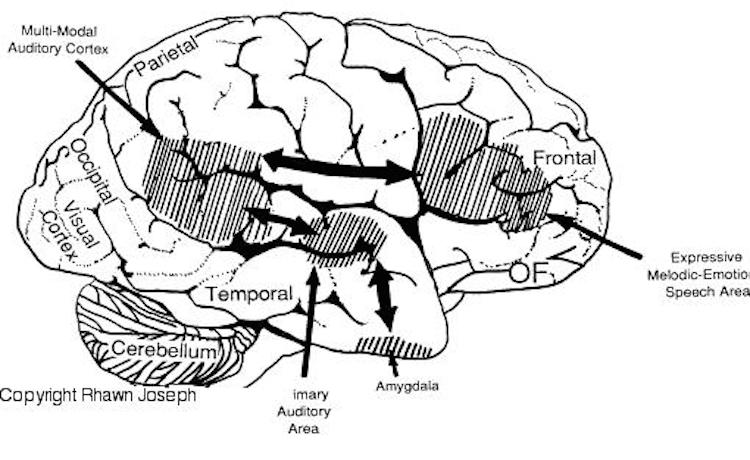
Hence, if I were to say, "Do you want to go outside?" although both hemispheres are able to determine whether a question vs. a statements has been made (Heilman et al. 2014; Weintraub et al. 2011), it is the right cerebrum which analyzes the paralinguistic emotional features of the voice so as to determine whether "going outside" will be fun or whether I am going to punch you in the nose. In fact, even without the aid of the actual words, based merely on melody and tone the right cerebrum can determine context and the feelings of the speaker (Blumstein & Cooper, 1974; DeUrso et al. 1986; Dwyer & Rinn, 2011). This may well explain why even preverbal infants are able to make these same determinations even when spoken to in a foreign language (Fernald, 1993; Haviland & Lelwica, 1987). The left hemisphere has great difficulty with such tasks.
For example, in experiments in which verbal information was filtered and the individual was to determine the context in which a person was speaking (e.g. talking about the death of a friend, speaking to a lost child), the right hemisphere was found to be dominant (Dwyer & Rinn, 2011). It is for these and other reasons that the right half of the brain sometimes is thought to be the more intuitive half of the cerebrum.
Correspondingly when the right hemisphere is damaged, the ability to process, recall, or even recognize these nonverbal nuances is greatly attenuated. For example, although able to comprehend individual sentences and paragraphs, such patients have difficulty understanding context and emotional connotation, drawing inferences, relating what is heard to its proper context, determining the overall gist or theme, and recognizing discrepancies such that they are likely to miss the point, respond to inappropriate details, and fail to appreciate fully when they are being presented with information that is sarcastic, incongruent or even implausible (Beeman 1993; Brownell et al. 1986; Foldi et al. 2013; Gardner et al. 2013; Kaplan et al. 2017; Rehak et al. 1992; Wapner et al. 2011).


Such patients frequently tend to be very concrete and literal. For example, when presented with the statement, "He had a heavy heart" and requested to choose several interpretations, right-brain damaged (vs. aphasic) patients are more likely to choose a picture of an individual staggering under a large heart vs. a crying person. They also have difficulty describing morals, motives, emotions, or overall main points (e.g. they lose the gestalt), although the ability to recall isolated facts and details is preserved (Delis et al. 1986; Hough 2017; Wapner et al. 2011) -details being the province of the left hemisphere.
Although they are not aphasic, individuals with right hemisphere damage sometimes have difficulty comprehending complex verbal and written statements, particularly when there are features which involve spatial transformations or incongruencies. For example, when presented with the question "Bob is taller than George. Who is shorter? ", those with right-brain damage have difficulties due, presumably, to a deficit in nonlingusitic imaginal processing or an inability to search a spatial representation of what they hear (Carmazza et al. 1976).
In contrast, when presented with "Bob is taller than George. Who is taller?" patients with right-hemisphere damage perform similar to normals, which indicates that the left cerebrum is responsible for providing the solution (Carmazza et al. 1976) given that the right hemisphere is injured and the question does not require any type of spatial transformation. That is, because the question "Who is shorter?" does not necessarily follow the first part of the statements (i.e., incongruent), whereas "Who is taller?" does, these differential findings further suggest that the right hemisphere is more involved than the left in the analysis of incongruencies.
Moreover, it appears that during the early stages of neonatal and infant development, that the role of the right hemisphere in language expression and perception was even more pronounced. As originally proposed by Joseph (1982, 2018a), language in the neonate and infant is dominated by the right hemisphere, which in turn accounts for the initial prosodic, melodic, and emotional qualities of their vocalizations. Of course, the left hemisphere is genetically programmed to gain functional dominance and to acquire the grammatical, temporal sequential, word-rich, and expressive-motor aspects of speech--as is evident neuronatomically by the presence of asymmetries in the fetal and neontal planum temporal (Wada et al., 2005; Witelson & Palli, 1973), and the fact that the left cerebral pyramidal tract descends and establishes synaptic contact with the brainstem and spinal cord in advance of the right (Kertesz & Geschwind 1971; Yakovlev & Rakic 1966).
However, as also based on evoked potential studies, the pattern of neurological activity, during the performance of language tasks, does not begin to resemble the adult pattern until the onset of puberty (Hollcomb et al., 1992). Moreover, although the left hemisphere gradually acquires language, the right hemisphere continues to participate even in non-emotional language processing, including reading, as demonstrated by functional imaging studies (Bottini et al., 2004; Cuenod, et al., 1995; Price et al., 1996).
For example, the right temporal and parietal areas are activated when reading (Bottini et al., 2004; Price et al., 1996), and the right temporal lobe becomes highly active when engaged in interpreting the figurative aspects of language (Bottini et al., 2004). Moreover, bilateral frontal activation is seen when speaking--though this activity is greater on the left (Passingham, 2013; Peterson et al., 2018). In part, however, these latter findings may well reflect those aspects of right hemisphere language processing (temporal-parietal) and expression (frontal-parietal) which are concerned with extracting and vocalizing emotional, motivational, personal, and contextual details.

For example, right frontal damage has been associated with a loss of emotional speech and emotional gesturing and a significantly reduced ability to mimic various nonlinguistic vocal patterns (Joseph 2018a; Ross, 2011, 1993; Shapiro & Danly, 1985). In these instances, speech can becomes flat and monotone or characterized by inflectional distortions.
With lesions that involve the right temporal-parietal area, the ability to comprehend or produce appropriate verbal prosody, emotional speech, or to repeat emotional statements is reduced significantly (Gorelick & Ross, 1987; Heilman et al. 2005; Lalande et al. 1992; Ross, 2011; Starkstein et al. 2004; Tucker et al. 2017). Indeed, when presented with neutral sentences spoken in an emotional manner, right hemisphere damage disrupts perception and discrimination (Heilman et al. 2005; Lalande et al. 1992) and the comprehension of emotional prosody (Heilman et al. 2014; Starkstein et al. 2004) regardless of whether it is positive or negative in content. Moreover, the ability to differentiate between different and even oppositional emotional qualities (e.g., "sarcasm vs irony" or "love" vs "hate") can become distorted (Cicone et al. 1980; Kaplan et al. 2017), and the capacity to appreciate and comprehend humor or mirth may be attenuated (Gardner et al. 2005).
The semantic-contextual ability of the right hemisphere is not limited to prosodic and paralinguistic features, however, but includes the ability to process and recognize familiar, concrete, highly imaginable words (J. Day, 2017; Deloch et al. 1987; Ellis & Shephard, 2005; Hines, 1976; Joseph 2018b; Landis et al., 1982; Mannhaupt, 2013), as well as emotional language in general.
The disconnected right hemisphere also can read printed words (Gazzaniga, 1970; Joseph, 1986b, 2018b; Levy, 2013; Sperry, 1982; Zaidel, 2013), retrieve objects with the left hand in response to direct and indirect verbal commands, e.g. "a container for liquids" (Joseph, 2018b; Sperry, 1982), and spell simple three- and four-letter words with cut-out letters (Sperry, 1982). However, it cannot comprehend complex, non-emotional, written or spoken language.
As noted, the right hemisphere dominance for vocal (and non-verbal) emotional expression and comprehension is believed to be secondary to hierarchical neocortical representation of limbic system functions. It may well be dominance by default, however. That is, at one time both hemispheres may well have contributed more or less equally to emotional expression, but with the evolution of language and right handedness, the left hemisphere gradually lost this capacity where it was retained in the right cerebrum (chapter 6). Even so, without the participation of the limbic system, the amygdala and cingulate gyrus in particular, emotional language capabilities would for the most part be nonexistent.
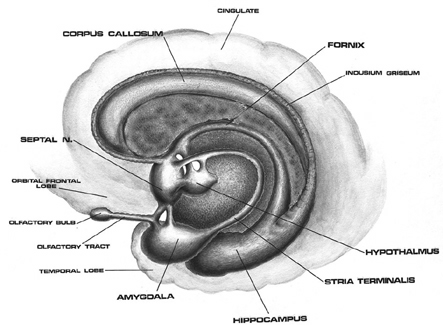

Conversely, subcortical right cerebral lesions involving the anterior cingulate, the amygdala, or these fiber interconnections can also result in emotional expressive and receptive disorders (chapter 15). Similarly, lesions to the left temporal or frontal lobe may result in disconnection which in turn may lead to distortions in the vocal expression or perception of emotional nuances -that is, within the left hemisphere.
SEX DIFFERENCES IN EMOTIONAL SOUND PRODUCTION & PERCEPTION
As detailed in chapter 5 and 15, the evolution of the anterior cingulate gyrus corresponded with the onset of long term maternal care, and presumably the advent of mother-infant vocalization and intercommunication. The presence of an adult female, in fact, appears to promote language production in infants, as well as adult males and other females. Thus non-threatening, complex social-emotional vocalizations are to some degree strongly associated with female-maternal behavior and the desire to form social-emotional attachments (chapter 15).
Across most social-living mammalian species, females tend to produce a greater range of social-emotional (limbic) vocalization (Joseph, 1993, 2009e). For example, human females tend to employ 5-6 different prosodic variations and to utilize the higher registers when conversing. They are also more likely to employ glissando or sliding effects between stressed syllables, and they tend to talk faster as well (Brend, 2005; Coleman, 1971; Edelsky, 1979). Men tend to be more monotone, employing 2-3 variations on average, most of which hovers around the lower registers (Brend, 2005; Coleman, 1971; Edelsky, 1979). Even when trying to emphasize a point males are less likely to employ melodic extremes but instead tend to speak louder. This is not, however, a function of sex differences in the oral-laryngeal structures, but are due to the greater capacity of the female right hemisphere (and limbic system) to express and perceive these nuance, including for example, the sounds of harmony (Evers et al., 2009)
For example, it has been repeatedly demonstrated that females are more emotionally expressive, and are more perceptive in regard to comprehending emotional verbal nuances (Burton & Levy, 2017; Hall, 1978; Soloman & Ali, 2001). This superior sensitivity includes the ability to feel and verbally express empathy (Burton & Levy, 2017; Safer, 2011) and the comprehension of emotional faces (Buck, Miller & Caul, 1974; Buck, Savin, Miller & Caul, 2001; see also Evans et al. 1995). In fact, from childhood to adulthood women appear to be much more emotionally expressive than males in general (Gilbert, 1969; see Brody, 1985; Burton & Levy, 2017 for review).
As detailed in chapters 6 and 7, in addition to the evolution of the "maternal" anterior cingulate gyrus, this language superiority appears to be in part a consequence of the differential activities engaged in by men (hunting) vs females (gathering, food preparation) for much of human history, and the possibility that the female right hemisphere has more neocortical space committed to emotional perception and expression (chapter 7).
As noted, this superiority is probably also a consequence of limbic system sexual differentiation, and the role of the female limbic system in promoting maternal care and communication (see chapter 15). Thus, regardless of culture, human mothers tend to emphasize and even exaggerate social-emotional, and melodic-prosodic vocal features when interacting with their infants (Fernald, 1992; Fernald et al. 2017), which in turn appears to greatly influence infant emotional behavior and attention (Fernald, 2013).


"I did not have sex with that woman..." -President Bill Clinton before his impeachment for lying under oath.
"I am not a crook" --President Richard Nixon before resigning from the Presidency.
It is not uncommon for individuals to lie. However, sometimes they believe their own lies, and this can be the basis for self-deception. In the extreme, however, some individuals following cerebral injury, make up lies that are so bizarre it takes on the form of confabulation.
In contrast to left frontal convexity lesions which can result in speech arrest (Broca's expressive aphasia) and/or significant reductions in verbal fluency, right frontal damage sometimes has been observed to result in speech release, excessive verbosity, tangentiality, and in the extreme, confabulation (Fischer et al. 1995; Joseph, 1986a, 2018a, 2009a).
When secondary to frontal damage, conflabulation seems to be due to disinhibition, difficulty monitoring responses, withholding answers, utilizing external or internal cues to make corrections, or suppressing the flow of tangential and circumstantial ideas (Shapiro et al. 2011; Stuss et al. 1978). When this occurs, the language axis of the left hemisphere becomes overwhelmed and flooded by irrelevant associations (Joseph, 1986a, 2018a, 2009a). In some cases the content of the confabulation may border on the bizarre and fantastical as loosely associated ideas become organized and anchored around fragments of current experience.
MEMORY AND CONFABULATION
As detailed in chapters 10, 19, confablation is associated with right frontal lesions in part because of flooding of the speech areas with irrelevant associations. However, yet another factor is loss of memory--or rather, an inability to retrieval autobiographical and episodic details; even those stored using language. Indeed, the right fontal lobe is directly implicated in episodic and autobiographical memory retrieval--as also recently demonstrated using functional imaging (see chapter 19).
Specifically, episodic memory memories are perceptual and are stored in an autobiographical context, and when engaged in episodic retrieval, there is a significant activation of the right frontal lobe, (Brewer et al., 1998; Dolan et al., 2013; Tulving et al., 2004; Kapur et al., 1995), right thalamus, and right medial temporal lobe (Dolan et al., 2013), even when the tasks requiring verbal processing. For example, in a test of unconscious memory, subjects were presented with word stems of complete words were previously presented there was increased blood flow in the right hippocampus and right frontal lobe (Squire, et al., 1992). Right frontal activation was also seen in a recognition tasks involving sentences viewed the day before (Tulving et al., 2004). Presumably, activity increases in the right frontal lobe as a function of retrieval effort (Kapur et al., 1995), whereas injury to the right frontal lobe results in retrieval failure and thus a gap in the information and memories access, coupled with disinhibition and a flooding of the language axis with irrelevant associations.
GAP-FILLING
Confabulation also can result from lesions that involve the posterior portions of the right hemisphere, immaturity or surgical section of the corpus callosum, or destruction of fiber tracts that lead to the left hemisphere (Joseph, 1982, 1986ab, 2018ab; Joseph et al. 2014). This results in incomplete information transfer and reception within discrete brain regions, so that one area of the brain and mind are disconnected from another.
As a consequence, because the language axis of the left hemisphere is unable to gain access to needed information, it attempts to fill the gap with information that is related in some manner to the fragments received. However, because the language areas are disconnected from the source of needed information, it cannot be informed that what it is saying (or, rather, making up) is erroneous, at least insofar as the damaged modality is concerned.
For example, in cases presented by Redlich and Dorsey (1945), individuals who were suffering from blindness or gross visual disturbances due to injuries in the visual cortex continued to claim that they could see even when they bumped into objects and tripped over furniture. Apparently, they maintained these claims because the areas of the brain that normally would alert them to their blindness (i.e. visual cortex) were no longer functioning.
Confabulation and delusional denial also often accompany neglect and body-image disturbances secondary to right cerebral (parietal) damage (Joseph 1982, 1986a, 2018a). For example, the left hemisphere may claim that a paralyzed left leg or arm is normal or that it belongs to someone other than the patient. This occurs in many cases because somesthetic body information no longer is being processed or transferred by the damaged right hemisphere; the body image and the memory of the left half of the body have been deleted. In all these instances, however, although the damage may be in the right hemisphere, it is the speaking half of the brain that confabulates.



Similarly, there have been reports that some musicians and composers who were suffering from aphasia and/or significant left hemisphere impairment were able to continue their work (Alajounine, 1948; Critchley, 1953; Luria, 1973). In some cases, despite severe receptive aphasia and/or although the ability to read written language (alexia) was disrupted, the ability to read music or to continue composing was preserved (Gates & Bradshaw, 2017; Luria, 1973). One famous example is that of Maurice Ravel, who suffered an injury to the left half of his brain in an auto accident. This resulted in ideomotor apraxia, dysgraphia, and moderate disturbances in comprehending speech (i.e., Wernicke's Aphasia). Nevertheless, he had no difficulty recognizing various musical compositions, was able to detect even minor errors when compositions were played, and was able to correct those errors by playing them correctly on the piano (Alajounine, 1948).

Right hemisphere damage also can disrupt the ability to sing or carry a tune and can cause toneless, monotonous speech, as well as abolish the capacity to obtain pleasure while listening to music (Reese. 1948; Ross, 2011; Shapiro & Danly, 1985), i.e., a condition also referred to as amusia. For example, Freeman and Williams (1953) report that removal of the right amygdala in one patient resulted in a great change in the pitch and timbre of speech and that the ability to sing also was severely affected. Similarly, when the right hemisphere is anesthetized the melodic aspects of speech and singing become significantly impaired (Gordon & Bogen, 1974).
It also has been demonstrated consistently in normals (such as in dictotic listening studies) and with brain-injured individuals that the right hemisphere predominates in the perception (and/or expression) of timbre, chords, tone, pitch, loudness, melody, meter, tempo, and intensity (Breitling et al.1987; Curry, 1967; R. Day et al.1971, Gates & Bradshaw, 2017; Gordon, 1970; Gordon & Bogen, 1974; Kester et al. 2013; Kimura, 1964; Knox & Kimura, 1970; McFarland & Fortin, 1982; Milner, 1962; Molfese et al. 2005; Piazza, 1980; Reese, 1948; Segalowitz & Plantery, 1985; Spellacy, 1970; Swisher et al. 1969; Tsunoda, 2005; Zurif, 1974)--the major components (in conjuction with harmony) of a musical stimulus.
For example, in a functional imaging study, it was found that when professional pianists played Bach (Bach's Italian concerto, third movement) that there was increased activity in the right but not left temporal lobe, whereas when they played scales, activity increased in the left but not right temporal lobe (Parsons & Fox, 2013). Likewise, Evers and colleagues (2009) in evaluating cerebral blood velocity, found that a right hemisphere increase in blood flow when listening to harmony (but not rhythm), among non-musicians in general, and especially among females.
However, this does not appear to be the case with professional musicians who in some respects tend to treat music as a mathematical language that is subject to rhythmic analysis. As noted when professional pianists played scaled, activity increased in the left but not right temporal lobe (Parsons & Fox 2013). Moroever, Evers and colleagues (2009) found that musicians displayed an increase in left hemisphere blood flow when listening to both harmony and rhythm.
ENVIRONMENTAL AND HUMAN/ANIMAL SOUNDS
In addition to music the right hemisphere has been shown to be superior to the left in discerning and recognizing nonverbal, animal, and environmental sounds (Curry, 1967; Joseph, 2018b; Kimura, 1963; King & Kimura, 2001; Knox & Kimura, 1970; Nielsen, 1946; Piazza, 1980; Roland et al. 2011; Schnider et al. 2004; Spreen et al. 1965; Tsunoda, 2005). Similarly, damage that involves select areas within the right hemisphere not only disturb the capacity to discern musical and social-emotional vocal nuances, but may disrupt the ability to perceive, recognize, or disciminate between a diverse number of sounds which occur naturally within the environment (Fujii et al., 2017; Joseph, 1993; Nielsen, 1946; Schnider et al. 2004; Spreen et al. 1965), such as water splashing, a door banging, applause, or even a typewriter; this is a condition which also plagues the disconnected left hemisphere (Joseph, 2018b).
A 47-year old woman I examined who was subsequently found to have a calcium cyst growing from the skull into the right superior temporal lobe, was able to name pictures of animals, tools and household objects. However, she was almost completely unable to recognize and correctly name animal and humans sounds (e.g. a baby crying, a crowd cheering, a lion roaring) which had been briefly presented, but was better able to recognize non-living sounds such as a creaking door, or a hammer hammering--though these abilities were also compromised. However, coupled with other findings to be reviewed below, there is some possiblility that the right temporal lobe is better able to recognize living and true environmental sounds, whereas the left may be better able to recognize and name non-living sounds.


For example, it is somewhat probable that primitive man and woman's first exposure to the sounds of music was environmentally embedded, for obviously musical sounds are heard frequently throughout nature (e.g. birds singing, the whistling of the wind, the humming of bees or insects). For example, bird songs can encompass sounds that are "flute-like, truly chime- or bell-like, violin-or guitar-like" and "some are almost as tender as a boy soprano" (Hartshorne, 1973, p. 36).

MUSIC AND EMOTION
Music is related strongly to emotion and, in fact, may not only be "pleasing to the ear," but invested with emotional significance. For example, when played in a major key, music sounds happy or joyful. When played in a minor key, music often is perceived as sad or melancholic. We are all familiar with the "blues" and perhaps at one time or another have felt like "singing for joy," or have told someone, "You make my heart sing!"
Interestingly, it has been reported that music can act to accelerate pulse rate (Reese, 1948), raise or lower blood pressure, and, thus, alter the rhythm of the heart's beat. Rhythm, of course, is a major component of music.

Left Hemisphere Musical Contributions.
There is some evidence to indicate that certain aspects of pitch, time sense and rhythm, are mediated to a similar degree by both cerebral hemispheres (Milner, 1962), with rhythm being associated with increased left cerebral activity (Evers et al., 2009). Of course, time sense and rhythm are also highly important in speech perception. These findings support the possibliity of a left hemisphere contribution to music. In fact, some authors have argued that receptive amusia is due to left hemisphere damage and that expressive amusia is due to right hemisphere dysfunction (Wertheim, 1969).
It also has been been reported that some musicians tend to show a left hemisphere dominance in the perception of certain aspects of music (Gates & Bradshaw, 2017), particularly rhythm (Evers et al., 2009). Indeed, when the sequential and rhythmical aspects of music are emphasized, the left hemisphere becomes increasingly involved (Breitling et al. 1987; Halperin et al. 1973). In this regard, it seems that when music is treated as a type of language to be acquired or when its mathematical and temporal-sequential features are emphasized (Breitling et al., 1987), the left cerebral hemisphere becomes heavily involved in its production and perception; especially in professional musicians (Evers et al., 2009).

MUSIC, MATH AND GEOMETRIC SPACE
Pythagoras, the great Greek mathematician, argued almost 2,000 years ago that music was numerical, the expression of number in sound (Durant 1939; McClain 1978).


This same golden rectangle is found in nature, i.e., the chambered nautilus shell, the shell of a snail, and in the ear --the cochleus. The geometric proportions of the golden rectangle were also employed in designing the Parthenon in Athens, and by Ptolemy in developing the "tonal calendar" and the "tonal Zodiac" (McCain, 1978) -the scale of ratios "bent round in a circle."
In fact, the first cosmologies, such as those developed by the ancient Egyptians, Hindus, Babylonians, and Greeks, were based on musical ratios (Durant, 1939; McClain, 1978). Pythagorus and Plato applied these same "musical proportions" to their theory of numbers, planetary motion, and to the science of stereometry --the gauging of solids (McClain, 1978). Indeed, Pythagorus attempted to deduce the size, speed, distance, and orbit of the planets based on musical ratios as well as estimates of the sounds generated (e.g., pitch and harmony) by their movement through space, i.e. "the music of the spheres" (Durant, 1939).
Interestingly, the famous mathematician and physicist Johannes Kepler in describing his laws of planetary motion also referred to them as based on the "music of the spheres." Thus music seems to have certain geometric properties, such as are expressed via the ratio. Indeed, Pythagrous, the "father" of arithmetic, geometry, and trigonometry, believed music to be geometric.
"Words and language,whether written or spoken,do not seem to lay any part in my thought processes. The psychological entities that serve as building blocks for my thought are certain signs or images, more or less clear, that I can reproduce at will." A. Einstein
As we know, geometry is employed in the measurement of land, the demarcation of boundaries, and, thus, in the analysis of space, shape, points, lines, angles, surfaces, and configuration.
In nature, one form of musical expression, that is, the songs of most birds, also is produced for geometric purposes. That is, a bird does not "sing for joy," but to signal others of impending threat, to attract mates, to indicate direction and location, to stake out territory, and to warn away others who may attempt to intrude on his space (Catchpole, 1979; Hartshorne, 1973).




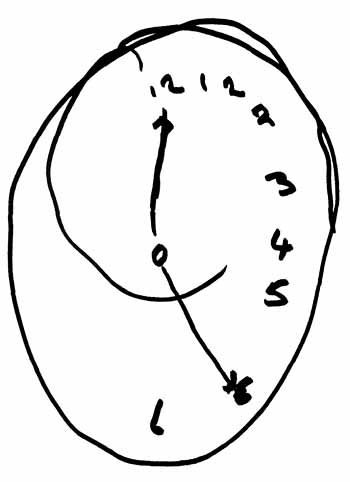
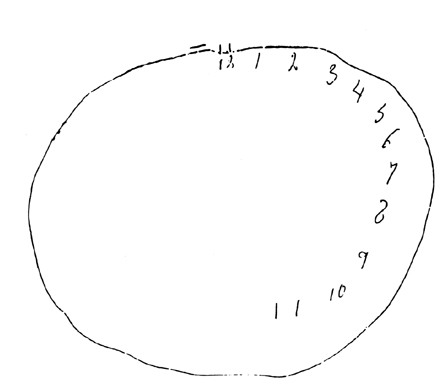
The right hemisphere perceives the left half of space. When damaged, patients neglect the left half of space. In this example, patients were instructed to "draw the face of a clock, put all the numbers in, and make it say 10 after 11."
For example, Kimura (1963) found that patients with right vs left temporal lobe injury were impaired when presented with overlapping nonsense shapes and then immediately tested for recognition. Likewise, Meier and French (1965), found that those with right vs left temporal lobe injuries were impaired when asked to make visual discriminations when presented with fragmented concentric circle patterns--skills which are also related to visual closure and gestalt formation.
In addition, the isolated right hemisphere has been found to be superior in "fitting designs into larger matrices, judging whole circle size from a small arc, discriminating and recalling nondescript shapes, making mental spatial transformations, sorting block sizes and shapes into categories, perceiving wholes from a collection of parts, and the intuitive perception and apprehension of geometric principles" (Sperry, 1982, p.1225).


Thus, it is the right hemisphere which enables us to find our way in space without getting lost, to walk and run without tripping and falling, to throw and catch a football with accuracy, to drive a car without bumping into things, to draw conclusions based on partial information, and to see the forest when looking at the trees. The right is also superior to the left in analyzing manipulo-spatial problems, drawing and copying simple and complex geometric-like figures and performing constructional tasks, block designs and puzzles (Benson & Barton, 1970; Black & Strub 1976; Critchley, 1953; DeRenzi, 1982; Gardner, 2005; Hecaen & Albert, 1978; Hier et al. 2013; Kertesz, 2013b; Levy, 1974; Luria, 1973, 1980; Piercy et al. 1960). It is for these and other reasons that the right brain is often viewed as the artistic half of the cerebrum.
The right hemisphere is also dominant over the left in regard to localizing and thus referencing the position of an object in space (Cook et al. 2004; Nunn et al., 2009; Ploner et al., 2009), as well as in aiming and closed loop throwing accuracy (Guiard et al. 2013; Haaland & Harrington, 2017). Of course, most individuals use the right hand to throw (as well as draw). Presumably the right hemisphere is able to guide right limb and related axial movements (Rapcsak et al. 1993) via bilateral SMA and parietal lobe innervation of the basal ganglia and lower motor neurons (see chapters 16, 19).


SEX DIFFERENCES IN SPATIAL ABILITY
Right cerebral visual-spatial and geometric
superiorities constitute skills that would enable an ancient hunter to visually track, throw a spear and dispatch various prey while maintaining a keen awareness of all else occurring within the environment. For most of human (and probably chimpanzee and baboon) history, males have typically been the hunters, whereas females (including female chimps) spend much more time gathering.
Moreover, some of these differences are present during childhood and have been demonstrated in other species (Dawson et al. 1978; Harris, 1978; Levy & Heller, 1992; Joseph, 1979; Joseph and Gallagher, 1982; Joseph et al. 1978). For example, male rats consistently demonstrate superior visual-spatial skills as compared to females (Joseph, 1979, Joseph & Gallagher, 1982; Joseph et al. 1978). In fact, these sex difference are ameliorated only when females are reared in a complex, socially and environmentally enriched environment and when males are placed in a restricted and impoverished environment; in which case males and females perform similarly (Joseph & Gallagher 1982). When environments are similar (enriched or deprived) males outperform females. Hence, environmental influences do not cause these sex differences which are obviously innate.
In large part, these sex differences in spatial ability are clearly a function of the presence or absence of testosterone during human fetal development (Joseph et al. 1978), as well as the differential activities of males vs females for much of human history; i.e. hunting vs gathering. Hence, over the course of evolution, natural males vs female superiorities have been enhanced.
However, in this regard, just as females appear to have more brain space devoted to language functions, it also appears that males have more neocortical space devoted to spatial-perceptual and related expressive functions (Joseph, 1993).
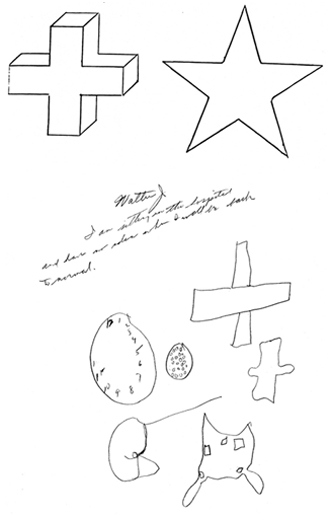
Patient with right cerebral injury was told to copy the star and the cross
Such individuals may misplace things, have difficulty with balance and stumble and bump into walls and furniture, become easily lost, confused and disoriented while they are walking or driving; and have difficulty following directions or even putting on their clothes. Indeed such patients easily can get lost while they are walking down familiar streets and even in their own homes (Benton, 1993; DeRenzi, 1982; DeRenzi et al., 1969; Ettlinger, 1960; Gardner, 2005; Landis et al., 1986; Lansdell, 1968a; 1970; Levy, 1974). Right brain damage also can also result in disorientation, problems in assuming different perspectives, and even with dressing (Hecaen, 1962; Hier et al., 2013).
In some instances, the deficit can be quite subtle and circumscribed. For example, one patients only complaint (3 months after he suffered a circumscribed blunt head injury that resulted in a subdural hemotoma over the right posterior temporal-parietal area) was that his golf game had deterioarted significantly and he was no longer as accurate when throwing wads of paper into the trash can in his office. Formal testing also indicated mild constructional and manipulo-spatial disturbances, with most other capacities in the high average to superior range.
In contrast, the right cerebrum is more involved in the overall perceptual analysis of visual and object interrelations including visual closure and gestalt formation. Thus, patients with right cerebral injuries have trouble with general shape and organization, although certain details may be drawn correctly. Drawings may also be grossly distorted and/or characterized by left sided neglect.
Right sided damage also can affect writing. When patients are asked to write cursively, writing samples may display problems with visual closure, as well as excessive segmentation due to left hemisphere release (Joseph, 2018a). That is, cursively the word "recognition" may be written "re cog n i tion," or letters such as "o" may be only partly formed.
Constructional deficits are more severe after right hemisphere damage (Arrigoni & DeRenzi, 1964; Black & Strub, 1976; Benson & Barton, 1970; Bradshaw & Mattingly, 1995; Critchley, 1953; Hier et al., 2013; Piercy et al., 1960). However, lesions to either hemisphere can create disturbances in constructional and manipulo-spatial functioning -including performance on the WAIS Block Design and Object Assembly subtests (Arrigoni & DeRenzi, 1964; Cubelli et al. 2013; Kimura, 1993; Mehta et al., 1987; Piercy et al., 1960).
If the left hemisphere is damaged, performance may be impaired due to motor programming errors and/or an inability to transform the percept into a motor action with preservation of good spatial-perceptual functioning (Warrington et al., 1966); in which case errors may be recognized by the patient. With right cerebral injuries, visual-spatial perceptual functioning becomes distorted (although motor activities per se are preserved) and the patient may not realize they have made an error (Hecaen & Assal, 1970).
Thus, visual motor deficits can result from lesions in either hemisphere (Arrigoni & DeRenzi, 1964; Bartolomeo et al. 2004; Kimura, 1993; Piercy et al., 1960), though visual-perceptual disturbances are more likely to result from right hemisphere damage. Lesions to the left half of the brain may leave the perceptual aspects undisturbed whereas visual motor functioning and selective organization may be compromised (Kim et al. 2014; Mehta et al., 1987; Poeck et al. 1973; Teuber & Weinstein, 1956) and attentional functioning may be disturbed (Bartolomeo et al. 2004; Cubelli et al. 2013).
In general, the size and sometimes the location of the lesion within the right hemisphere have little or no correlation with the extent of the visual-spatial or constructional deficits demonstrated (Kimura, 1993), although right posterior lesions tend to be worst of all. For example, in a restrospective study of 656 patients with unilateral lesions that employed a short form of the WAIS, Warrington, James and Maciejewski (1986), found that those with right posterior (vs. anterior or left hemisphere) damage had the lowest Performance IQs as well as difficulty performing the block design and picture arrangment subtests.
Conversely, visual-perceptual disturbances associated with left hemisphere damage are positively correlated with lesion size; and left anterior lesions are worse than left posterior (Benson & Barton, 1970; Black & Bernard, 2014; Black & Strub, 1976; Lansdell, 1970). The larger the lesion, the more extensive the deficit. Kimura (1993), however, argues that males are more likely to demonstrate these disorders with left posterior lesions, whereas in females the lesion tends to be more anterior.
As pertaining to WAIS PIQ-VIQ differences, based on an extensive review of the literature, Bornstein and Matarazzo (1982) have have confirmed what is now generally well known; i.e. that those with left hemisphere lesions have lower VIQs, whereas those with right sided damage have lower mean PIQs. Reading and Math.
Because of its importance in visual orientation, the right hemisphere also participates in math and reading. Visual-spatial orientation is important when performing a variety of math problems, such as in correctly aligning numbers when adding multiple digits. Conversely, right sided lesions may cause the patient to neglect the left half of digit pairs while adding or subtracting (Hecaen & Albert, 1978; Luria, 1980).
Moreover, some aspects of math, such as what Dehaene and colleagues (2009. p. 970), refer to as "approximate" arithmetic, is language independent and "relies on a sense of numerical magnitudes, and recruits bilateral areas of the parietal lobes involved in visuo-spatial processing." Hence, right parietal lesions can disrupt this more intuitive aspect of mathematical reasoning.
Moreover, via the analysis of position, orientation, etc., the right cerebrum enables a human to read the words on this page without losing their place and jumping half-hazardly from line to line. Conversely, when damaged, patients may fail to attend to the left half of written words, or even the left half of the page (Critchely, 1953; Gainotti, et al. 2001, 1986).
Because the right half of the cerebrum can make conclusions based on partial information, e.g. closure and gestalt formation, humans need not read every or all of each word in order to know what they have read. For example, when presented with incomplete words or perceptually degraded written stimuli, there is an initial right hemisphere superiority in processing (Hellige & Webster, 1979); i.e. visual closure, which enables an individual to fill-in these gaps and thus comprehend. Of course, sometimes people draw the wrong conclusions from incomplete perception (e.g. reading "word power" as "world power"), a problem that can become exaggerated if the right half of the brain has been damaged.
In addition, when the visual-figural characteristics of written language are emphasized, such as when large gothic scriptis employed (e.g., in Tachistoscopic studies) the right hemisphere is dominant (Bryden & Allard, 1976; Wagner & Harris, 2004). Similarly, when presented with unfamiliar written words, or a foreign alphabet, there is an initial right hemisphere perceptual dominance (Silverberg et al. 1979), apparently as the left hemisphere does not immediately recognize these stimuli as "words," whereas the right hemisphere attends to their shape and form.
Indeed, based on an extensive analysis of the evolution of written language , there is some evidence to suggestan initial right hemisphere dominance, particularly in that much of what was written was at first depicted in a pictorial, gestalt-like fashion (see chapter 6). The use of images preceded the use of signs (Campbell 2018; Chiera, 1966; Joseph 1993; Jung, 1964).
When shown their paralyzed or neglected left arm or leg, such patients may deny that it is their own and claim that it belongs to the doctor or a patient in the next room. Indeed, "patients with severe unilateral neglect behave as if a whole system of beliefs had vanished, as if one half of the inner model of the environment were simply deleted from their mind (Bisiach et al. 2013, p. 35). In the less extreme cases, patients may seem inattentive such that when their attention is directed to the left half of the environment, they are able to respond appropriately (see Jeannerod, 1987; and Umilta 1995, for a detailed review).
Imaginal and spatial-postional memory functioning also are disrupted (Nunn et al., 2009) such that patients may fail to attend to the left half of images recalled from memory. For example, Bisiach and Luzzatti (1978), found that when right brain damaged patients were asked to recall and describe a familiar scene from different perspectives, regardless of perspective (e.g. imagining a street from one direction and then from another) patients consistently failed to report details that fell to their left--although the same details were recalled when imagined from the opposite direction, their right. Bisiach and Luzzatti (1978) suggest that visual images and scenes may be split into two images when conjured up, such that the right hemisphere images the left half of space and the left brain the right half of space. Similar results were presented by Meador, Loring, Bowers, and Heilman, (1987).
Thus right cerebral injuries can result in visual-spatial as well as (imaginal) representational neglect. In some severe cases, patients may demonstrate both forms, whereas in others visual-spatial (but not representational) neglect may be found in isolation (Bartolomeo et al. 2004).
Neglect also can be influenced by the task demands (Bartolomeo et al. 2004; Binder et al 1992; Starkstein et al. 1993; Umilta 1995) and may be differentially expressed in the horizontal and vertical spatial dimension and in near vs far pepipersonal space (Mennemeir et al. 1992). For example, some patients may demonstrate neglect in the visual vs tactile modality (Umilta 1995), whereas yet others may adequately draw simple figures, but fail to correctly place numerals in the right half of space when drawing a clock.

Thus a patient with a left cerebral injury may not respond to a verbal question or command, or they may tend to ignore objects or written material that falls to their extreme right. Hence, left brain damaged patients also may show unilateral inattention or neglect (Albert, 1973; Bartolomeo et al. 2004; Cubelli et al. 2013; Denny-Brown et al. 1952; Gainotti et al., 1986), albeit in a less severe form.
These patients therefore also tend to demonstrate variable degrees of left sided neglect, and may also have considerable difficulty activating memories and correctly engaging in memory search (see chapter 19). In this regard, Meador et al., (1987) found that by turning the head to the left (thus supposedly more greatly activating the right hemisphere) that although still deficient, patients were able to recall more left sided objects and stimuli.
On the other hand, with smaller lesions involving the right frontal convexity, rather than a loss of arousal there can result a loss of control over arousal, and such patients can respond in a highly disinhibited fashion (Joseph, 1986a, 2009a). Such patients may also, however, demonstrate neglect. Presumably, due to the loss of counterbalancing right frontal input, the left frontal region may be unable to disengage from attending to the right half of auditory, visual and physical space so as to explore the (neglected) left half of space. However, this may be partly overcome by requiring the patient to physically orient toward the left (e.g. Meador et al. 1987).
In addition, unlike the left, the right hemisphere is responsive to tactual stimuli which impinge on either side of the body (Desmedt, 2017; Pardo et al. 2013). Indeed, a somesthetic image of the entire body appears to be maintained by the parietal lobe of the right half of the brain (Joseph, 2018a); and not just a body image, but memories of the body, the left half in particular.
When confronted by their unused or paralyzed extremities, such patients may claim that they belong to the doctor or a person in the next room or, conversely, seem indifferent to their condition and/or claim that their paralyzed limbs are normal --even when unable to comply with requests to move them.
"When asked why she could not move her hand she replied, 'somebody has ahold of it.' Another patient, when asked if anything was wrong with her hand said, 'I think it's the weather. I could warm it up and it would be alright.' One woman, when asked whether she could walk said, 'I could walk at home, but not here. It's slippery here." Another patient, when asked why he couldn't raise his arm said, 'I have a shirt on'" (Nathanson et al., 1952; p. 383).
Neglect & Denial: Emotional Reactions.
Many patients also appear indifferent and/or make inappropriate emotional remarks about their disability. Given right cerebral dominance for emotion, perhaps not surprisingly, many patients may appear and behave inappropriately and may even laugh and joked about being paralyzed.
In an extensive examination of these disturbances, E. Weinstein and Kahn (1950) found that of 22 patients (only 3 of whom were thought to have predominantly left hemisphere dysfunction), 15 were euphoric and manifested an air of serenity or bland unconcern about their condition despite the fact that they were suffering from disorders such as hemiplegia, blindness, loss of memory, and incontinence. Ten of these individuals also behaved in a labile or transiently paranoid fashion.
Right cerebral lesions have been reported to slow the appearance of phantom limbs on either side of the body and can result in the loss of phantom limb pain (S. Weinstein, 1978). In contrast, left sided lesions seem to have little effect. In general, like neglect and inattention, lesions which result in body image disturbances tend to involve the right parietal or right frontal lobe (Bradshaw & Mattingly, 1995; Critchley, 1953; Joseph, 1986a, 2018a; Stuss & Benson, 1986). In this regard, the fact that lesions of the left hemisphere rarely result in neglect or body image disturbances suggests that the right hemisphere maintains a bilateral representation, and memories of the body, whereas the left cerebrum maintains a unilateral representation and a memory of only the right half of the body -memories which are stored in the parietal lobe, a portion of which evolved from the hippocampus which is also concerned with memory, including memory of the body in space (chapters 13, 14). This bilateral representation of the body image vs the unilateral representation maintained by the left hemisphere, also explain the greater degree of hemiplegia and hemianesthesia which is seen after right vs left parietal lesions (Sterzi et al., 1993).
Hence, when the left hemisphere is damaged, the right hemisphere continues to monitor (and remember) both halves of the body and there is little or no neglect --an impression supported by findings which indicate that the right hemisphere electrophysiologically responds to stimuli that impinge on either side of the body, whereas the left hemisphere predominantly responds only to right sided stimulation (Desmedt, 2017; Pardo et al. 2013).
For example, one 48 year old housewife complained of diffuse, poorly localized (albeit intense) pain in her left leg, which occurred in spasms that lasted minutes. She subsequently was found to have a large tumor in the right parietal area, which, when removed, alleviated all further attacks. Head and Holmes (1911) reported a patient who suffered brief attacks of "electric shock"-like pain that radiated from his foot to the trunk; a glioma in the right parietal area subsequently was discovered. McFie and Zangwill (1960) reported an individual who began to experience intense, extreme pain in a phantom arm after a right posterior stroke.
In another instance reported by York et al., (1979), a 9-year-old boy experienced spontaneous attacks of intense scrotal and testicular pain and was found to have seizure activity in the right parietal area. Ruff (1980) reports two cases who experienced paroxsymal episodes of spontaneous and painful orgasm, which was secondary to right parietal seizure activity. In one patient the episodes began with the sensation of clitoral warmth, engorgement of the breasts, tachycardia, etc., all of which rapidly escalated to a painful climax. Interestingly, in the normal intact individual, orgasm is associated with electrohysiological arousal predominantly within the right hemisphere (H. Cohen et al. 1976).
It is important to note, however, that although the predominant focus for paroxysmal pain is the right hemisphere, pain also has been reported to occur with tumors or seizures activity that involves the left parietal region (Bhaskar, 1987; McFie & Zangwill, 1960).
Unfortunately, when the patient's symptoms are not considered from a neurological perspective, their complaints with regard to pain may be viewed as psychogenic in origin. This is because the sensation of pain, stiffness, engorgement, is, indeed, entirely "in their head" and based on distorted perceptual functioning at the level of the neocortex. Physical exam may reveal nothing wrong with the seemingly affected limb or organ -unless neocortical damage is extensive, in which case the patient may display paresis, sensory loss, etc. Even under these latter condtions such patients may be viewed as hysterical or hypochondriacle, particularly in that right hemisphere damage also disrupts emotional functioning. For example, in one case a woman patient suspected of hysteria subsquently was found to have a right frontal-parietal and mid-temporal cyst (Joseph, 2018a). This same individual had difficulty recognizing or mimicking emotional and environmental sounds.
It is noteworthy that individuals suspected of suffering from hysteria are two to four times more likely to experience pain and other distortions on the left side of the body (Axelrod et al. 1980; Galin, Diamond & Braff 2017; Ley 1980; D. Stern 2017); findings which in turn, suggest that the source of the hysteria may be a damaged right hemisphere.
This supposition is supported further by the raw MMPI data provided by Gasparrini, Satz, Heilman, and Cooldige (1978) in their study of differential effects of right vs. left hemisphere damage on emotional functioning. That is, whereas depression (elevated MMPI scale 2) is more likely subsequent to left cerebral damage, a pattern suggestive of hysteria and conversion reactions (elevations on scales 1 and 3, reductions on scale 2; i.e., the Conversion "V") are more likely subsequent to right hemispheric lesions.
The findings of Gasparrini et al. (1978) were replicated based on a restrospective case analysis of individuals with long standing right vs. left hemisphere injury (verified by neuropsychological exam coupled with CT-scan and/or EEG). In this study the MMPI Conversion V profile was found to be associated signficantly and almost exclusively with right hemisphere injuries (among males only), whereas elevated scale 2 was associated with those with left sided damage (Joseph, unpublished data). However, not every patient demonstrated this pattern.
At least one investigator, however, reporting on psychiatric patients has attributed hysteria to left cerebral damage (Flor-Henry 2013). In part this may be a function of the statistical permutations utilized in analyzing his data. However, this also may reflect the nature of the population studied (i.e., psychiatric rather than neurological) and, thus, the differential effect of long-standing drug treatments and biochemical and congenitial disturbances vs. recently acquired neuroanatomical lesions.

Be it the face of a friend or that of a stranger, the right hemisphere superiority for facial recognition is augmented by the additional display of facial emotion (Ley & Bryden, 1979; Suberi & McKeever, 2017). Indeed, not only is it predominant in perceiving facial emotion, regardless of the emotion conveyed (Buchtel et al. 1978; Dekosky et al. 1980; Landis et al. 1979; Strauss & Moscovitch, 2011; Suberi & McKeever, 2017) faces also are judged to be more intensely emotional when viewed exclusively by the right hemisphere (Heller & Levy, 2011).
Conversely, with right (but not left) cerebral injuries, patients tend to demonstrate an overall impairment in recalling, imaging, identifying and visualizing facial emotional expressions (Bowers et al. 2013; Young et al. 1995), and with right temporal atrophy, patients may suffer a progressive prosopagnosia (Evans et al. 1995).
In addition the left side of the face has been found to be more emotionally expressive (Campbell 1978; Chaurasis & Goswami 2005; Moreno et al. 2017; Sackheim et al. 1978) and to be perceived as more intensely emotional as well (Borod & Caron 1980; Sackheim & Gur 1978). In response to emotional stimuli, the left half of the face becomes more activated and a significant majority of individuals respond with conjugate lateral eye movements to the left (Schwartz et al. 2005; Tucker 2011); the left half of the body being under the control of the right hemisphere.
Conversely, damage to the right (but not left) hemisphere significantly reduces facial emotional expressiveness (Blonder et al. 1993).
In fact, the right hemisphere superiority for facial recognition is augmented by the additional display of facial emotion (Ley & Bryden 1979; Moreno, et al. 2017; Suberi & McKeever 2017), regardless of the emotion conveyed (Buchtel et al. 1978; Dekosky et al. 1980; Landis et al. 1979; Strauss & Moscovitch 2011; Suberi & McKeever, 2017). Faces also are judged to be more intensely emotional when viewed exclusively by the right hemisphere (Heller & Levy 2011) and the right hemisphere is dominant in regard to memory for facial expression as well (Weddell, 2017; reviewed in Bradshaw & Mattingly, 2013). Hence, the right half of the cerebrum is dominate for visual, facial, and auditory modes of emotional expression and perception including memory for faces and facial emotion.


Bill and Hillary Clinton. Tom Cruise
For example, one patient could not identify his wife, and although tested for 7 hours by the same examiner was unable to recognize him at the end of the session (De Renzi, 1986). Another patient was unable to recognize relatives, her pets, or even discriminate between people on the basis of sex but instead had to rely on the presence of details (such as lipstick, rouge, hair length, a moustache) to make discriminations (Levine, 1978).
DELUSIONS & FACIAL RECOGNITION
As noted above, lesions to the right cerebrum may result in difficulty recognizing, distinguishing or differentiating facial emotion (Bowers et al. 2013; DeKosky et al. 1980; Evans et al. 1995). That is, patients are unable to recognize or determine what others are feeling via facial expression.
Electrical stimulation of the posterior portion of the right middle temporal gyrus also results in an inability to correctly label the emotion shown in faces, whereas posterior right temporal stimulation disrupts visual-spatial memory for faces in general (Fried et al., 1982). Hence, the right hemisphere is clearly dominant for perceiving, recognizing, differentiating, expressing, and even recalling facial emotion.
In some instances, depending on the extent of damage, rather than a frank failure to recognize, patients may notice that friends, lovers, or their children, look different, strange, or unfamiliar --perceptions which may give rise to a host of abnormal emotional reactions and upheavals including frank paranoia, for example, fear that one's wife may have been replaced by an imposter.
Delusional misperception of familiar and unfamiliar individuals, as well as disturbances such as Capgras syndrome (delusional doubles; reduplication) or false identification, also can result from right hemisphere and/or (bilateral) frontal damage (Alexander et al. 1979; Benson et al. 1976; Hecaen, 1964; Jacocic & Staton 1993). For example, one patient who was looking into a dark tachistoscope suddenly said in an emotional voice, "I see my daughter --oh, she 's gone" and was unable to recognize ward personel or relatives when present (Levine, 1978).
As noted, a 47-year old woman who was suffering from a right superior temporal lobe, was able to name pictures of animals, tools and household objects but was almost completely unable to recognize and correctly name animal and humans sounds. However, she was better able to recognize non-living sounds such as a creaking door, or a hammer hammering--though these abilities were also compromised.
These findings, which require independent confirmation, raises the possibility that the right temporal lobe is specialized for perceiving faces and living creatures (and the sounds they make) whereas the left is somewhat more adept at perceiving and naming non-living things, such as tools and household objects.
There is some (albeit, controversial) evidence that the left hemisphere processes positive emotions, and is likely to view negative and neutral emotions as positive (Davidson, 2014; Davidson et al. 1985, 1987; Dimond & Farrington, 2017; Dimond, et al, 1976; Gainotti, 2001; Lee, et al. 2017, 1993; Ostrove et al. 2017; Otto et al. 1987; Rossi & Rosadini, 1967; Sackeim, et al. 1982; Schiff & Lamon, 2017; Silberman & Weingartner 1986; Terzian,1964). However, this appears to be a bias rather than a perceptual capability and in fact appears to be based on an inability to correctly perceive emotion. For example, when right cerebral influences are eliminated, such as due to right hemisphere damage or anesthetization, many patients are likely to view and report neutral and even negative events in a positive manner and to exhibit a positive mood including laughter (Gainotti, 2001; Lee et al. 2017; Rossi & Rosadini, 1967; Sackeim et al. 1982; Terzian 1964).
Rossi and Rosadini (1967), for example, injected sodium amytal into the left vs right hemisphere and found that in 68% of those with left hemisphere inactivation reacted with depression (expressed presumably by the awake right hemisphere). In contrast, 84% of those with right sided inactivation responded with euphoria (expressed presumably by the completely awake left hemisphere) and 16% responded in a depressed fashion (see also Gainotti, 2001; Lee et al. 2017; Rossi & Rosadini, 1967; Terzian 1964). These differences are not always observed, however. Indeed, I have observed approximately a dozen sodium amytal tests and never witnessed these changes in affect.
Nevertheless, when the left anterior region of the brain has been damaged or is dysfunctional, individuals are likely to respond with severe depression, or anger, irritability, and paranoia (Gainotti, 2001; Gruzelier & Manchanda, 1982; Hillbom, 1960; Joseph, 2009a; Lebrun, 1987; Robinson & Benson, 2011; Robinson & Szetela, 2011; Sherwin et al. 1982; Sinyour et al., 1986).
This suggests that when left cerebral ("positive") influences are negated, positive emotions are replaced by "negative" feeling states which in turn are a consequence of right cerebral emotional dominance; i.e. the right half of the brain is accurately perceiving the consequences of the injury and is understandably upset and depressed. In fact, with right prefrontal transcranial stimulation, patients report a significant reduction in depression (Kelin et al., 2009). However, in some cases those with massive left posterior and temporal lobe damage may respond with euphoria which in turn may be due to subsequent emotional disorganization in response to loss of comprehension. That is, they become euphoric as they no longer comprehend.
Therefore, although the left plays a minor role in regard to emotions, there is evidence to suggest that it is inclined to view or express emotional material in either a neutral or positive light, regardless of its actual affective value. This may also explain why following massive right frontal injuries (at least in males), "positive" emotions are sometimes expressed indiscriminately and inappropriately . Indeed, affected patients may appear to be in a manic state (see below).
OVERVIEW
Hence, when the right cerebrum is damaged there can result a myriad of peculiar disturbances that involve a number of modalities. Patients with body-image disturbances may seem emotionally abnormal and possibly hysterical rather than neurologically impaired. Those with facial agnosia may become paranoid and convinced that friends or lovers have been replaced by imposters. Individuals with intonational-melodic and emotional-linguistic deficiences may be unable to adequately vocally express their feelings, fail to recognize or misinterpret the feelings conveyed by others, as well as "miss the point" or fail to recognize discrepancies in speech, such as when presented with implausible information. Conversely, their own speech patterns and behavior may become abnormal, tangential, disinhibited, and contaminated by implausible, confabulatory, and delusional ideation.
Hence, in all instances, regardless of where within the right hemisphere damage occurs, social-emotional abnormalities may result. Indeed, emotional disturbances may be the dominant or only manifestation of a patient's illness. Unfortunately, if not accompanied by gross neurological signs, the possiblity of right hemisphere damage may be overlooked.
In short this formerly highly impressive and dignified man acted for a long time period after his stroke in a highly unusual and bizzare manner.

For example, M. Cohen and Niska (1980) report an individual with a subarachnoid hemorrhage and right temporal hematoma who developed an irritable mood; shortened sleep time; loud, grandiose, tangential speech; flight of ideas; and lability and who engaged in the buying of expensive commodities. Similarly, Oppler (1950) documented an individual with a good premorbid history who began to deteriorate over many years' time. Eventually, the patient developed flight of ideas, emotional elation, increased activity, hypomanic behavior, lability, extreme fearfulness, distractability, jocularity, and argumentativeness. The patient was also overly talkative and produced a great deal of tangential-circumstantial ideation with fears of persecution and delusions. Eventually a tumor was discovered (which weighed over 74 grams) and removed from the right frontal-parietal area.
Similarly, Spreen et al., (1965) describe a 65 year old man who following a right sided stroke (with left hemiparesis), developed extremely unpredictable behavior and lability. Regardless of external circumstances he would begin crying at one moment and at the next demonstrate irritability, happiness, or extreme depression. Secondary mania also has been reported with right frontal encephalopathy accompanied by biolectric epileptiform activity (Jack et al. 2013).
Over the course of the last 20 years, I have examined two dozen patients who who developed manic like symptoms after suffering a right frontal stroke or trauma to the right hemisphere (some of whom are described in Joseph, 1986a, 2018a); the majority of whom were males. All but three of the males had good premorbid histories and had worked steadily at the same job for over 3-5 years. Upon recovering from their injuries, all developed delusions of grandeur, pressured speech, flight of ideas, decreased need for sleep, indiscriminant financial activity, extreme emotional lability, and increased libido.
One patient, formerly very reserved, quiet, conservative, and dignified with more than 20 patents to his name, and who had been married to the same woman for over 25 years, began patronizing up to 4 different prostitutes a day and continued this activity for months. He left his job, began thinking up and attempting to act upon extravagant, grandiose schemes, and camped out at Disneyland and attempted to convince personnel there to finance his ideas for developing an amusement park on top of a mountain. At night he frequently had dreams in which either John F. or Robert Kennedy would appear and offer him advice --and he was a Republican!
RIGHT HEMISPHERE MENTAL FUNCTIONING
The right and left half of the brain are functionally lateralized. However, they also interact with each half if the brain assisting the other. This enables tasks to be performed more efficiently, and gives rise to creativity, insight, and greater mental ability--literally "two heads are better than one," except in the case of the human head, there are two brains, the right and left half of the brain, each specialized, and both communicating and share information via a massive rope of nerve fibers, the corpus callosum.
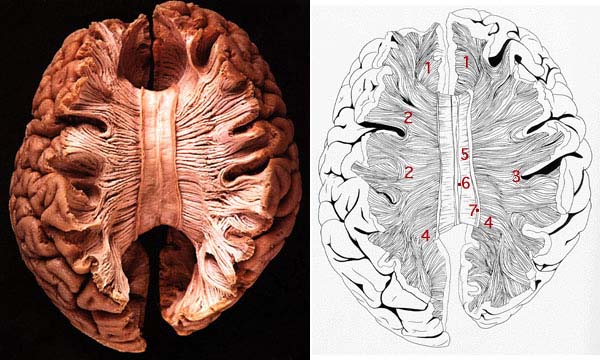

For example, when split-brain patients are tactually stimulated on the left side of the body, their left hemispheres demonstrate marked neglect when verbal responses are required, they are unable to name objects placed in the left hand, and they fail to report the presence of a moving or stationary stimulus in the left half of their visual fields (Bogen, 1979; Gazzaniga & LeDoux, 1978; Joseph, 2018b; Levy, 1974, 2013; Seymour et al. 2004; Sperry, 1982). They (i.e., their left hemisphere's) cannot verbally describe odors, pictures or auditory stimuli tachistoscopically or dichotically presented to the right cerebrum, and have extreme difficulty explaining why the left half of their bodies responds or behaves in a particular purposeful manner (such as when the right brain is selectively given a command). In addition, they demonstrate marked difficulties in naming incomplete figures (and thus forming visual closure), as well as a reduced ability to name and identify nonlinguistic and environmental sounds (Joseph, 1986b, 2018b)--capacities associated with the functional integrity of the right hemisphere.
However, by raising their left hand (which is controlled by the right half of the cerebrum) the disconnected right hemisphere is able to indicate when the patient is tactually or visually stimulated on the left side. When tachistoscopically presented with words to the left of visual midline, although unable to name them, when offered mutiple visual choices in full field their right hemispheres are usually able to point correctly with the left hand to the word viewed.

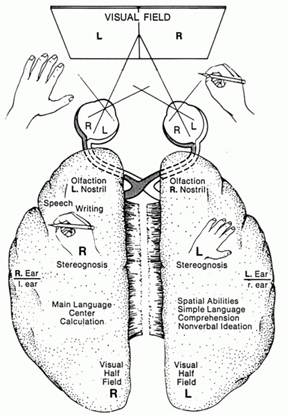

In this regard, when presented with words like "toothbrush", such that the word "tooth" falls in the left visual field (and thus, is transmitted to the right cerebrum) and the word "brush" falls in the right field (and goes to the left hemisphere), when offered the opportunity to point to several words (i.e., hair, tooth, coat, brush, etc.), the left hand usually will point to the word viewed by the right cerebrum (i.e., tooth) and the right hand to the word viewed by the left hemisphere (i.e., brush). When offered a verbal choice, the speaking (usually the left) hemisphere will respond "brush" and will deny seeing the word "tooth."
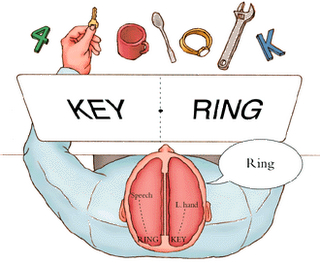
Overall, this indicates that the disconnected right and left cerebral hemispheres, although unable to communicate and directly share information, are nevertheless fully capable of independently generating and supporting mental activity (Bogen, 1969, 1979; Gazzaniga & LeDoux, 1978; Joseph, 1986b, 2018b; Levy, 1974, 2013; Sperry, 1982). Hence, in the right hemisphere we deal with a second form of awareness that accompanies in parallel what appears to be the "dominant" temporal-sequential, language dependent stream of consciousness in the left cerebrum.
Moreover, as has been demonstrated by Sperry, Bogen, Levy, and colleagues, the isolated right cerebral hemisphere, like the left, is capable of self-awareness, can plan for the future, has goals and aspirations, likes and dislikes, social and political awareness, can purposefully initiate behavior, guide responses choices and emotional reactions, as well as recall and act upon certain desires, impulses situations or environmental events --without the aid, knowledge or active (reflective) participation of the left half of the brain.
In its most subtle manifestations the disconnected right hemisphere may attempt to provide the left with clues when the left (speaking) hemisphere is called upon to describe or guess what type of stimulus has been secretly shown to the right (such as in a T-scope experiment). Because the corpus callosum has been severed transfer and information exchange is not otherwise possible.
Hene, when a picture has been shown to the right and the left has been asked to guess, the right hemisphere may listen and then nod the head or clear the throat so as to give clues or indicate to the left cerebrum that it has guessed incorrectly. In one case the right hemisphere attempted to trace or write an answer on the back of the right hand (e.g. Sperry et al. 1979). For example, after the right hemisphere was selectively shown a picture of Hitler, and then asked to indicate their attitude toward it before verbally describing it, the patient signalled "thumbs down".
EX: "That's another 'thumbs-down'?"
LB: "Guess I'm antisocial."
EX: "Who is it?"
LB: "GI came to mind, I mean..." Subject at this point was seen to be tracing letters with the first finger of the left hand on the back of his right hand.
EX: "You're writing with your left hand; let's keep the cues out."
LB: "Sorry about that."
Nevertheless, the behavior of the right hemisphere is not always cooperative, and sometimes it engages in behavior which the left cerebrum finds objectionable, embarrassing, puzzling, mysterious, and/or quite frustrating. This is probably true for the normal as well as the "split-brain" individual.
For example, Akelaitis (1945, p. 597) describes two patients with complete corpus callosotomies who experienced extreme difficulties making the two halves of their bodies cooperate. "In tasks requiring bimanual activity the left hand would frequently perform oppositely to what she desired to do with the right hand. For example, she would be putting on clothes with her right and pulling them off with her left, opening a door or drawer with her right hand and simultaneously pushing it shut with the left. These uncontrollable acts made her increasingly irritated and depressed."
Another patient experienced difficulty while shopping, the right hand would place something in the cart and the left hand would put it right back again. Both patients frequently experienced other difficulties as well . "I want to walk forward but something makes me go backward. ' A recently divorced male patient noted that on several occasions while walking about town he found himself forced to go some distance in another direction. Later (although his left hemisphere was not conscious of it at the time) it was discovered (by Dr. Akelaitis) that this diverted course, if continued, would have led him to his former wife's new home.
Geschwind (2011) reports a callosal patient who complained that his left hand on several occasions suddenly struck his wife--much to the embarrassment of his left (speaking) hemisphere. In another case, a patient's left hand attempted to choke the patient himself and had to be wrestled away (Goldstein; cited by Geschwind, 2011).

Brion and Jedynak (cited by Geschwind, 2011) indicate that this type of independent left sided (right hemisphere) activity was common in their split-brain patients and termined it the "alien hand."
In addition, Bogen (1979, p. 333) indicates that almost all of his "complete commissurotomy patients manifested some degree of intermanual conflict in the early postoperative period." One patient, Rocky, experienced situations in which his hands were uncooperative; the right would button up a shirt and the left would follow right behind and undo the buttons. For years, he complained of difficulty getting his left leg to go in the direction he (or rather his left hemisphere) desired. Another patient often referred to the left half of her body as "my little sister" when she was complaining of its peculiar and independent actions.
A split-brain patient described by Dimond (1980, p. 434) reported that once when she had overslept her "left hand slapped me awake." This same patient, in fact, complained of several instances where her left hand had acted violently. Similarly, Sweet (1945) describes a female patient whose left hand sometimes behaved oppositionally and in a fashion which on occasion was quite embarrassing.


Similar difficulties plagued a split-brain patient on whom I reported on (Joseph 2018b). Indeed, after callosotomy, this patient (2-C) frequently was confronted with situations in which his left extremities not only acted independently, but engaged in purposeful and complex behaviors --some of which he (or rather, his left hemisphere) found objectionable and annoying.
For example, 2-C complained of instances in which his left hand would perform socially inappropriate actions (e.g. attempting to strike a relative) and would act in a manner completely opposite to what he expressively intended, such as turn off the TV or change channels, even though he (or rather his left hemisphere) was enjoying the program. Once, after he had retrieved something from the refrigerator with his right hand, his left took the food, put it back on the shelf and retrieved a completely different item "Even though that's not what I wanted to eat!" On at least one occasion, his left leg refused to continue "going for a walk" and would only allow him to return home.
In the laboratory, he often became quite angry with his left hand, he struck it and expressed hate for it. Several times, his left and right hands were observed to engage in actual physical struggles. For example, on one task both hands were stimulated simultaneously (while out of view) with either the same or two different textured materials (e.g., sandpaper to the right, velvet to the left), and the patient was required to point (with the left and right hands simultaneously) to an array of fabrics that were hanging in view on the left and right of the testing apparatus. However, at no time was he informed that two different fabrics were being applied.
After stimulation the patient would pull his hands out from inside the apparatus and point with the left to the fabric felt by the left and with the right to the fabric felt by the right.
Surprisingly, although his left hand (right hemisphere) responded correctly, his left hemisphere vocalized: "Thats wrong!" Repeatedly he reached over with his right hand and tried to force his left extremity to point to the fabric experienced by the right (although the left hand responded correctly! His left hemisphere didn't know this, however.). His left hand refused to be moved and physically resisted being forced to point at anything different. In one instance a physical struggle ensued, the right grappling with the left.
Moreover, while 2-C was performing this (and other tasks), his left hemisphere made statements such as: "I hate this hand" or "This is so frustrating" and would strike his left hand with his right or punch his left arm. In these instances there could be little doubt that his right hemisphere was behaving with purposeful intent and understanding, whereas his left brain had absolutely no comprehension of why his left hand (right hemisphere) was behaving in this manner.
LATERALIZED GOALS AND ATTITUDES
Why the right and left cerebral hemispheres in some situations behave cooperatively and yet in others in an oppositional fashion is in part a function of functional lateralization and specialization and the differential representation of social-emotional analytical abilities predominantly within the right hemisphere. Hence, because each hemisphere is concerned with different types of information, even when analyzing ostensibly the same stimulus may react, interpret and process it differently and even reach different conclusions (Joseph, 2018b; Levy & Trevarthen, 1976). Moreover, even when the goals are the same, the two halves of the brain may produce and attempt to act on different strategies.
Functional lateralization may thus lead to the development of oppositional attitudes, goals and interests. For example, one split brain individual's left hand would not allow him to smoke, and would pluck lit cigarettes from his mouth or right hand and put them out. Apparently, although his left ceebrum wanted to smoke, his right hemisphere didn't approve -apparently he had been trying to quite for years.
As noted above, 2-C experienced conflicts when attempting to eat, watch TV, or go for walks, his right and left brain apparently enjoying different TV programs or types of food (Joseph 2018b). Nevertheless, these difficulties are not limited to split-brain patients, for conflicts of a similar nature often plague the intact individual as well.

For example, it is well known that the left hemisphere is responsible for the encoding and recall of verbal memories, whereas the right cerebrum is dominant in regard to visual-spatial, non-verbal, and emotional memory functioning (Barr, Goldberg, Wasserstein & Novelly 2017; Brewer et al., 1998; Fried et al. 1982; Frisk & Milner 2017; Hecaen & Albert, 1978; Kimura, 1963. Levy, 2013; Milner, 1962, 1968; Nunn et al., 2009; Sperry et al., 1979; Squire, 1992; Suberi & McKever, 2017; Wechsler, 1973; Whitehouse, 2011). If the left temporal lobe were destroyed, verbal memory functioning would become impaired since the right cerebrum does not readily store this type of information. Conversely, the left has great difficulty storing or remembering nonlinguistic, visual, spatial, and emotional information.
Specifically, left temporal lobectomy, seizures or lesions involving the inferior temporal areas can moderately disrupt immediate and severely impair delayed memory for verbal passages, and the recall of verbal paired-associates, consonant trigrams, word lists, number sequences, and conversations (Barr et al. 2017; Delaney et al. 1980; Kapur et al. 1992; Meyer & Yates,1955; Milner 1968; Milner & Teuber 1968; Samson & Zatorre, 1992; Weingartner 1968). Similarly, severe anterograde and retrograde memory loss for verbal material has been noted when the left anterior and posterior temporal regions (respectively) are electrically stimulated (Ojemann et al. 1968, 1971), lobectomized or injured (Barr et al. 2017; Kapur et al. 1992).
In contrast, right temporal lesions or lobectomy significantly impairs recognition memory for tactile and recurring visual stimuli such as faces and meaningless designs, memory for object position and orientation, and visual-pictorial stimuli, and short-term memory for melodies (Corkin 1965; Delaney et al. 1980; Kimura 1963; Milner 1968; Nunn et al., 2009; Ploner et al., 2009; Samson & Zatorre, 2018, 1992; Taylor 1979). Similarly, memory for emotional material is also significantly impaired with right vs left cerebral lesions (Cimino et al. 2013; Wechsler 1973) including the ability to recall or recognize emotional faces (DeKosky, et al. 1980; Fried et al. 1982; Weddell, 2017). Individuals with right hemisphere damage also have more difficulty recalling personal emotional memories (Cimino et al. 2013).
Hence, it is the left hemisphere which is responsible for the encoding and recall of verbal, temporal-sequential, and language related memories, whereas the right cerebrum is dominant in regard to visual-spatial, non-verbal, and social emotional memory functioning. Each hemisphere stores the type of material that it is best at recognizing, processing, and expressing.
UNILATERAL MEMORY STORAGE
In the intact, normal brain, even non-emotional memory traces appear to be stored unilaterally rather than laid down in both hemispheres (Bures & Buresova 1960; Doty & Overman 2017; Hasegawa et al., 1998; Kucharski et al. 2017; Levy, 1974; Risse & Gazzaniga, 1979). Moreover when one hemisphere learns, has certain experiences, and/or stores information in memory, this information is not always available to the opposing hemisphere; one hemisphere cannot always gain access to memories stored in the other half of the brain (Bures & Buresova 1960; Doty & Overman 2017; Hasegawa et al., 1998; Joseph, 1986b, 2018ab, 1992b; Kucharski et al. 2017; Levy, 1974; Risse & Gazzaniga 1979).
To gain access to these lateralized memories, one hemisphere has to activate the memory banks of the other brain half via the corpus callosum (Doty & Overman, 2017; Hasegawa et al., 1998) or anterior commissure (Kucharski et al. 2017). This has been demonstrated experimentally in primates. For example, after one hemisphere had been trained to perform a certain task, although either hemisphere could respond correctly once it was learned, when the commissures were subsequently cut, only the hemisphere that originally was trained was able to perform--i.e., could recall it. The untrained hemisphere acted as though it never had been exposed to the task; its ability to retrieve the original memories was now abolished (Doty & Overman, 2017; see also Hasegawa et al., 1998).
In a conceptually similar study, Risse and Gazzaniga (1979) injected sodium amytal into the left carotid arteries of intact patients so as to anesthetize the left cerebral hemisphere. After the left cerebrum was inactivated, the awake right hemisphere, although unable to speak, was still able to follow and behaviorally respond to commands, e.g., palpating an object with the left hand.
Once the left hemisphere had recovered from the drug, as determined by the return of speech and motor functioning, none of the eight patients studied was able to verbally recall what objects had been palpated with the left hand, "even after considerable probing." Although encouraged to guess most patients refused to try and insisted that they did not remember anything. However, when offered multiple choices in full field, most patients immediately raised the left hand and pointed to the correct object!
According to Risse and Gazzaniga (1979), although the memory of touching and palpating the object was not accessible to the verbal (left hemisphere) memory system, it was encoded in a nonverbal form within the right hemisphere and was unavailable to the left hemisphere when normal function returned. The left (speaking) hemisphere was unable to gain access to information and memories stored within the right half of the brain. Nevertheless, the right brain not only remembered, but was able to act on its memories.
This indicates that when exchange and transfer is not possible, is in some manner inhibited, or if for any reason the two halves of the brain become functionally disconnected and are unable to share information, the possibility of information transfer at a later time is precluded (Bures & Buresova, 1960; Hasegawa et al., 1998; Kucharski et al. 2017; Risse & Gazzaniga, 1979) -even when the ability to transfer is acquired or restored. The information is lost to the opposite half of the cerebrum.
Moreover, because some types of information are processed by the right and left hemisphere in a wholly different fashion, they are unable to completely share or gain access to the data or even the conclusions reached by the other -as they are unable to process or recognize it -which in turn precludes complete interhemispheric transfer (Berlucchi & Rizzolatti, 1968; Hicks, 1974; Joseph, 1982, 2018a; Marzi, 1986; Merriam & Gardner, 1987; Miller, 2017, 2013; Myers, 1959, 1962; Rizzolatti et al. 1971; Taylor & Heilman 1980); information is lost during the transfer process.
Nevertheless, although inaccessible or lost, these memories, details, and attached feelings can continue to influence whole brain functioning in subtle as well as in profound ways. That is, one hemisphere may experience and store certain information in memory and at a later time in response to certain situations act on those memories, much to the surprise, perplexity, or chagrin of the other half of the brain; one hemisphere cannot always gain access to memories stored in the other half of the brain.
Dreaming.
Of course, complete functional deactivation is probably quite rare in the normal brain. However, there is some evidence to suggest that interhemispheric communication is reduced, for example, during sleep and possibly during dreaming (Banquet, 2013; Joseph, 2018a).
Most dreaming occurs during REM, which possibly is associated with right hemisphere activation and low-level left hemisphere arousal (Goldstein et al. 2001; Hodoba, 1986; Meyer et al. 1987). It also becomes progressively more difficult to recall one's dreams as one spends time in or awakens during NonREM (Wolpert & Trosman, 1958), which is associated with high left hemisphere and low right brain activation (Goldstein et al. 2001). Thus are dreams really forgotten, or are they locked away in a code which is not accessible to the speaking left hemisphere?
Most individuals awakened during REM report dream activity approximately 80% of the time. When awakened during the N-REM period, dreams are reported approximately 20% of the time (Foulkes, 1962; Goodenough et al. 1959; Monroe et al. 1965) However, the type of dreaming that occurs during REM vs. N-REM is quite different. For example, N-REM dreams (when they occur) are often quite similar to thinking and speech (i.e. lingusitic thought), such that a kind-of rambling verbal monologue is experienced in the absence of imagery (Foulkes 1962; Monroe et al. 1965) It is also during N-REM in which an individual is most likely to talk in his or her sleep (Kamiya, 1961). In contrast, REM dreams involve a considerable degree of visual imagery, emotion, and tend to be distorted and implausible to various degrees (Foulkes, 1962; Monroe et al. 1965).
REM is characterized by high levels of activity within the brainstem, occipital lobe, and other nuclei (Hobson, et al. 1986; Steriade & McCarley 2017; Vertes 2017) It also has been reported that electrophysiologically the right hemisphere becomes highly active during REM, whereas, conversely, the left brain becomes more active during N-REM (Goldstein et al. 2001; Hodoba, 1986). Similarly, measurements of cerebral blood flow have shown an increase in the right temporal and parietal regions during REM sleep and in subjects who upon wakening report visual, hypnagogic, hallucinatory and auditory dreaming (Meyer et al., 1987).
Interestingly, abnormal and enhanced activity in the right temporal and temporal-occipital area acts to increase dreaming and REM sleep for an atypically long time period. Similarly, REM sleep increases activity in this same region much more than in the left hemisphere (Hodoba, 1986), which indicates that there is a specific complementary relationship between REM sleep and right temporal-occipital electrophysiological activity.
At least one group of investigators, however, have failed to find significant hemispheric EEG differences between REM and NREM (Ehrlichman et al. 1985).
DAY DREAMS, NIGHT DREAMS, AND HEMISPHERIC OSCILLATION
There is some evidence to suggest that during the course of the day and night the two cerebral hemispheres oscillate in activity every 90 to 100 minutes and are 180 degrees out of phase --a cycle that corresponds to changes in cognitive efficiency, the appearance of day dreams, REM (dream sleep), and, conversely, N-REM sleep (Bertini et al. 2013; Broughton, 1982; Gordon et al. 1982; cited by Hodoba, 1986; Klein & Armitage, 1979; Kripke & Sonnenschein, 1973; Levie et al. 2013, cited by Hodoba, 1986). That is, like two pistons sliding up and down, it appears that when the right cerebrum is functionally at its peak of activity, the left hemisphere is correspondingly at its nadir.
Similarly, shifts in cognitive abilities associated with the right and left hemisphere have been found during these cyclic changes during the day and after awakenings from REM and N-REM sleep. That is, performance across a number of tasks associated with left hemisphere cognitive efficiency is maximal during N-REM, whereas, conversely, right hemisphere performance (e.g., point localization, shape identification, orientation in space) is maximal after REM awakenings (Bertini et al.,2013; Gordon et al., 1982; Levie et al., 2013; cited by Hodoba, 1986). Moreover, Bertini et al., (2013) found that left hand motor dexterity (in right handed subjects) was superior to the right when awakened during REM and that the opposite relationship was found during NREM, i.e. right hand superiority (see Hodoba, 1986, for review.)
Conversely, there have been reports of patients with right cerebral damage who have ceased dreaming altogether or to dream only in words (Humphrey & Zangwill, 1951; Kerr & Foulkes, 1978, 2011). For example, defective dreaming, deficits that involve visual imagery, and loss of hypnagogic imagery have been found in patients with focal lesions or hypoplasia of the posterior right hemisphere and abnormalities in the corpus callosum (Botez et al. 1985; Kerr & Foulkes, 2011; Murri et al. 2014).
An absence or diminished amount of dreaming during sleep also has been reported after split-brain surgery; i.e., as reported by the disconnected left hemispehere (Bogen & Bogen, 1969; Hoppe & Bogen, 2017). Similarly, a paucity of REM episodes have been noted in other callosotomy patients, although these particular individuals continued to report some dream activity (Greenwood, Wilson, & Gazzaniga, 2017).
On the other hand it has been reported that when the left hemisphere has been damaged, particularly the posterior portions (i.e. aphasic patients), the ability to verbally report and recall dreams also is greatly attenuated (Murri et al., 2014; Pena-Casanova & Roig-Rovira, 1985; Schanfald et al. 1985). Of course, aphasics have difficulty describing much of anything, let alone their dreams. Moreover, Language Axis disconnection from the right hemisphere would also account for this failure to verbally report dreams and related imagery.
In some respects, however, a parallel between these latter findings and those of Risse and Gazzaniga (1979) in their amytal studies may be explanatory regarding the failure to report dreams with left hemisphere lesions. That is, with left hemisphere damage or when it is at a low level of arousal, the ability to verbally recall or report events experienced or generated by the right hemisphere appears to be reduced; i.e., the left (speaking) half of the brain cannot remember because it cannot gain access to right cerebral memory centers.
Thus, it appears that the right hemisphere provides the physiological foundation from which dreams in part derive their source and origin (Goldstein, et al. 2001; Hodoba, 1986; Joseph, 2018a; Meyer et al.,1987). However, in some instances in which these dream centers are disconnected from the language dominant left hemisphere, due to posterior right or left hemisphere lesions or after callosotomy, the ability to recall, report, and/or to produce vivid visual and hypnogogic dream imagery is attenuated (Joseph, 2018a).
However, also important in the capacity to engage in memory search and retrieval, or to dream and fantasize, are the frontal lobes (chapter 19). Likewise, frontal lobe damage and lobotomy also have been reported to abolish dreaming (Freeman & Watts, 1942, 1943).
HALLUCINATIONS
In addition to dream production, the right hemisphere also appears to be the dominant source for complex non-linguistic hallucinations. Specifically, tumors or electrical stimulation of the right hemisphere or temporal lobe are much more likely to result in complex visual as well as musical and singing hallucinations, whereas left cerebral tumors or activation gives rise to hallucinations of words or sentences (Berrios, 2017; Halgren, et al. 1978; Hecaen & Albert, 1978; Jackson, 1880; Mullan & Penfield, 1959; Penfield & Perot, 1963; Teuber et al. 1960). Conversely, LSD induced hallucinations are significantly reduced following right but not left temporal lobe surgical removal (Serafetinides, 1965), and dreaming is sometimes abolished with right but not left temporal lobe removals (Kerr & Foulkes, 2011). In one study, however, it was reported that an alcoholic patient with a right subcortical injury and left sided neglect, experienced hallucinations only in the right (non-neglected) half of visual-space (Chamorro et al. 2017).

Unfortunately, those making these claims appear to have confused apraxia, agraphia, and disturbances of language as somehow indicative of imagery deficits, and/or the ability to quickly verbally describe a verbal "image" as somehow synonymous with imagery production.
For example, in some studies, patients were required to verbally describe and/or draw the object they have "visualized" (e.g. Farah et al. 2018). In other words, imagery production was not assessed, but drawing and verbal ability. those imagery experiments with individuals with left posterior lesions are equally suspect in that a common abnormality would be word finding difficulty. Thus, these left cerebral "imaginal" deficits appear to be more a function of naming and word finding disturbances coupled with visual agnosia and/or apraxia -disturbances associated with left posterior (parietal) injuries (see also Trojano & Grossi 2004).
Individuals with left hemisphere damage have difficulty accurately describing much of anything, let alone visual images which is clearly more the productive province of the right hemisphere. Indeed, those who champion a left cerebral dominance for imagery have very little evidence to support their claims and instead appear to be confusing disconnection syndromes and apraxic and language disorders for imagery deficits (see also Sergent 2017).

Nevertheless, the left cerebrum no doubt can produce a variety of images, especially in response to a verbal command (Goldenberg 2017). However, as noted above, right (but not left) cerebral injuries can result a failure or inability to generate half of visual images (Bartolomeo et al. 2004) -which indicates that in the absence of right cerebral input, the left hemisphere is producing only a partial image and is therefore lacking dominance in this regard.

DREAM STIMULANTS
Since the right hemisphere utilizes a form of visual-spatial and emotional language which the left hemisphere does not speak, dream imagery often seems incomprehensible to the language dependent half of the brain, even when it is providing the accompanying narrative or dialogue. Rather, the non-temporal, often gestalt nature of dreams appears to require that they be consciously scrutinized from multiple angles in order to discern their meaning, for the last may be first and what is missing may be just as significant as what is present (Freud 1900; Jung 1945, 1964). This is because the right hemisphere analyzes and expresses information in a manner much different form the left.
Moreover, in that the right hemisphere is at a higher level of activation during REM, it also tends to predominate when attempting to analyze internal and external sensory input during this stage of sleep. For example, although sensory perception is restricted, the right cerebrum may respond to a sensation experienced during sleep by creating a dream to explain it. However, when this occurs we sometimes dream backwards.
A case in point, "Trish" dreams she is walking in San Francisco lugging large bags of gifts. Feeling tired she sets them down on the sidewalk. She looks for a bus and see a cable car coming. As it pulls up the conductor begins to ring its bell. The sound of the bell grows louder and then jolts her awake. Fully awake she realizes that someone is ringing her doorbell. In this regard, the hearing of the bell seemed to be a natural part of the dream, and it is. What seems paradoxical, however, is that the dream seemed to lead up to the bell so that its ringing made sense in the context of the dream.
The dream did not lead up to the bell, however, for the bell initiated the dream. The dream was produced, via the unique language of the right hemisphere during sleep (as well as amygdala activation), so as to explain the sound of the bell. The bell was heard and the dream was instantly produced in explanation and association. The bell stimulated the dream (perhaps by startling the amygdala; see chapter 13) which may have only last a second.
Since most dreams often last only a few seconds (although they may seem to take place over the course of hours) and since the right hemisphere does not analyze in temporal and linear units, the sequence of events is not all that important, that is, to the right half of the brain.
Fortunately, backward dreams are the most easily comprehended because the left cerebrum recalls the dream from its ending forward, and then like a reflection from a mirror reverses all that is perceived so that it makes temporal-sequential sense. Backward dreams also tend to be devoid of hidden meanings.
For example, one individual (described by Freud, 1900) dreamt he was in 18th Century France in the midst of the French Revolution. After a trial in which he was been found guilty, he was being led down a street lined with yelling and cursing Frenchmen and women. At the end of the street he could see the gallows where the heads of various political criminals were being chopped off at the neck. Mad with fright he felt and saw himself led up the stairs and his head being placed in the yoke of the chopping block. The executioner gave the signal, the crowd screamed its approval, he could hear and sense the blade falling, and with a loud crack it struck him across the neck. Indeed, it struck him with such a jolt that he awoke to find that his poster bed had broke and that a railing had fallen and struck him across the side and back of his neck.
Dream Patterns.
Although dreams probably serve a number of purposes, and at times are highly improbable and bizzare, they sometimes reflect something significant about the mental and emotional life of the dreamer, as well as other issues of concern to the right half of the brain. For example, when subjects are awakened repeatedly during REM over the course of several days, often an evolving thematic pattern, like an unfoldinig story, can be discerned (Cartwright et al. 1980). These patterns frequently reflect mental-emotional activity concerned with the solution of particular problems (Cartwright et al. 1980; Freud, 1900; Joseph 1992b; Jung, 1945, 1964; Kramer et al. 1964).
For example, one subject, a student, noted that "after being woken many times and seeing three or four dreams a night, I could realize there was a certain problem being worked out, like coping with responsibilities that were thrust upon me, but that weren't necessarily my own but I took on anyway. It was working out the feelings of resentment of taking somebody else's responsibility, but I met them well in my dreams. A good thing about spending time in the sleep lab was you could relate a common bond to some of the" dreams (Cartwright et al., 1980, p. 277). Similar bonds and patterns were, of course, recognized by Freud (1900) and Jung (1945) many years ago.
DREAMS & EMOTIONAL TRAUMA
Given right cerebral dominance for emotion and dream production, it is perhaps not surprising that emotional conflicts and traumas are often represented via dream imagery (Freud, 1900, Joseph, 1992b; Jung, 1945).
Consider, for example, Sara, who was desribed by her parents as a "very good and obedient" 6 year old girl who suffered from "night terrors" (Joseph, 1992b). Specifically, she frequently woke up at night screaming about the "creek," that ran near her home. According to her mother, Sara was afraid because she had been told that hoboes lived under the bridge and to never go there without her mommy or daddy, because they would "get" her.
I had Sara tell me her dream.
Sara: "I'm walking on the sidewalk near the big creek. Then I go to the edge and stare down at the big rocks at the bottom. All at once the whole world starts to shake. Like it's turning upside down. It's trying to throw me into the creek. I get scared and start to be afraid and start grabbing at the trees and bushes to keep from falling into the creek and onto the big rocks. Sometimes I see this hole and I crawl in. Then everything is OK. Sometimes I fall and fall and fall and I can see the big rocks coming closer and I know I'm going to fall on them. When I fall on them I wake up 'cause it hurts."
Sara had this particular dream repeatedly. Was it really because she was afraid of the hoboes and the creek?
In Sara's case it is noteworthy that her parents fought, screamed, yelled, and argued almost non-stop and there were constant threats of divorce. From talking within Sara it was also apparent that she was traumatized by her parents constant fighting -although they were both ostensibly quite good to her. Nevertheless, Sara's world was being turned up side down and was in complete chaos, which was reflected in her dreams.
Sara also had another troubling and recurrent dream where she went riding on her bike and when she came back to her street, her house was gone. Every house on the street was the same, including the neighbors. But when she asked about her family, no one knew what she was talking about and no one recognized her.
Although the symbolic content of these dreams was not apparent to Sara or her mother, one need not be a psychiatrist to decipher their obvious meaning. Sara's emotional world was literally being turned upside down and was in chaos due to the horrible fighting engaged in by her parents. She was terrified of losing her home and the catastrophe she perceived as befalling her family. Her very identity and functional integrity as a person was at stake, for if she lost her family she lost her Self.
The dream imagery involving the hole that she climbs into is also very interesting in that it suggests the desire to return to the safety and security of the womb. On the other hand, the hole" may have been exactly that, a hole. Indeed, dreams frequently mean exactly what they seem to mean (Jung, 1945).
Nevertheless, it might reasonably be asked, if dreams are of importance and not merely reflective of random and purely confabulatory ideation, why are they so difficult to recall? In part, as noted in the introduction to this section, this may be a function of lateralization, alterations in hemispheric arousal and activity, differential memory storage, and decreased interhemispheric communication during REM. Perhaps they only can be recalled by the right hemisphere. Of course, in some instances, dreams are probably nothing more than mundane and confabulatory noise.
Information processed and experienced during infancy vs. adulthood is stored via certain transformations and retrieval strategies which are quite different. As the brain matures and new information processing strategies are learned and developed, the manner in which information is processed and stored is altered. Although these early memories are stored within the brain, the organism no longer has the means of retrieving them, i.e., the key no longer fits the lock.
That is, early experiences may be unrecallable because infants use a different system of codes to store memories whereas adults use symbols and associations (such as language) not yet fully available to the child (Dollard & Miller, 1950; Joseph, 1982, 2017; Piaget, 1952, 1962, 1974). Much of what was experienced and committed to memory during early childhood took place prior to the development of lingusitic labeling ability and was based on a pre- or nonlinguistic code (Dollard & Miller, 1950; Freud, 1900). Hence, the adult, who is relying on more sophisticated and language-related coding systems, cannot find the right set of neural programs to open the door to childhood memories. The key does not fit the lock because the key and the lock have changed.
The inability to recall early memories is also a function of programmed cell death--the loss of memory-laden neurons which are shed by the millions over the course of early development and the immaturity of the corpus callosum in children (Joseph, 2009b, 2017). That is, non-verbal information perceived and processed by the right vs left hemisphere is generally stored in the right vs left hemisphere. Later, when the commissures mature, this information cannot be transferred except under special conditions.
However, under conditions of traumatic memory loss; e.g. repression, not just the the right and left hemisphere, but the differential activation of the amygdala and hippocampus are contributory; and later the recall of such memories may be opposed by the frontal lobes; the right frontal in particular (see chapter 19)
EMOTION AND RIGHT BRAIN FUNCTIONING IN CHIDLREN
As is now well known, the developing organism is extremely vulnerable to early experience during infancy such that the nervous system, perceptual functioning and behavior may be altered dramatically (Bowlby 1982; Casagrande & Joseph 1978; 1980; Diamond, 1985, 2013; Denenberg 2011; Ecknerode et al. 1993; Harlow & Harlow 1965; Joseph 1979; Joseph & Casagrande 1980; Joseph & Gallagher 1980; Joseph et al. 1978; Langmeier & Matejcek 2005; Rosenzweig 1971; Salzinger et al.1993; Sternberg et al. 1993). Interestingly, there is some evidence that the right cerebral hemisphere and the right amygdala may be more greatly affected (see Denenberg 2011; Diamond, 1985).
Moreover during these same early years our traumas, fears, and other emotional experiences, like those of an adult, are mediated not only by the limbic system, but also via the nonlinguistic, social-emotional right hemisphere. And, just as they are in adulthood, these experiences are stored in the memory banks of the right cerebrum.
However, much of what was experienced and learned by the right half of the brain during these early years was not always shared or available for left hemisphere scrutiny (and vice-versa). That is, a childs two hemispheres are not only functionally lateralized, but limited in their ability to share and transfer information. In many ways, infants and young children have split-brains (Deruelle & de Schonen, 2013; Finlayson 2005; Gallagher & Joseph 1982; Galin et al. 1979ab; Joseph & Gallagher, 1985; Joseph et al. 2014; Kraft et al. 1980; Molfese et al. 2005; O'Leary 1980; Ramaeker & Njiokiktjien, 2013; Salamy 1978; Yakovlev & Lecours 1967).
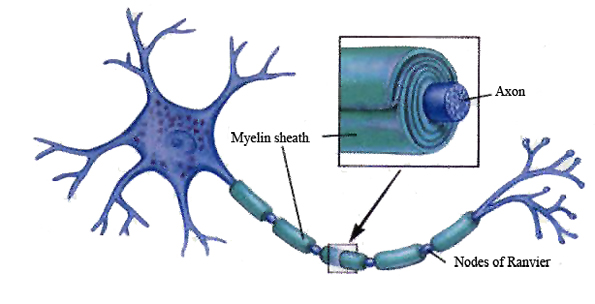
Nerves are insulted with myelin. Myelin is like the rubber coating that covers naked wires. This insulation, be it on a naked wire or an axonal appendage of a neurons, enables information to travel more efficiently between neurons. However, myelination of axons can take up to 10 years after a child is born. Therefore, information process is not as efficient in the brain of a child vs an adult.

The corpus callosum, the major axonal fiber pathway that connects the right and left half of the brain, takes over 10 years to fully myelinate.
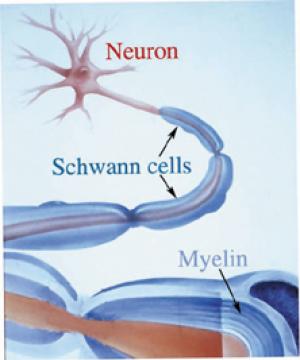
Due to the immaturity of the corpus callosum and in particular, the slow rate of axonal myelin within the callosum (Yakovlev & Lecours, 1967), communication is so poor that children as old as age 4 have mild difficulty transfering tactile, auditory, or visual information between the hemispheres, e.g., accurately describing complex pictures shown to the right brain (Joseph et al. 2014). That is, in addition to differential functional specialization, the slow development of the myelination process can in turn slow and disrupt axonal information transmission (e.g. Konner, 2013; Ritchie, 2014; Salamy 1978).
Indeed, although pain can be transmitted and received via axons devoid of myelin, this type of data is completely lacking in complexity and is devoid of initial cognitive attributes. Pain transmitting axons have very simple requirements. By contrast as data complexity increases, so too does the complexity of those neurons which transmit these signals (Konner, 2013; Ritchie, 2014).
For example, as axon diameter increases, so too does the extent of myelination. Similarly, quantities of axoplasm and cytoplasm, nuclear diameter, and neuronal packing density are also correlated with myelination (Konner, 2013). Conversely, lack of myelin, or those neurons which have not yet myelinated, are associated with an increased susceptibility to conduction failure and interference due extraneous influences, including signal modification by neighboring axons (Konner, 2013; Ritchie, 2014).
Moreover, particularly when one is dealing with complex or emotional information situations probably sometimes arise in which one brain half has little or no knowledge as to what is occurring in the other (Dimond, 1980; Dimond & Beaumont, 1974; Dimond et al. 2001; Geschwind, 1965; Joseph, 1982, 2018a, 1992b; Joseph et al. 2014; Marzi, 1986; Myers, 1959). In part, this is a consequence of lateralized specialization. Certain forms of information can only be processed and, thus, recognized by the right or left half of the brain. Even information that is transferred may be subject to interpretation and miss-interpretation (Joseph, 1982, 1986ab, 2018ab; Joseph et al., 2014) and this includes even the learning of sequential and fine motor movements (Hicks 1974; Taylor & Heilman 1980; see also Parlow & Kinsbourne 2017).
In addition, one brain half can be prevented from knowing what is occurring in the opposite half due to inhibitory or suppressive actions initiated by, for example, the frontal lobes, such that certain forms of information are suppressed, censored, and inter-hemispheric (as well as intra-hemispheric) information transmission prevented (Hoppe, 2017; Joseph, 1982, 2018a, 1992b). Thus, there sometimes results a functional commissurotomy (Hoppe, 2017; see also Galin, 1974).
Therefore, these three conditions --lateralized specialization, frontal lobe inhibitory activity, and incomplete myelination of callosum axons-- can reduce the ability of the two hemispheres to communicate among normal, intact individuals. Hence, in many ways the brain of even a normal adult is functionally split and disconnected, and for good reason. These conditions protect the brain and linguistic consciousness from becoming overwhelmed. As we have seen with frontal lobe damage, when communication is allowed to occur freely (due to disinhibition) the overall integrity of the brain to function normally is curtailed dramatically.
Nevertheless, a unique side effect of having two brains that are not always able to communicate completely and successsfully is intra-psychic conflicts. That is, we sometimes find ourselves feeling happy, sad, depressed, angry, etc. without a clue as to the cause. In other instances, we actually may commit certain thoughtless, impulsive, overly emotional, or embarassing actions and "have no idea" as to "what came over" us. To posit the notion that we have such experiences simpl "because" is absurd. Nor, among "normals," are such experiences always due to biochemical fluctuations or the result of "unconscious" urges. Rather, unbeknownst to the left brain, sometimes the right perceives, remembers, or responds to some external or internal source of experience and/or to its own memories and, thus, reacts in an emotional manner. The left (speaking ) hemisphere in turn only knows that it is feeling something but is unsure what or why , or, conversely, confabulates various denials, rationalizations and explanations which it accepts as fact.
THE ONTOLOGY OF EMOTIONAL CONFLICT
Because of lateralization and limited exchange, the effects of early "socializing" experience can have potentially profund effects. "As a good deal of this early experience is likely to have unpleasant if not traumatic moments, it is fascinating to consider the later ramifications of early emotional learning occurring in the right hemisphere unbeknownst to the left; learning and associated emotional responding that later may be completely inaccessibile to the language centers of the left half of the brain. That is, although limited transfer in children confers advantages, it also provides for the eventual development of a number of very significant psychic conflicts --many of which do not become apparent until much later in life."
Moreover, due to the immaturity of the callosum, children frequently can encounter situations in which the right and left cerebrum not only differentially perceive what is going on, but are unable to link these experiences so as to understand fully what is occurring or to correct misperceptions (Galin, 1974; Joseph, 1982, 1992b). Consider, for example, a young divorced mother with ambivalent feelings toward her young son (Galin, 1974; Joseph, 2018a, 1992b).
Although she does not express these feelings verbally, she conveys them through her tone of voice, facial expression, and in the manner in which she touches her son. She knows that she should love him, and at some level she does. She wants to be a good mother and makes herself go through the motions. However, she also resents her son because she has lost her freedom, he is a financial burden, and he may hinder her in finding a desirable mate. She is confronted by two opposing attitudes, one of which is unacceptable to the image she has of a good mother. Like many of us, she must prevent these feelings from reaching linguistic consciousness. However, this does not prevent them from being expressed nonlingusitically via. the right brain.
Her son, of course, also has a right hemisphere which perceives her tension and amibivalance. The right half of his brain notes the stiffness when his mother holds or touches him and is aware of the manner in which she sometimes looks at him . Worse, when she says, "I love you," his right hemisphere senses the tension and tone of her voice and correctly perceives that what she means is, "I don't want you" or, "I hate you. " His left hemisphere hears, however, "I love you" and notes only that she is attentive. He is in a "double bind" conflict, with no way for his two cerebral hemispheres to match impressions.
The right half of this little boy's brain feels something painful when the words "I love you" are spoken. When his mother touches him, he becomes stiff and withdrawn because his right hemisphere, via the analysis of facial expression, emotional tone, tactile sensation, etc. is fully aware that she does not want him.
Later, as an adult, this same young man has one failed relationship after another. He feels that he can't trust women, often feels rejected, and when a girl or woman says "I love you," it makes him want to cringe, run away, or strike out. As an adult, his left hemisphere hears "Love," and his right cerebrum feels pain and rejection.
Because the two halves of his cerebrum were not in communication during early childhood, his ability to gain insight into the source of his problems is greatly restricted. The left half of his brain cannot access these memories. It has "no idea" as to the cause of his conflicts.
In this regard, this asymmetrical arrangement of hemispheric function and maturation may well predispose the developing child in later life to come upon situations in which it finds itself responding emotionally, nervously, anxiously, or neurotically, without linguistic knowledge, or without even the possiblity of linguistic comprehension as to the cause, purpose, eliciting stimulus, or origin of its behavior. As a child or an adult, it may find itself faced with behavior that is mysterious, embarrassing, etc. "I don't know what came over me."
It also appears to be more involved than the left in the production of certain forms of visual imagery (see Ehrlichman & Barrett, 2013, Trojano & Gross, 2004, for contradictory evidence), dreams during REM sleep, as well as day dreams during waking. Conversely, the left brain appears to be associated with non-REM sleep and the thinking-type of mentation that sometimes occurs during this stage. However, the left hemisphere probably provides much of the dialogue and commentary that accompany dream activity.
A considerable body of evidence indicates that the right hemisphere is dominant in the comprehension and expression of prosodic, melodic, and emotional features of speech, the expression and perception of visual, facial, and verbal affect, and the ability to determine a persons mood, attitude and intentions via the analysis of gesture, facial expression, vocal-melodic and intonational qualities. However, the female right hemisphere appears to have more neocortical space devoted to these social-emotional and related language functions, whereas the in males, more neocortical space is devoted to spatial perceptual processing.
Because the right half of the brain is dominant in regard to most if not all aspects of social-emotional functioning, when it is damaged a myriad of affective disturbances may result. These include mania, depression, hysteria, gross social-emotional disinhibition, euphoria, childishness, puerility, or, conversely, complete indifference and apathy.
Patients may become delusional, engage in the production of bizzare confabulations, and experience a host of somatic disturbances that range from pain and body-perceptual distortions to seizure induced sexual activity and orgasm. They may fail to recognize the left half of their own bodies or, in other instances, fail to recognize the faces of friends, loved ones, or even their pets. In fact, individuals with right-sided lesions develop a greater degree of hemiplegia and hemianesthesia (Sterzi et al., 1993) and show less recovery and are more likely to die as compared to those with left sided destruction (Denes et al. 1982; Hobhouse, 1936; Hurwitz & Adams, 2001; Knapp, 1959; Marquardsen, 1969).
On the other hand, geriatric patients with left hemisphere strokes are more likely to develop decrease immunological functioning and severe infection (Kawarharda & Urasawa, 1992). Moreover, lympocyte counts, total T cells, and suppressor cells have been found to be reduced with left hemisphere resections but increased after right hemisphere resections (Meador et al., 2009). These results indicate the immune functioning is more severely effected with left hemisphere damage.
Hence, whereas those with right cerebral lesions are likely to remain healthy and then die, those with left hemisphere lesions are more likely to get sick and then live. Although these effects are obviously due to hemispheric differences in the control over the immune system, exactly what the responsible mechanisms might be is currently unknown. However, perhaps these differences are due to right vs left hemisphere emotional reactions to injury. As noted, with left hemisphere lesions, patients develop apathy, depression, emotional blunting, and schizophrenia, although euphoria sometimes accompanies receptive aphasia and loss of comprehension (Gainotti, 2001; Gasparrini et al., 1978; Geschwind, 1965; Gruzelier & Manchanda, 1982; Hillbom, 1960; Robinson & Szetela, 2011; Robinson et al. 2014; Sherwin et al. 1982; Sinyour et al., 1986), whereas with right hemisphere injuries they may become indifferent. Indifference may promote immune reactions, whereas depression and apathy may interfere. On the other hand, depression and apathy may be yet another symptom associated with depressed immune functioning. Or it could be that the patients reported above, i.e. geriatric and those requiring surgical resection, have brains that are so different from the norm, that these findings cannot be generalized.
In any case, although there is evidence of considerable functional overlap as well as interhemispheric cooperation on a number of tasks, it certainly appears that the mental system maintained by the right cerebral hemisphere is highly developed, social-emotional, bilateral, and in many ways dominant over the temporal-sequential, language-dependent half of the cerebrum. Indeed, the right cerebrum can independently recall and act on certain memories with purposeful intent; is the dominant source of our dreams, psychic conflicts and desires, and is fully capable of motivating, initiating as well as controlling behavioral expression --often without the aid or even active (reflective) participation of the left half of the brain.











